JANUMET ® un médicament à base de sitagliptine et de metformine
GROUPE THÉRAPEUTIQUE : Agents hypoglycémiants oraux associés

Indications JANUMET ® - Sitagliptine + Metformine
JANUMET ® est utilisé dans le traitement du diabète de type II, lorsque la metformine seule, même à doses maximales, n'a pas pu garantir un bon contrôle glycémique.
Chez les patients présentant une décompensation métabolique sévère, JANUMET ® peut être inclus dans une trithérapie par sulfamide hypoglycémiant.
Mécanisme d'action JANUMET ® - Sitagliptine + Metformine
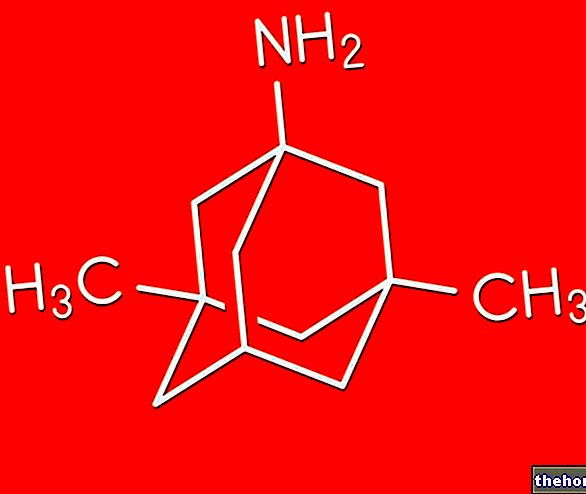
JANUMET ® représente un hypoglycémiant oral, obtenu à partir de l'association de principes actifs appartenant à la catégorie des inhibiteurs de la DPP-4 et des biguanides.
L'effet thérapeutique, qui s'effectue grâce à un contrôle intégré du métabolisme du glucose, est médié par la sitagliptine, capable d'augmenter la disponibilité des incrétines (réduction de l'activation de la DPP-4, impliquée dans l'hydrolyse irréversible), hormones responsables de la sensibilisation du pancréas. cellule bêta utile pour assurer la libération d'insuline, et de metformine, capable d'agir sur divers tissus sensibles à l'insuline en augmentant la captation du glucose sanguin, et dans le foie en inhibant le processus de gluconéogenèse et de glycogénolyse.
D'un point de vue métabolique, donc, l'effet hypoglycémiant est soutenu d'une part par l'augmentation de la sécrétion d'insuline et d'autre part par la diminution de la production endogène de glucose.
Du point de vue pharmacocinétique, la prise de JANUMET ® maintient les caractéristiques des deux principes actifs pris individuellement quasiment inchangées.
Études réalisées et efficacité clinique
1. L'EFFICACITÉ DE LA THÉRAPIE COMBINÉE SITAGLIPTINE / METFORMINE
Médicaments. 12 février 2011 ; 71 : 349-61.
Association à dose fixe sitagliptine/metformine : chez les patients diabétiques de type 2.
Chwieduk CM.
Des études ont montré que l'association de la metformine et de la sitagliptine pouvait assurer un bon contrôle glycémique chez les patients atteints de diabète sucré de type II, pour lesquels la metformine seule était inefficace. Dans ces cas, une réduction supplémentaire de la glycémie a été observée. et le contrôle glycémique postprandial et un risque réduit d'hypoglycémie.
2. SITAGLIPTINE / METFORMINE, AU-DELÀ DE LA PRATIQUE CLINIQUE
Diabetes Res Clin Pract. 21 février 2011
Effet de la sitagliptine et de la metformine sur la fonction des cellules , l'intégrité des îlots et l'expression des gènes des îlots chez les rats gras diabétiques Zucker.
Han SJ, Choi SE, Kang Y, Jung JG, Yi SA, Kim HJ, Lee KW, Kim DJ.
Les succès thérapeutiques importants que l'association entre la metformine et la sitagliptine a obtenus en pratique clinique semblent essentiellement liés aux effets thérapeutiques uniques des deux principes actifs.Cependant, des études in vitro ont montré comment cette association peut agir en synergie sur la cellule bêta, en préservant fonction et intégrité, probablement en activant des gènes anti-apoptotiques impliqués dans la survie cellulaire.
3. SITAGLIPTINE / METFORMINE EN UNE SEULE FORMULATION
Clin Drug Investig. 2010 ; 30 : 855-66.
Bioéquivalence des comprimés de l'association fixe sitagliptine/metformine et administration concomitante de sitagliptine et de metformine chez des sujets adultes sains : étude randomisée, en ouvert, croisée.
Migoya EM, Miller JL, Gutierrez M, Zheng W, Johnson-Levonas AO, Liu Q, Matthews CZ, Wagner JA, Gottesdiener KM.
Des études pharmacocinétiques et des essais cliniques de divers types ont conclu que l'effet thérapeutique observé après la prise de JANUMET, peut être tout à fait comparable à celui obtenu avec l'administration séparée mais simultanée de metformine et de sitagliptine, avec la même fréquence d'effets secondaires.
Mode d'emploi et posologie
JANUMET® Comprimés à 50/850 ou 50/1000 mg de sitagliptine et de metformine :
le traitement par JANUMET ® doit commencer à partir de la dose administrable la plus faible, puis éventuellement corriger ou valider la posologie susmentionnée, en fonction du résultat métabolique observé en termes de glycémie.
La dose maximale ne doit pas dépasser celle de 100 mg de sitagliptine, éventuellement prise en deux administrations différentes en même temps que les repas pour réduire les effets secondaires gastro-intestinaux.
En cas d'utilisation d'une trithérapie par sulfamides hypoglycémiants, des posologies inférieures à celles prévues pourront également être utilisées.
Dans tous les cas, il appartient à votre médecin d'évaluer et de formuler la posologie appropriée.
Mises en garde JANUMET ® - Sitagliptine + Metformine
Il est très important d'assurer un bon contrôle glycémique, que le traitement par JANUMET ® s'accompagne d'une surveillance périodique de la glycémie et de l'application concomitante de mesures non pharmacologiques telles que l'activité physique et une alimentation équilibrée.
Il est également nécessaire de surveiller la fonction rénale, pour éviter que l'accumulation de metformine n'induise chez le patient un état d'acidose métabolique potentiellement létale.
Le traitement par JANUMET ® doit être suspendu dans les cas où l'utilisation de produits de contraste ou de médicaments iodés ou de procédures qui compromettent la fonction rénale est requise.
Des conditions hypoglycémiques, potentiellement vérifiables suite à des dosages incorrects, à l'administration d'ingrédients actifs ou à d'autres causes, pourraient réduire les capacités de perception et de réaction du patient, rendant l'utilisation de machines et la conduite de voitures dangereuses.
LA GROSSESSE ET L'ALLAITEMENT
L'utilisation de JANUMET ® pendant la grossesse est fortement contre-indiquée, en raison de la présence d'études montrant un risque accru de malformations congénitales chez les nouveau-nés accouchés de femmes traitées par metformine.
Au contraire, l'expérimentation avec la sitagliptine n'a pas donné de résultats statistiquement significatifs pour le moment.
De plus, la sécrétion éventuelle de ces principes actifs dans le lait maternel expose le nourrisson à un risque potentiel d'hypoglycémie, au point d'étendre la contre-indication également à la période de lactation.
Interactions
Dans ce cas également, les interactions médicamenteuses observées pour JANUMET ® sont à attribuer de manière distincte à la présence des deux principes actifs.
Par conséquent, alors que la sitagliptine est peu réactive, avec des modifications pharmacocinétiques observées après l'administration de digoxine et de cyclosporine, la metformine peut répondre négativement à l'administration concomitante de glucocorticoïdes, de bêta-agonistes, de cimétidine et de diurétiques.
Plusieurs études ont également évalué la présence d'autres interactions possibles in vitro, qui revêtent cependant une pertinence clinique modeste in vivo.
Il est également utile de se rappeler que l'administration concomitante de rifampicine ou de produits de contraste iodés pourrait altérer la fonction rénale normale, augmentant l'exposition de l'organisme à ces médicaments hypoglycémiants.
Contre-indications JANUMET ® - Sitagliptine + Metformine
JANUMET® est contre-indiqué en cas d'hypersensibilité aux principes actifs ou aux excipients, diabète de type I, acidocétose diabétique, modifications sévères de la fonction hépatique et rénale, déshydratation, choc, pathologies aiguës, insuffisance cardiaque et respiratoire, choc, alcoolisme, grossesse et allaitement . . .
Effets indésirables - Effets secondaires
Les effets secondaires décrits à la suite d'un traitement par JANUMET ® sont essentiellement attribuables à ceux observés lors d'une monothérapie par la sitagliptine et la metformine.
En particulier, la phase initiale du traitement peut s'accompagner de troubles gastro-intestinaux tels que nausées, vomissements, constipation ou diarrhée, infections des voies respiratoires supérieures, maux de tête et vertiges.
Des effets indésirables cliniquement plus significatifs n'ont été observés que rarement, avec l'apparition de réactions d'hypersensibilité telles qu'œdème de Quincke, rash et urticaire, des modifications du tableau hématologique et du tracé électrocardiographique, pour lesquelles il a été nécessaire d'interrompre le traitement.
En revanche, les troubles hypoglycémiques ont été retrouvés surtout au cours d'une trithérapie par sulfamides hypoglycémiants.
Noter
JANUMET ® ne peut être vendu que sur prescription médicale stricte
Les informations sur JANUMET ® - Sitagliptine + Metformine publiées sur cette page peuvent être périmées ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.