Qu'est-ce que Hetlioz -Tasimelteon et à quoi sert-il ?
Hetlioz est un médicament indiqué dans le traitement du syndrome d'altération du cycle veille-sommeil (syndrome "non-24") chez l'adulte aveugle. Le syndrome non-24 est une affection qui affecte presque exclusivement les personnes aveugles, qui ont un cycle veille-sommeil qui n'est pas synchronisé avec le jour et la nuit, souvent plus long que le cycle normal de 24 heures. En conséquence, les patients s'endorment et se réveillent à des heures inhabituelles.
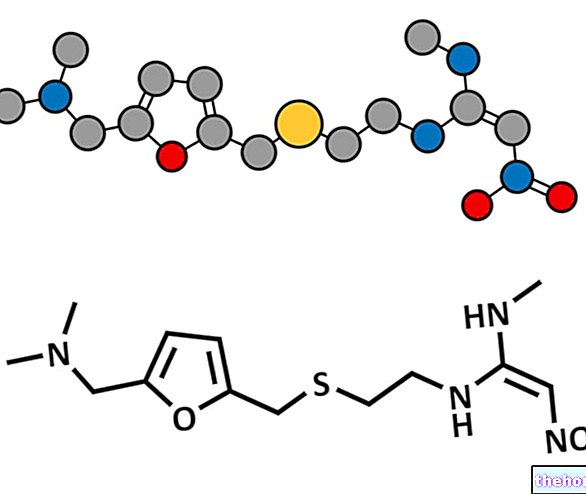
Hetlioz contient le principe actif tasimeltéon.
Étant donné que le nombre de patients atteints du syndrome non-24 est faible, la maladie est considérée comme « rare », et Hetlioz a été désigné « médicament orphelin » (un médicament utilisé dans les maladies rares) le 23 février 2011.
Comment Hetlioz-Tasimelteon est-il utilisé ?
Hetlioz est disponible sous forme de gélules (20 mg) et n'est délivré que sur ordonnance.
Hetlioz est destiné à un usage prolongé, la dose recommandée est d'une gélule par jour, à prendre une heure avant le coucher, à la même heure chaque soir. Le médicament doit être pris à jeun.
Comment fonctionne Hetlioz-Tasimelteon ?
Une hormone appelée mélatonine joue un rôle fondamental dans la coordination du cycle de sommeil du corps.Chez les personnes ayant une perception normale de la lumière et de l'obscurité, la mélatonine est produite pendant les heures d'obscurité et favorise le sommeil en agissant sur les récepteurs de la mélatonine présents dans des zones spécifiques. L'ingrédient d'Hetlioz, le tasimelteon, agit sur les mêmes récepteurs que la mélatonine pour stimuler le sommeil et réguler le rythme du sommeil. Pris chaque jour à une heure appropriée, il peut aider à rétablir un cycle veille-sommeil plus normal.
Quel bénéfice Hetlioz-Tasimelteon a-t-il démontré au cours des études ?
Hetlioz s'est avéré efficace pour « aider les patients à s'adapter à leur rythme de sommeil normal de 24 heures » dans 2 études principales.
La première étude, portant sur un total de 84 patients aveugles atteints du syndrome non-24, a comparé Hetlioz à un placebo (un traitement fictif). Le principal critère d'évaluation de l'efficacité était le pourcentage de patients capables de s'adapter au rythme veille-sommeil sur 24 heures, calculé en observant les modifications du métabolisme de la mélatonine dans l'urine des patients au fil du temps. Vingt pour cent des patients traités par Hetlioz (8 sur 40) ont pu s'adapter au rythme veille-sommeil de 24 heures après 1 mois de traitement, contre environ 3 % des patients traités par placebo (1 sur 38). De meilleurs résultats ont été observés dans un sous-ensemble de patients après 7 mois de traitement, ce qui suggère que les patients peuvent prendre des semaines ou des mois pour répondre au traitement.
Dans la seconde étude, 57 patients ont été initialement traités par Hetlioz pendant environ 11 semaines. Les patients qui ont pu s'adapter au rythme veille-sommeil sur 24 heures (20 au total) ont ensuite été traités par Hetlioz ou un placebo pendant 8 semaines supplémentaires, pour voir si l'effet d'Hetlioz se maintenait dans le temps. ont continué à recevoir le médicament, à la fin de l'étude, 9 personnes avaient maintenu leur rythme veille-sommeil normal sur 24 heures contre 2 des 10 patients traités par placebo.
Quel est le risque associé à Hetlioz-Tasimelteon ?
Les effets indésirables les plus fréquents avec Hetlioz (pouvant affecter plus de 3 personnes sur 100) sont les maux de tête, la somnolence, les nausées et les vertiges. Ces effets sont généralement légers à modérés et de nature transitoire.
Pour la liste complète des effets indésirables et des limitations rapportés avec Hetlioz, voir la notice.
Pourquoi Hetlioz -Tasimelteon a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) de l'Agence a estimé que les bénéfices d'Hetlioz sont supérieurs à ses risques et a recommandé qu'il soit approuvé pour une utilisation dans l'UE.Le CHMP a noté que seulement environ 20% des patients pourraient bénéficier d'Hetlioz Cependant, étant donné l'absence de thérapies approuvées pour le syndrome non-24, qui est une maladie débilitante, cette réponse, bien que modeste, a été considérée comme importante. Cependant, un traitement prolongé est nécessaire pour maintenir les effets bénéfiques du médicament. En ce qui concerne la sécurité, il a été démontré que Hetlioz est bien toléré, ne provoquant que quelques effets secondaires bénins.
Quelles mesures sont prises pour garantir une utilisation sûre et efficace de Hetlioz-Tasimelteon ?
Un plan de gestion des risques a été élaboré pour s'assurer que Hetlioz est utilisé de la manière la plus sûre possible. Sur la base de ce plan, des informations de sécurité ont été ajoutées au résumé des caractéristiques du produit et à la notice d'Hetlioz, y compris les précautions appropriées à suivre par les professionnels de santé et les patients.
Plus d'informations sur Hetlioz-Tasimelteon
Le 3 juillet 2015, la Commission européenne a délivré une "Autorisation de mise sur le marché" pour Hetlioz, valable dans toute l'Union européenne.
Pour plus d'informations sur le traitement par Hetlioz, veuillez lire la notice (jointe à l'EPAR) ou contacter votre médecin ou votre pharmacien.
Dernière mise à jour de ce résumé : 07-2015.
Les informations sur Hetlioz -Tasimelteon publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.