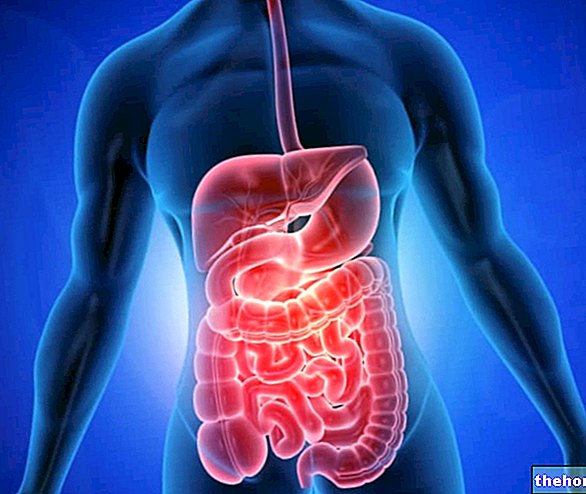
PROTÉASES (ou peptidases) : enzymes hydrolytiques impliquées dans la digestion des protéines. Par leur action, les protéases sont capables de rompre les liaisons peptidiques qui unissent les différents acides aminés et dont proviennent l'enchaînement répété des molécules protéiques.

SOURCE
- Pepsine : produite par les cellules peptiques de l'estomac sous une forme inactive (pepsinogène). Activé par l'acide chlorhydrique, il intervient principalement sur les liaisons peptidiques qui engagent les acides aminés aromatiques (tels que la tyrosine, le tryptophane et la phénylalanine).
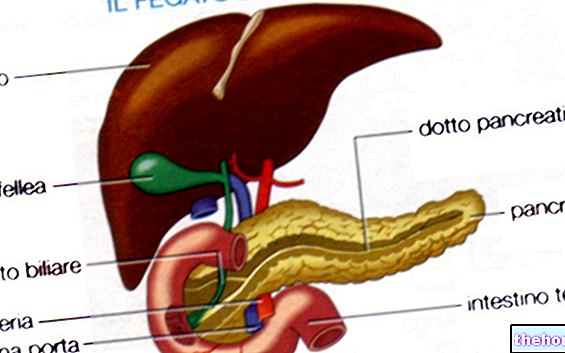
- Trypsine : produite par le pancréas exocrine sous une forme inactive (trypsinogène). Activé par l'entéropeptidase duodénale, il intervient principalement sur les liaisons peptidiques qui engagent les acides aminés basiques (tels que l'arginine et la lysine)
- Chymotrypsine : produite par le pancréas exocrine sous une forme inactive (chymotrypsinogène). Activé par la trypsine. Il intervient principalement sur les liaisons peptidiques qui engagent les acides aminés aromatiques (tels que la tyrosine, le tryptophane et la phénylalanine).
- Élastase : produite par le pancréas exocrine sous une forme inactive (pré-pro-élastase). Activé par la trypsine. La seule enzyme capable d'attaquer l'élastine et donc très importante pour la digestion des aliments carnés.
- Carboxypeptidase : produite et sécrétée par le pancréas exocrine en partie sous forme active et en partie sous forme inactive. Ils interviennent sur les liaisons peptidiques placées en bout carboxyle de la chaîne d'acides aminés.
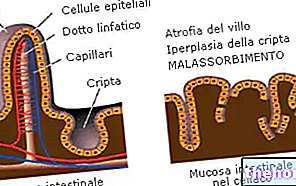
- Aminopeptidases : produites et sécrétées par la muqueuse duodénale. Ils interviennent sur les liaisons peptidiques placées en bout carboxyle de la chaîne d'acides aminés.
- Les dipeptidases : sécrétées par les entérocytes de l'intestin grêle, elles hydrolysent la liaison peptidique qui maintient ensemble les paires d'acides aminés
Les protéases utilisées pour la digestion des protéines d'origine alimentaire ne représentent qu'une petite partie de la grande famille à laquelle elles appartiennent. Si l'on pense que les enzymes, comme de nombreuses hormones, sont des molécules de nature protéique, on se rend compte à quel point le rôle joué par les protéases est important. Quoi de mieux pour réguler l'activité de ces molécules que de les dégrader si nécessaire au moyen de protéases spécifiques ? Voici donc que les cellules du système immunitaire contiennent des protéases pour digérer la membrane cellulaire des micro-organismes étrangers, qui à leur tour (on parle de bactéries) ont d'une part la capacité de sécréter des enzymes protéolytiques pour envahir la cellule et d'autre part de libérer des toxines protéiques (exotoxines) contre lesquelles il faut se défendre. Certaines protéases plasmatiques, comme l'antithrombine III et la plasmine, jouent un rôle important dans la coagulation, empêchant une « activation excessive de ce mécanisme, qui est au contraire renforcé par la thrombine (appartenant également à la grande famille des protéases).
L'action modulatrice des protéases est donc fondamentale dans la régulation de diverses fonctions corporelles ; si, par exemple, des protéines anormales proviennent de la synthèse des protéines, il est très important qu'elles soient dégradées le plus tôt possible. croître à l'infini, mais elle doit être limitée par des protéases spécifiques.En outre, si l'on considère que les protéases dégradent et détruisent les molécules protéiques, il faut s'attendre à ce qu'il existe des facteurs, y compris d'autres protéases, capables de réguler leur fonction et ainsi empêcher les phénomènes protéolytiques exaltés finissent par "causer des dommages aux tissus de" l'organisme.
Les phénomènes protéolytiques affectant les fibres musculaires sont activés par le jeûne et l'exercice physique prolongé afin de produire de l'énergie et du glucose à partir de certains acides aminés glucogéniques.
Les protéases végétales sont également synthétisées par les plantes afin de se défendre contre les agents pathogènes, dégrader les peptides et modifier la structure et la fonction d'autres protéines. La tige d'ananas (bromélaïne), la papaye (papain) et les graines germées (malt d'orge) en sont particulièrement riches. L'ingestion de ces aliments ou de leurs extraits aide à réguler la fonction digestive, en aidant l'action des protéases endogènes libérées dans le tube digestif.