Qu'est-ce que Victoza ?
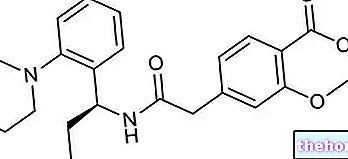
Victoza est une solution injectable qui contient le principe actif liraglutide, disponible en stylos préremplis (6 mg/ml).
A quoi sert Victoza ?
Victoza est utilisé chez les patients adultes atteints de diabète de type 2 (diabète non insulino-dépendant) pour contrôler le taux de glucose (sucre) dans le sang. Victoza est utilisé en association avec :
• la metformine ou un sulfamide hypoglycémiant (médicaments antidiabétiques), chez les patients dont le contrôle glycémique est insuffisant malgré la dose maximale tolérée de metformine ou de sulfamide hypoglycémiant seul ;
• la metformine et un sulfamide hypoglycémiant ou la metformine et une thiazolidinedione (un autre groupe de médicaments antidiabétiques) chez les patients dont le contrôle glycémique est insuffisant malgré une bithérapie.
Le médicament ne peut être obtenu que sur ordonnance
Comment Victoza est-il utilisé ?
Victoza est administré une fois par jour et peut être injecté par voie sous-cutanée (sous la peau) dans l'abdomen, la cuisse ou le haut du bras.Le médicament peut être pris à tout moment, indépendamment des repas, de préférence à la même heure tous les jours.
La dose initiale de Victoza est de 0,6 mg. Après au moins une semaine, la dose doit être augmentée à 1,2 mg. Pour certains patients, la dose peut encore être augmentée à 1,8 mg après une semaine supplémentaire pour obtenir un meilleur contrôle de la glycémie.
Dans les cas où Victoza est ajouté à un traitement existant par la metformine ou la thiazolidinedione, il n'est pas nécessaire de varier les doses de ces médicaments. Cependant, si Victoza est ajouté à un traitement par un sulfamide hypoglycémiant, il appartiendra au médecin de décider s'il faut ou non diminuer la dose du sulfamide hypoglycémiant pour réduire le risque d'hypoglycémie (faible taux de sucre dans le sang).
Comment fonctionne Victoza ?
Le diabète de type 2 est une maladie dans laquelle le pancréas ne produit pas suffisamment d'insuline pour contrôler le taux de glucose (sucre) dans le sang, ou lorsque le corps est incapable d'utiliser l'insuline efficacement. Le principe actif de Victoza, le liraglutide, est un « mimétique des incrétines » : il agit de la même manière que les incrétines (hormones produites dans l'intestin) en augmentant la quantité d'insuline libérée par le pancréas en réponse à la nourriture. Cela aide à contrôler la nourriture. les niveaux de glucose dans le sang.
Comment Victoza a-t-il été étudié ?
Les effets de Victoza ont d'abord été testés dans des modèles expérimentaux avant d'être étudiés chez l'homme.
Le médicament a fait l'objet de cinq études principales portant sur 3 978 adultes diabétiques de type 2, à savoir :
• une étude en « monothérapie » comparant Victoza, administré seul, au glimépiride (une sulfonylurée) ;
• deux études de « bithérapie » comparant Victoza en association avec la metformine ou Victoza en association avec le glimépiride avec la metformine ou le glimépiride pris avec un placebo (un traitement fictif) ;
• deux études de « trithérapie » comparant Victoza en association avec la metformine et le glimépiride ou la rosiglitazone (une thiazolidinedione), avec des traitements incluant un placebo ou un autre médicament contre le diabète au lieu de Victoza.
Le principal critère d'évaluation de l'efficacité était la modification de la quantité d'une substance dans le sang appelée hémoglobine glycosylée (HbA1c) après six mois ou un an. "L'HbA1c offre une indication de la qualité du contrôle de la glycémie."
Quel bénéfice Victoza a-t-il démontré au cours des études ?
Les combinaisons incluant Victoza se sont avérées plus efficaces pour contrôler la glycémie que les combinaisons où le médicament n'apparaissait pas. Les bithérapies, contenant du Victoza et de la metformine ou un sulfamide hypoglycémiant, ont permis des réductions de l'HbA1c d'environ 1%, alors qu'aucune réduction n'a été enregistrée en l'absence de Victoza.Les trithérapies comprenant la metformine et un sulfamide hypoglycémiant ou une thiazolidinedione une réduction de 1,3-1,5% par rapport à une réduction de 0,5 % ou moins sans Victoza autorisé. En monothérapie, une réduction plus importante de l'HbA1c a également été observée avec Victoza qu'avec le glimépiride. Cependant, l'étude n'était pas suffisante pour soutenir l'utilisation de Victoza seul.
Quel est le risque associé à Victoza ?
Les effets indésirables les plus fréquemment observés avec Victoza, utilisé en association avec d'autres médicaments antidiabétiques (chez plus de 1 patient sur 10) sont l'hypoglycémie (faible glycémie), les maux de tête, les nausées et la diarrhée. Pour la liste complète des effets indésirables rapportés avec Victoza, voir la notice.
Victoza ne doit pas être utilisé chez les personnes pouvant présenter une hypersensibilité (allergie) au liraglutide ou à l'un des autres composants.
Pourquoi Victoza a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) a estimé que les bénéfices de Victoza sont supérieurs à ses risques pour le traitement des adultes atteints de diabète de type 2 pour obtenir un contrôle glycémique en bithérapie, en association avec la metformine ou un sulfamide hypoglycémiant. - un traitement médicamenteux en association avec la metformine et un sulfamide hypoglycémiant ou une thiazolidinedione. Le comité a donc recommandé l'octroi d'une autorisation de mise sur le marché pour Victoza.
Autres informations sur Victoza :
Le 30 juin 2009, la Commission européenne a accordé à Novo Nordisk A/S une « autorisation de mise sur le marché » pour Victoza, valable dans toute l'Union européenne.
Pour la version complète de l'EPAR de Victoza, cliquez ici.
Dernière mise à jour de ce résumé : 05-2009.
Les informations sur Victoza - liraglutide publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.