Qu'est-ce que NovoRapid ?
NovoRapid est une solution injectable qui contient le principe actif insuline asparte. Il est disponible en flacons, cartouches (PenFill) et stylos préremplis (NovoLet, FlexPen et InnoLet).
A quoi sert NovoRapid ?
NovoRapid est utilisé pour traiter les adultes, les adolescents et les enfants de plus de 2 ans atteints de diabète.
Le médicament ne peut être obtenu que sur ordonnance.
Comment NovoRapid est-il utilisé ?
Novorapid est administré par injection sous la peau (sous la peau) dans la paroi abdominale (ventre), la cuisse, le bras, l'épaule ou la fesse. NovoRapid est une « insuline à action rapide. Normalement, elle est administrée immédiatement avant un repas, mais, si nécessaire, elle peut être administrée après un repas. NovoRapid est généralement utilisé avec une » insuline à action intermédiaire ou prolongée administrée au moins une fois. un jour. Pour trouver la dose efficace la plus faible, le patient doit faire contrôler régulièrement sa glycémie (sucre). La dose habituelle varie entre 0,5 et 1,0 U/kg/jour. Lorsqu'il est pris au cours d'un repas, 50 à 70 % des besoins en insuline sont fournis par NovoRapid et le reste par une insuline "d'action intermédiaire ou longue" NovoRapid peut être administré aux femmes enceintes.
NovoRapid peut également être utilisé avec un système de pompe à perfusion d'insuline en continu. Il peut être administré dans une veine, mais uniquement par un médecin ou une infirmière.
Comment fonctionne NovoRapid ?
Le diabète est une maladie dans laquelle le corps ne produit pas suffisamment d'insuline pour contrôler le taux de glucose dans le sang. NovoRapid est une « insuline de remplacement, qui est très similaire à l'insuline produite par le corps ». Le principe actif de NovoRapid, « l'insuline asparte, est produit par une méthode connue sous le nom de « technique de l'ADN recombinant », c'est-à-dire qu'il est fabriqué par une levure dans laquelle est inséré un gène (ADN) qui la rend capable de produire insuline asparte.
L'insuline asparte diffère très peu de l'insuline humaine ; grâce à cette différence, elle est absorbée plus rapidement par l'organisme, elle peut donc agir plus rapidement que l'insuline humaine. L'insuline de remplacement agit comme l'insuline produite naturellement et favorise la pénétration du glucose dans les cellules du sang.En contrôlant le taux de glucose dans le sang, les symptômes et les complications du diabète sont réduits.
Comment NovoRapid a-t-il été étudié ?
NovoRapid a été étudié dans deux études portant sur 1954 patients atteints de diabète de type 1 (dans lequel le pancréas est incapable de produire de l'insuline) et dans une étude portant sur 182 patients atteints de diabète de type 2 (dans laquelle le corps n'est pas capable de produire de l'insuline). pour utiliser efficacement l'insuline). Dans ces études, NovoRapid a été comparé à l'insuline humaine en mesurant le niveau dans le sang d'une substance appelée hémoglobine glycosylée (HbA1c) qui donne une indication du degré de contrôle de la glycémie. La sécurité de NovoRapid a également été comparée à celle de l'homme. l'insuline dans deux études portant sur 349 femmes enceintes atteintes de diabète de type 1 ou de diabète gestationnel (causé par la grossesse). NovoRapid n'a pas été testé chez les enfants de moins de deux ans.
Quel bénéfice NovoRapid a-t-il démontré au cours des études ?
NovoRapid a donné à peu près les mêmes résultats que l'insuline humaine. Dans les deux études sur le diabète de type 1, NovoRapid a réduit les taux d'HbA1c de 0,12 % et 0,15 %, respectivement, par rapport à l'insuline humaine après six mois. NovoRapid utilisé pendant la grossesse a présenté le même profil de sécurité que l'insuline humaine.
Quel est le risque associé à NovoRapid ?
L'effet indésirable le plus fréquent avec NovoRapid (observé chez 1 à 10 patients sur 100) est l'hypoglycémie (faible taux de glucose dans le sang). Pour la liste complète des effets indésirables rapportés avec NovoRapid, voir la notice.
NovoRapid ne doit pas être utilisé chez les personnes pouvant présenter une hypersensibilité (allergie) à l'insuline asparte ou à l'un des autres composants. De plus, les doses de NovoRapid peuvent devoir être ajustées lorsqu'il est administré avec d'autres médicaments pouvant avoir un effet sur le sang. glycémie La liste complète se trouve dans la notice.
Pourquoi NovoRapid a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) a décidé que les bénéfices de NovoRapid sont supérieurs à ses risques pour le traitement du diabète sucré. Le comité a recommandé l'autorisation de mise sur le marché de NovoRapid.
Plus d'informations sur NovoRapid
Le 7 septembre 1999, la Commission européenne a accordé à Novo Nordisk A/S une "Autorisation de mise sur le marché" pour NovoRapid, valable dans toute l'Union européenne. L'"Autorisation de mise sur le marché" a été renouvelée le 7 septembre 2004 et le 7 septembre 2009.
Pour la version complète de l'EPAR de NovoRapid, cliquez ici.
Dernière mise à jour de ce résumé : 09-2009.
Les informations sur NovoRapid - insuline aspart publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.

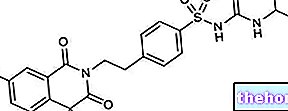
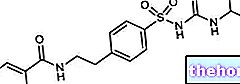




.jpg)