ONGLYZA ® un médicament à base de saxagliptine
GROUPE THÉRAPEUTIQUE : Agents hypoglycémiants oraux - Inhibiteurs de la DPP-4

Indications ONGLYZA ® - Saxagliptine
ONGLYZA ® est utilisé comme aide pharmacologique dans le diabète de type 2, en complément de la metformine, des sulfonylurées et des agonistes du PPAR gamma, lorsque les monothérapies n'ont pas produit l'effet thérapeutique souhaité.
Mécanisme d'action ONGLYZA ® - Saxagliptine
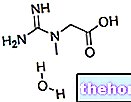
La saxagliptine, principe actif d'ONGLYZA ® est un inhibiteur sélectif de la DPP-4, enzymes responsables de la dégradation irréversible des incrétines (GLP-1 et GIP), hormones gastro-intestinales capables de moduler l'activité pancréatique, augmentant l'activité des cellules bêta, entraînant par une augmentation des concentrations d'insuline, et une sensibilisation des alpha, avec une diminution de la sécrétion de glucagon, responsable de la production endogène de glucose.
Du point de vue métabolique, les deux effets se traduisent par une baisse significative des taux glycémiques, donnée par le doublement des concentrations sanguines d'incrétine, induite par la saxagliptine.
Pris par voie orale, cet ingrédient actif atteint sa concentration plasmatique maximale vers la troisième heure, exerçant l'action inhibitrice de la DPP-4 pendant environ 24 heures et est ensuite métabolisé également dans le foie en métabolites modérément actifs, éliminés principalement par l'urine.
Des variations modestes de l'exposition médicamenteuse ont été observées chez des patients présentant une insuffisance hépatique et rénale, pour lesquels une surveillance médicale est strictement nécessaire.
Études réalisées et efficacité clinique
1. L'EFFICACITÉ DE LA THÉRAPIE COMBINÉE
Diabète Obes Métab. 22 février 2011. doi: 10.1111 / j.1463-1326.2011.01385.x.
Le traitement combiné initial avec la saxagliptine et la metformine permet un contrôle glycémique soutenu et est bien toléré jusqu'à 76 semaines.
Pfützner A, Paz-Pacheco E, Allen EM, Frederich B, Chen R.
Le traitement du patient diabétique de deuxième type avec une thérapie combinée saxagliptine/metformine a assuré un excellent contrôle glycémique, réduisant les concentrations d'hémoglobine glycosylée d'environ 2 points de pourcentage en 76 semaines de traitement, sans effets secondaires cliniquement pertinents. Les résultats obtenus étaient bien meilleurs que les monothérapies.
2. QUEL INHIBITEUR UTILISER ?
Diabetes Res Clin Pract. 2010 novembre ; 90 : 131-40. Publication en ligne du 13 août 2010.
Inhibiteurs de la DPP-4 : quels peuvent être les différenciateurs cliniques ?
Gerich J.
Le développement récent de nombreux principes actifs capables d'inhiber l'enzyme DPP-4, et l'expérimentation même de nouveaux, nécessitent un contrôle et des algorithmes utiles au médecin dans la prescription correcte du médicament.Il est donc important qu'il y ait guides de lignes, qui peuvent aider le spécialiste à formuler le bon plan thérapeutique, à protéger la santé du patient et à augmenter l'efficacité de la thérapie.
3. LA SAXAGLIPTINE, PAS SEULEMENT HYPOGLYCÉMISANTE
Diabète Metab Syndr Obes. 10 mai 2010, 3 : 117-24.
Nouveaux traitements dans la prise en charge du diabète de type 2 : un bilan critique de la saxagliptine.
Gallwitz B.
Le traitement de la pathologie diabétique de second type par la saxagliptine s'est avéré utile non seulement pour améliorer certains marqueurs du métabolisme du glucose tels que l'hémoglobine glycosylée et la glycémie à jeun et postprandiale, mais également pour moduler et réduire le risque cardiovasculaire.
Mode d'emploi et posologie
ONGLYZA® Comprimés de saxagliptine 5 mg :
la posologie recommandée est d'un comprimé que, pris indépendamment des repas, en association avec la metformine, les thiazolidinediones ou les sulfonylurées.
Bien que la posologie susmentionnée soit la plus recommandée, il est d'une importance fondamentale que votre médecin organise le bon plan thérapeutique en fonction des caractéristiques physiopathologiques du patient, de sa situation métabolique et de la prise éventuelle d'autres médicaments.
Mises en garde ONGLYZA ® - Saxagliptine
Le traitement pharmacologique avec ONGLYZA ® doit être soutenu et accompagné de mesures non pharmacologiques telles qu'une alimentation équilibrée et un mode de vie sain, utiles pour lutter contre la pathologie diabétique.
Une surveillance médicale étroite est nécessaire chez les patients insuffisants rénaux ou hépatiques, traités par sulfamides hypoglycémiants, compte tenu du risque d'hypoglycémie, et chez les patients âgés.
ONGLYZA® contient du lactose, par conséquent l'administration chez les patients présentant un déficit en enzyme lactase, une intolérance au lactose ou un déficit d'absorption du glucose/galactose, pourrait s'accompagner de la survenue concomitante d'effets secondaires concentrés notamment au niveau gastro-intestinal.
Le risque d'hypoglycémie, particulièrement évident en cas de prise concomitante de sulfonylurées, pourrait rendre dangereux l'utilisation de machines et la conduite de véhicules à moteur.
LA GROSSESSE ET L'ALLAITEMENT
L'utilisation d'ONGLYZA ® n'est pas recommandée dans le traitement du diabète gestationnel, compte tenu de l'absence d'études cliniques capables de tester le profil de sécurité du médicament sur la santé du fœtus, et compte tenu de la possibilité d'utiliser des médicaments mieux caractérisés.
ONGLYZA ® ne doit pas non plus être pris dans la phase de lactation ultérieure, étant donné la possibilité de retrouver le principe actif dans le lait maternel, potentiellement dangereux pour la santé du nourrisson.
Interactions
Bien que le nombre d'interactions cliniquement significatives soit faible, des modifications pharmacocinétiques affectant la saxagliptine sont décrites suite à la prise concomitante de diltiazem, de kétoconazole et de rifampicine, des médicaments puissants modulant le système enzymatique CYP3A4, responsable du métabolisme de la saxagliptine.
Contre-indications ONGLYZA ® - Saxagliptine
ONGLYZA® est contre-indiqué en cas d'hypersensibilité à la substance active ou à l'un de ses composants et pendant la période d'allaitement et de grossesse.
Ce médicament ne doit pas être utilisé pour le traitement des patients diabétiques de premier type ou des patients diabétiques céto acidosidiabiques, et encore moins chez les patients présentant une insuffisance hépatique et rénale.
Effets indésirables - Effets secondaires
Les données relatives au profil de tolérance de la saxagliptine sont essentiellement issues de l'essai clinique qui a testé ce principe actif sur plus de 3000 patients.
Bien que le traitement ait été bien toléré, parmi les effets indésirables les plus fréquents, des infections affectant les voies respiratoires supérieures, des maux de tête et des vertiges, des vomissements, de la diarrhée et de la constipation, et un œdème périphérique ont été observés à la fois en monothérapie et en association.
Des tableaux cliniques plus graves n'ont été observés que dans de rares cas, et chez des patients hypersensibles à la substance active, tandis que des épisodes d'hypoglycémie ont été principalement décrits suite à la prise combinée de saxagliptine et de glibenclamide.
Noter
ONGLYZA ® ne peut être commercialisé que sur prescription médicale stricte.
Les informations sur ONGLYZA ® - Saxagliptine publiées sur cette page peuvent être périmées ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.








-propriet-nutrizionali.jpg)