LEVEMIR ® un médicament à base d'insuline detemir
GROUPE THÉRAPEUTIQUE : Insuline détémir à usage injectable - insulines et analogues

Indications LEVEMIR ® - Insuline detemir
LEVEMIR ® est indiqué dans le traitement des patients diabétiques nécessitant une insulinothérapie pour rétablir un équilibre glycémique correct.
Mécanisme d'action LEVEMIR ® - Insuline detemir
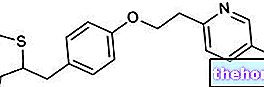
LEVEMIR ® est un médicament à base d'insuline détémir, un analogue de l'insuline obtenu en substituant la thréonine en position B30 et en associant la lysine en position B29, un acide gras à 14 atomes de carbone appelé acide myristique.
Ces deux variations importantes permettent respectivement de lier l'albumine plasmatique de manière réversible, rendant progressive l'action activatrice de l'hormone sur les récepteurs de l'insuline et facilitant en même temps la formation d'hexamères dans la couche sous-cutanée qui retardent l'absorption de cet analogue de l'insuline.
Ces types de particularités rendent l'insuline detemir plus stable du point de vue pharmacocinétique, réduisant significativement les variations intra et interindividuelles, et reproduisant une cinétique d'action plus ou moins similaire chez les différents patients, ce qui est capable de manière dose-dépendante de prolonger la " effet biologique jusqu'à 24 heures après administration sous-cutanée.
Cependant, les variations structurelles importantes permettent également à l'insuline detemir de médier son effet hypoglycémiant par l'activation des récepteurs de l'insuline exprimés par les tissus sensibles à l'insuline et en même temps l'inhibition de la production et de la libération de glucose par le foie, d'une manière complètement identique à ce qu'il fait l'hormone endogène.
Études réalisées et efficacité clinique
1. L'INSULINE DETEMIR DANS L'ÂGE PÉDIATRIQUE
Pédiatre Diabète. 21 mars 2011
Traitement par insuline détémir ou insuline NPH chez les enfants âgés de 2 à 5 ans atteints de diabète de type 1.
Thalange N, Bereket A, Larsen J, Hiort LC, Peterkova V.
L'administration d'insuline détémir chez des patients pédiatriques atteints de diabète sucré de type 1 a assuré un excellent contrôle glycémique, une réduction de la glycémie à jeun, une réduction des événements hypoglycémiques et une prise de poids réduite sur de courtes périodes de traitement. Ces résultats étaient significativement meilleurs que ceux obtenus avec un autre analogue d'insuline intermédiaire tel que l'insuline isophane.
2. EFFICACITÉ DE L'INSULINE DETEMIR
Diabète Technol Ther. 12 décembre 2010 : 1019-27.
Relation entre la dose d'insuline, l'abaissement de l'A1c et le poids dans le diabète de type 2 : comparaison de l'insuline glargine et de l'insuline détémir.
Dailey G, Admane K, Mercier F, Owens D.
Le traitement par insuline détémir chez les patients diabétiques de type II s'est avéré particulièrement utile pour assurer une réduction significative de l'hémoglobine glycosylée et de la glycémie à jeun, évitant la prise de poids observée suite à l'administration d'insuline glargine.
3. INSULINE THÉRAPIE ET AUGMENTATION DE POIDS
Prim Care Diabète. Avr 2010 ; 4 Suppl 1 : S24-30.
Diabète et gestion du poids.
Lau DC.
La prise de poids est l'une des manifestations les plus fréquentes présentes chez le patient diabétique qui est souvent accentuée par l'administration d'insuline en début de traitement.Parmi les différents analogues, l'insuline détémir est celle capable de déterminer la moindre prise de poids. Cependant, dans ces conditions, il serait conseillé d'associer un mode de vie sain et une alimentation adéquate.
Mode d'emploi et posologie
LEVEMIR® 100 UI/ml d'insuline détémir pour injection, stylos préremplis de 3 ml :
la posologie appropriée d'insuline détémir doit être définie par le médecin en fonction du type de traitement, de l'administration concomitante d'autres insulines, de l'état physiopathologique du patient et de ses concentrations glycémiques.
Dans la plupart des cas, l'insuline detemir est prise une à deux fois par jour par voie sous-cutanée par injections dans la cuisse, la paroi abdominale ou le deltoïde.
La zone d'administration différente affecte, comme on le sait, le moment de l'absorption du médicament.
Mises en garde LEVEMIR ® - Insuline detemir
La thérapie pharmacologique avec l'insuline, et en détail avec l'insuline detemir, doit prévoir la participation active du patient afin de maintenir un bon contrôle glycémique et d'éviter les effets secondaires désagréables.
En effet, le patient doit être suffisamment instruit sur les bonnes méthodes de conservation, de préparation et d'administration du médicament et doit être capable de reconnaître rapidement les signes avant-coureurs d'une hypoglycémie afin de recourir rapidement à des remèdes évitant l'apparition de réactions secondaires graves.
Cependant, une surveillance médicale est nécessaire tout au long du traitement thérapeutique avec une pertinence particulière dans les périodes pouvant nécessiter un ajustement thérapeutique telles que les états fébriles, les maladies infectieuses, la chirurgie, les changements d'habitudes alimentaires et de mode de vie.
L'apparition possible d'une hypoglycémie pourrait s'accompagner d'une diminution significative des capacités de perception du patient, rendant l'utilisation de machines et la conduite de voitures dangereuses.
LA GROSSESSE ET L'ALLAITEMENT
La commercialisation récente de l'insuline détémir n'a pas été suffisamment précédée d'études et d'essais cliniques capables d'évaluer l'impact de cet analogue de l'insuline sur la santé du fœtus lorsqu'il est pris pendant la grossesse.
Cependant, le profil d'innocuité sur les modèles animaux était excellent au même titre que les autres analogues.
Interactions
Comme d'autres analogues, l'insuline detemir peut interagir avec différents principes actifs en faisant varier ses propriétés pharmacocinétiques.
En effet, la prise simultanée d'agents hypoglycémiants oraux, d'octréotide, d'anti-MAO, d'agents bêta-bloquants, d'inhibiteurs de l'ECA, de salicylates, d'alcool et de stéroïdes anabolisants pourrait renforcer l'effet thérapeutique de LEVEMIR ®, augmentant significativement le risque d'hypoglycémie.
A l'inverse, l'administration concomitante de contraceptifs oraux, de thiazidiques, de glucocorticoïdes, d'hormones thyroïdiennes et de sympathomimétiques pourrait réduire l'effet hypoglycémiant de l'insuline détémir, nécessitant un éventuel ajustement de la posologie.
Il est également utile de se rappeler comment les sympatholytiques peuvent masquer certains signes importants d'hypoglycémie, augmentant le risque de réactions graves.
Contre-indications LEVEMIR ® - Insuline detemir
LEVEMIR ® est contre-indiqué en cas d'hypoglycémie et d'hypersensibilité à l'insuline humaine ou à ses excipients.
Effets indésirables - Effets secondaires
Le traitement à l'insuline detemir a les mêmes effets secondaires que les autres analogues de l'insuline.
En conséquence, une rougeur, une douleur, un œdème et des démangeaisons ou une lipoatrophie peuvent apparaître au site d'injection en raison de ne pas changer le site d'injection.
Des réactions systémiques telles que des difficultés respiratoires, un œdème et une hypotension peuvent être dues à une hypersensibilité à l'insuline ou à l'un de ses excipients.
Cependant, le principal effet indésirable décrit après l'insulinothérapie est représenté par l'hypoglycémie, qui peut se manifester par une pâleur, des sueurs froides, de la fatigue, une hypotension, une somnolence et qui peut conduire dans les cas graves à une perte de conscience et un coma.
Noter
LEVEMIR ® vendu uniquement sur prescription médicale.
LEVEMIR ® fait partie de la classe de dopage : hormones et substances apparentées (interdites en et hors compétition)
Les informations sur LEVEMIR ® - Insulin detemir publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.