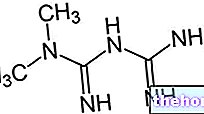
Ingrédients actifs : Nitroglycérine
TRINITRINA comprimés enrobés NITROGLYCÉRINE 0,3 mg
Les notices d'emballage de la trinitrine sont disponibles pour les tailles de conditionnement :- Comprimés enrobés de TRINITRINA NITROGLYCÉRINE 0,3 mg
- TRINITRINA 5mg/1,5ml solution à diluer pour perfusion
- TRINITRINE 50 mg/50 ml solution à diluer pour perfusion
Pourquoi Trinitrina est-il utilisé ? Pourquoi est-ce?
Groupe pharmacothérapeutique
Vasodilatateur utilisé dans les maladies cardiaques (C01DA02).
Indications thérapeutiques
Traitement de la crise aiguë d'angine de poitrine.
Contre-indications Quand la Trinitrine ne doit pas être utilisée
- choc
- choc cardiogénique, sauf si une pression diastolique aortique adéquate est maintenue
- état d'hypotension dans l'infarctus aigu du myocarde avec faible pression de remplissage
- hypotension sévère (pression artérielle systolique)
- hypovolémie sévère
- augmentation de la pression intracrânienne, traumatisme crânien et hémorragie cérébrale
- insuffisance myocardique due à une obstruction (par exemple en présence d'une sténose aortique ou mitrale ou d'une péricardite constrictive)
- cardiomyopathie hypertrophique obstructive
- oedème pulmonaire toxique
- anémie sévère
- glaucome à angle fermé
- généralement contre-indiqué pendant la grossesse et l'allaitement et à l'âge pédiatrique
- en association avec le sildénafil (voir rubrique "Interactions")
- hypersensibilité aux nitrates organiques et/ou à l'un des excipients
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Trinitrine
L'efficacité de la nitroglycérine prise par voie sublinguale chez les patients atteints d'insuffisance cardiaque congestive n'a pas été établie. Par conséquent, une surveillance clinique ou hémodynamique attentive doit être effectuée chez ces patients afin de détecter la possibilité d'hypotension et de tachycardie.
Une hypotension sévère, en particulier orthostatique, peut survenir après la prise même de petites doses de nitroglycérine. Par conséquent, le médicament doit être utilisé avec prudence chez les patients présentant une hypovolémie ou une hypotension préexistante. L'hypotension provoquée par la nitroglycérine peut s'accompagner d'une bradycardie paradoxale et d'une augmentation des douleurs angineuses.
Le traitement par nitrate peut aggraver l'angine chez les patients atteints de cardiomyopathie hypertrophique. À utiliser avec prudence chez les patients présentant une hypoxémie anémique sévère, car la biotransformation de la nitroglycérine est réduite chez ces patients.
Les comprimés enrobés de trinitrine doivent être arrêtés si des troubles visuels (vision trouble) ou une bouche sèche apparaissent. Une dose excessive de nitroglycérine peut provoquer des maux de tête sévères. Seule la dose minimale nécessaire pour soulager la crise angineuse aiguë doit être prise. L'utilisation excessive de nitroglycérine peut conduire à une intolérance médicamenteuse.
Les comprimés enrobés de trinitrine doivent être pris par voie sublinguale et ne pas être avalés. L'ingestion des comprimés n'est pas nocive, mais annule l'effet thérapeutique.
Les comprimés enrobés de trinitrine contiennent du glucose, du lactose et du saccharose anhydres. Les patients présentant des formes rares de malabsorption du glucose et du galactose, des troubles héréditaires rares d'intolérance au galactose ou au fructose, un déficit en lactase ou en sucrase isomaltase ne doivent pas prendre le médicament.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de la Trinitrine
Des effets hypotenseurs additifs peuvent être observés chez les patients prenant des médicaments antihypertenseurs, des -bloquants, des phénothiazines et des nitrates. Une hypotension orthostatique marquée a été observée lorsque des médicaments bloquant le calcium et des nitrates organiques, tels que la nitroglycérine, sont administrés ensemble.
L'utilisation concomitante d'alcool et de nitrates peut provoquer une hypotension.
Les effets vasodilatateurs et hémodynamiques de la nitroglycérine peuvent être renforcés par l'administration concomitante d'aspirine.
La vasodilatation périphérique de la nitroglycérine est entravée par l'administration concomitante de l'indométacine anti-inflammatoire.
Les antidépresseurs tricycliques (amitriptyline, désipramine, doxépine et autres) et les médicaments anticholinergiques peuvent provoquer une sécheresse de la bouche et une diminution de la sécrétion salivaire. Cela peut rendre difficile la dissolution de la nitroglycérine sublinguale. Une augmentation de la salivation avec des produits de salive artificielle peut s'avérer bénéfique dans de tels cas.
L'administration orale de nitroglycérine diminue nettement le métabolisme de premier passage de l'ergotamine et augmente donc la biodisponibilité orale de cette dernière. L'ergotamine est connue pour déclencher des crises d'angine. Par conséquent, les patients prenant de la nitroglycérine sublinguale ne doivent pas prendre d'ergotamine et de médicaments apparentés, ou être surveillés de près pour des symptômes d'ergotisme s'il n'est pas possible d'éviter de prendre des dérivés de l'ergot.
Le sildénafil potentialise les effets hypotenseurs des nitrates et, par conséquent, sa co-administration avec des nitrates organiques, tels que la nitroglycérine, est contre-indiquée (voir « Contre-indications »).
Une diminution de l'effet thérapeutique de la nitroglycérine sublinguale peut être observée chez les patients prenant des nitrates à longue durée d'action.
Interactions avec les tests de laboratoire
Les nitrates peuvent interférer avec le dosage colorimétrique Zlatkis/Zak provoquant un faux résultat de diminution du cholestérol plasmatique.
Informez votre médecin ou pharmacien si vous prenez ou avez pris récemment tout autre médicament, même sans ordonnance.
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Il n'existe pas de données cliniques et épidémiologiques adéquates sur l'utilisation de la nitroglycérine pendant la grossesse.Les études animales sont insuffisantes pour établir les effets de la nitroglycérine sublinguale sur la grossesse, le développement embryonnaire et/ou postnatal et la parturition. Le risque potentiel chez l'homme est inconnu.
Les comprimés enrobés de trinitrine ne doivent pas être utilisés pendant la grossesse, en particulier au cours des trois premiers mois, sauf en cas de stricte nécessité.
On ne sait pas si la nitroglycérine est excrétée dans le lait maternel. Comme cette possibilité ne peut être exclue, une prudence particulière doit être exercée lors de la prise de nitroglycérine par une patiente qui allaite.
Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Les comprimés enrobés de trinitrine, en raison de certains effets indésirables (hypotension orthostatique, nausées, vertiges), peuvent avoir une influence majeure sur l'aptitude à conduire des véhicules ou à utiliser des machines. Ces effets indésirables peuvent réduire la capacité de réaction de la personne.
En cas d'intolérance avérée aux sucres, contactez votre médecin avant de prendre le médicament.
Posologie et mode d'utilisation Comment utiliser Trinitrina : Posologie
Généralement un comprimé suffit pour résoudre les symptômes douloureux ou prévenir la crise d'angine. Si nécessaire, un deuxième comprimé peut être pris après quelques minutes. Les comprimés doivent être écrasés avec les dents et laissés se dissoudre sous la langue. nocif, mais annule l'effet thérapeutique.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Surdosage Que faire si vous avez pris trop de Trinitrine
Les signes et symptômes sont principalement dus à une vasodilatation marquée et à une méthémoglobinémie. Les manifestations sont les suivantes : hypotension et tachycardie réflexe, peau froide et pâle, céphalées, vertiges, confusion mentale, troubles visuels, nausées et vomissements avec coliques douloureuses abdominales éventuellement associées à une diarrhée aqueuse. Palpitations, syncope et collapsus cardiovasculaire.
Le traitement des symptômes dus à une hypotension marquée et généralisée nécessite le positionnement du patient intoxiqué en position de Trendelenburg (couché et effectuant des mouvements passifs du membre) avec administration de fluides par voie intraveineuse. Si nécessaire, administrer des agonistes alpha-adrénergiques (c'est-à-dire de la méthoxamine ou de la phényléphrine). L'épinéphrine et les substances similaires sont inefficaces en cas d'hypotension marquée due à un surdosage.
Traitement de la cyanose due à la méthémoglobinémie : à partir d'un taux de 0,8 g/100 ml de méthémoglobine, le traitement consistera en l'administration intraveineuse de 1% de bleu de méthylène (1-2 mg/kg). kg doit être administré par voie orale Un traitement dans un centre spécialisé est recommandé.
En cas de prise accidentelle d'une dose excessive du médicament, informez immédiatement votre médecin ou rendez-vous à l'hôpital le plus proche.
Effets secondaires Quels sont les effets secondaires de la Trinitrine
Comme tous les médicaments, cela peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Un mal de tête, même sévère et persistant causé par une vasodilatation cérébrale, peut survenir immédiatement après la prise de nitroglycérine.
Vertiges, confusion, faiblesse, palpitations et autres manifestations d'hypotension orthostatique peuvent être ressentis occasionnellement et en particulier chez les sujets en position orthostatique immobile.
Chez les sujets particulièrement sensibles aux effets hypotenseurs des nitrates, des nausées, des vomissements, une transpiration, une pâleur et une lipothymie peuvent survenir aux doses thérapeutiques.
En revanche, la nitroglycérine ne peut que rarement induire une bradycardie et des signes d'hypervagotonie. De plus, chez les patients traités par nitrates, l'apparition de rougeurs cutanées et de dermatite exfoliative est possible.
Des effets indésirables tels que bouffées vasomotrices, céphalées et hypotension orthostatique peuvent limiter le traitement, en particulier aux stades précoces ou lorsque l'angine est sévère ou lorsque les patients sont hypersensibles aux effets des nitrates.
Les événements indésirables signalés sont énumérés ci-dessous, par classe d'organes.
- Troubles du système sanguin et lymphatique
Rare : méthémoglobinémie
- Troubles du système nerveux
vertiges, maux de tête
- Troubles oculaires
Vision floue
- Maladies cardiaques
tachycardie, palpitations, bradycardie paradoxale, syncope
- Troubles vasculaires
hypotension orthostatique
- Problèmes gastro-intestinaux
nausées, troubles digestifs
- Troubles cutanés et sous-cutanés
éruption
- Troubles généraux et altérations du site d'administration
Troubles généraux : bouffées de chaleur avec érythème, faiblesse, sueurs
Très rare : cyanose
Modification du site administratif
Brûlure, érythème.
La méthémoglobinémie a été associée à des traitements prolongés ou à fortes doses.
Si l'un des effets indésirables devient grave ou si vous remarquez un effet indésirable non mentionné dans cette notice, veuillez en informer votre médecin ou votre pharmacien.
Expiration et conservation
Conserver à une température ne dépassant pas 25°C.
N'utilisez pas le médicament après la date de péremption indiquée sur l'emballage.
La date de péremption fait référence au produit dans un emballage intact et correctement stocké.
Gardez ce médicament hors de la portée et de la vue des enfants.
Les médicaments ne doivent pas être jetés au tout-à-l'égout ou avec les ordures ménagères.
Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus. Cela contribuera à protéger l'environnement.
Composition
Chaque comprimé enrobé contient :
Principe actif: nitroglycérine 0,3 mg
Excipients: saccharose, amidon de maïs, lactose monohydraté, mannitol, talc, cellulose microcristalline, silice, dioxyde de titane, glucose anhydre, gomme arabique, amidon de pomme de terre, carbonate de magnésium, stéarate de magnésium, stéarate de calcium, gélatine, alcool polyvinylique, vanilline, p-hydroxybenzoate de méthyle .
Forme et contenu pharmaceutiques
Des comprimés enrobés. 2 plaquettes thermoformées de 35 comprimés.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
TRINITRINA 0,3 MG COMPRIMÉS ENROBÉS
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Chaque comprimé enrobé contient :
Principe actif: nitroglycérine 0,3 mg.
Pour les excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Des comprimés enrobés
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Traitement de la crise aiguë d'angine de poitrine.
04.2 Posologie et mode d'administration
Généralement, un comprimé suffit pour résoudre les symptômes douloureux ou prévenir la crise d'angine.Si nécessaire, un deuxième comprimé peut être pris après quelques minutes.
Les comprimés doivent être écrasés avec les dents et laissés se dissoudre sous la langue. L'ingestion des comprimés n'est pas nocive, mais annule l'effet thérapeutique.
04.3 Contre-indications
- choc
- choc cardiogénique, sauf si une pression diastolique aortique adéquate est maintenue
- état d'hypotension dans l'infarctus aigu du myocarde avec faible pression de remplissage
- hypotension sévère (pression systolique)
- hypovolémie sévère
- augmentation de la pression intracrânienne, traumatisme crânien et hémorragie cérébrale
- insuffisance myocardique due à une obstruction (par exemple en présence d'une sténose aortique ou mitrale ou d'une péricardite constrictive)
- cardiomyopathie hypertrophique obstructive
- œdème pulmonaire toxique
- anémie sévère
- glaucome à angle fermé
- généralement contre-indiqué pendant la grossesse, l'allaitement et l'âge pédiatrique en association avec le sildénafil (voir rubrique « Interactions avec d'autres médicaments et autres formes d'interactions »)
- hypersensibilité aux nitrates organiques et/ou à l'un des excipients
04.4 Mises en garde spéciales et précautions d'emploi appropriées
L'efficacité de la nitroglycérine prise par voie sublinguale chez les patients atteints d'insuffisance cardiaque congestive n'a pas été établie. Par conséquent, une surveillance clinique ou hémodynamique attentive doit être effectuée chez ces patients afin de détecter la possibilité d'hypotension et de tachycardie.
Une hypotension sévère, en particulier orthostatique, peut survenir après la prise même de petites doses de nitroglycérine. Par conséquent, le médicament doit être utilisé avec prudence chez les patients présentant une hypovolémie ou une hypotension préexistante. L'hypotension provoquée par la nitroglycérine peut s'accompagner d'une bradycardie paradoxale et d'une augmentation des douleurs angineuses.
Le traitement par nitrate peut aggraver l'angine chez les patients atteints de cardiomyopathie hypertrophique. À utiliser avec prudence chez les patients présentant une hypoxémie anémique sévère, car la biotransformation de la nitroglycérine est réduite chez ces patients.
Les comprimés enrobés de trinitrine doivent être arrêtés si des troubles visuels (vision trouble) ou une sécheresse buccale apparaissent.Une dose excessive de nitroglycérine peut provoquer de graves maux de tête.
Seule la dose minimale nécessaire pour soulager la crise angineuse aiguë doit être prise.L'utilisation excessive de nitroglycérine peut conduire à une intolérance médicamenteuse.
Les comprimés enrobés de trinitrine doivent être pris par voie sublinguale et ne pas être avalés. L'ingestion des comprimés n'est pas nocive, mais annule l'effet thérapeutique.Les comprimés enrobés de trinitrine contiennent du glucose, du lactose et du saccharose anhydres. Les patients présentant des formes rares de malabsorption du glucose et du galactose, des troubles héréditaires rares d'intolérance au galactose ou au fructose, un déficit en lactase ou en sucrase isomaltase ne doivent pas prendre le médicament.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Des effets hypotenseurs additifs peuvent être observés chez les patients prenant des médicaments antihypertenseurs, des -bloquants, des phénothiazines et des nitrates. Une hypotension orthostatique marquée a été observée lorsque des médicaments bloquant le calcium et des nitrates organiques, tels que la nitroglycérine, sont administrés ensemble.
L'utilisation concomitante d'alcool et de nitrates peut provoquer une hypotension.
Les effets vasodilatateurs et hémodynamiques de la nitroglycérine peuvent être renforcés par l'administration concomitante d'aspirine.
La vasodilatation périphérique de la nitroglycérine est entravée par l'administration concomitante de l'indométacine anti-inflammatoire.
Les antidépresseurs tricycliques (amitriptyline, désipramine, doxépine et autres) et les médicaments anticholinergiques peuvent provoquer une sécheresse de la bouche et une diminution de la sécrétion salivaire. Cela peut rendre difficile la dissolution de la nitroglycérine sublinguale. Une augmentation de la salivation avec des produits de salive artificielle peut s'avérer bénéfique dans de tels cas.
L'administration orale de nitroglycérine diminue nettement le métabolisme de premier passage de l'ergotamine et augmente donc la biodisponibilité orale de cette dernière. L'ergotamine est connue pour déclencher des crises d'angine. Par conséquent, les patients prenant de la nitroglycérine sublinguale ne doivent pas prendre d'ergotamine et de médicaments apparentés, ou être surveillés de près pour des symptômes d'ergotisme s'il n'est pas possible d'éviter de prendre des dérivés de l'ergot.
Le sildénafil potentialise les effets hypotenseurs des nitrates et, par conséquent, sa co-administration avec des nitrates organiques, tels que la nitroglycérine, est contre-indiquée (voir « Contre-indications »).
Une diminution de l'effet thérapeutique de la nitroglycérine sublinguale peut être observée chez les patients prenant des nitrates à longue durée d'action.
Interactions avec les tests de laboratoire
Les nitrates peuvent interférer avec le dosage colorimétrique Zlatkis/Zak provoquant un faux résultat de diminution du cholestérol plasmatique.
04.6 Grossesse et allaitement
Il n'existe pas de données cliniques et épidémiologiques adéquates sur l'utilisation de la nitroglycérine pendant la grossesse.Les études animales sont insuffisantes pour établir les effets de la nitroglycérine sublinguale sur la grossesse, le développement embryonnaire et/ou postnatal, et la parturition (voir paragraphe « Données de sécurité précliniques »). Le risque potentiel chez l'homme est inconnu.
Les comprimés enrobés de trinitrine ne doivent pas être utilisés pendant la grossesse, en particulier au cours des trois premiers mois, sauf en cas de stricte nécessité.
On ne sait pas si la nitroglycérine est excrétée dans le lait maternel. Comme cette possibilité ne peut être exclue, une prudence particulière doit être exercée lors de la prise de nitroglycérine par une patiente qui allaite.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Les comprimés enrobés de trinitrine, en raison de certains effets indésirables (hypotension orthostatique, nausées, vertiges), peuvent avoir une influence majeure sur l'aptitude à conduire des véhicules ou à utiliser des machines. Ces effets indésirables peuvent réduire la capacité de réaction de la personne.
04.8 Effets indésirables
Un mal de tête, même sévère et persistant causé par une vasodilatation cérébrale, peut survenir immédiatement après la prise de nitroglycérine.
Vertiges, confusion, faiblesse, palpitations et autres manifestations d'hypotension orthostatique peuvent être ressentis occasionnellement et en particulier chez les sujets en position orthostatique immobile.
Chez les sujets particulièrement sensibles aux effets hypotenseurs des nitrates, des nausées, des vomissements, une transpiration, une pâleur et une lipothymie peuvent survenir aux doses thérapeutiques.
En revanche, la nitroglycérine ne peut que rarement induire une bradycardie et des signes d'hypervagotonie. De plus, chez les patients traités par nitrates, l'apparition de rougeurs cutanées et de dermatite exfoliative est possible.
Des effets indésirables tels que bouffées vasomotrices, céphalées et hypotension orthostatique peuvent limiter le traitement, en particulier aux stades précoces ou lorsque l'angine est sévère ou lorsque les patients sont hypersensibles aux effets des nitrates.
Les événements indésirables signalés sont énumérés ci-dessous, par classe d'organes. Troubles du système sanguin et lymphatique
- Rare : méthémoglobinémie
Troubles du système nerveux
- vertiges, maux de tête
Troubles oculaires
- Vision floue
Maladies cardiaques
- tachycardie, palpitations, bradycardie paradoxale, syncope
Troubles vasculaires
- hypotension orthostatique
Problèmes gastro-intestinaux
- nausées, troubles digestifs
Troubles cutanés et sous-cutanés
- éruption
Troubles généraux et altérations du site d'administration
- Troubles généraux : bouffées de chaleur avec érythème, faiblesse, sueurs
- Très rare : cyanose
Modification du site administratif
- Brûlure, érythème
La méthémoglobinémie a été associée à des traitements prolongés ou à fortes doses.
04.9 Surdosage
Les signes et symptômes sont principalement dus à une vasodilatation marquée et à une méthémoglobinémie. Les manifestations sont les suivantes : hypotension et tachycardie réflexe, peau froide et pâle, céphalées, vertiges, confusion mentale, troubles visuels, nausées et vomissements avec coliques douloureuses abdominales éventuellement associées à une diarrhée aqueuse. Palpitations, syncope et collapsus cardiovasculaire.
Le traitement des symptômes dus à une hypotension marquée et généralisée nécessite le positionnement du patient intoxiqué en position de Trendelenburg (couché et effectuant des mouvements passifs du membre) avec administration de fluides par voie intraveineuse. Si nécessaire, administrer des agonistes alpha-adrénergiques (c'est-à-dire de la méthoxamine ou de la phényléphrine). L'épinéphrine et les substances assimilées sont inefficaces sur une hypotension marquée due à un surdosage Traitement de la cyanose due à la méthémoglobinémie : à partir d'un taux de 0,8 g/100 ml de méthémoglobine, le traitement consistera en une administration intraveineuse à 1, % de bleu de méthylène (1-2 mg/kg). Dans les cas moins graves, la dose de 50 mg/kg doit être administrée par voie orale. Un traitement dans un centre spécialisé est recommandé.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : vasodilatateurs utilisés dans les maladies cardiaques, nitrates organiques.
Code ATC : C01 DA 02.
La nitroglycérine a un effet relaxant sur les muscles lisses de l'organisme humain.Dans le compartiment vasculaire, elle agit principalement sur le système veineux et sur les grandes artères coronaires, avec un effet plus important sur les premières.
Dans l'angine de poitrine le principal mécanisme d'action de la nitroglycérine est représenté par l'augmentation de la capacité veineuse qui détermine une diminution du retour sanguin vers le cœur, donc la pression du ventricule gauche en fin de diastole (pré-charge) et le volume de remplissage. diminution, entraînant une diminution de la demande en oxygène du myocarde. Cette diminution de la consommation d'oxygène s'accompagne de l'amélioration de la perfusion myocardique résultant de l'action antispastique sur les branches épicardiques coronaires et de la redistribution du flux en faveur des couches sous-épicardiques du muscle cardiaque.De plus, la nitroglycérine dilate la sténose athéroscléreuse en cas de athérome excentrique et produit un relâchement du vasospasme spontané et induit par l'ergonovine.
Au niveau systémique, la nitroglycérine exerce un effet dilatateur dose-dépendant sur le système vasculaire artériel ; ceci détermine la réduction de la résistance vasculaire périphérique (postcharge) et de la tension systolique de la paroi ventriculaire gauche, deux événements contribuant à la diminution de la consommation d'oxygène du myocarde.
Corrélats cyniques
En corrélation avec l'amélioration clinique de l'angine de poitrine, la pléthysmographie digitale révèle que le début de l'effet vasodilatateur survient environ 1 minute après l'administration sublinguale de nitroglycérine et atteint son maximum environ 5 minutes après l'administration.Cet effet persiste pendant au moins 1 minute25. minutes après la prise.
Mécanisme d'action
La nitroglycérine est à l'origine du radical libre NO (oxyde nitrique) qui active la guanyl cyclase, avec une augmentation conséquente du GMP cyclique dans les muscles lisses et d'autres tissus. Cela conduit à une déphosphorylation des chaînes légères de la myosine, qui régule l'état de contraction des muscles lisses, entraînant une vasodilatation.
05.2 "Propriétés pharmacocinétiques
Absorption:
La nitroglycérine est rapidement absorbée après l'administration sublinguale de comprimés enrobés de Trinitrine. Les concentrations plasmatiques maximales surviennent 6 à 7 minutes après l'administration (voir Tableau).La concentration plasmatique maximale (Cmax) et l'aire sous la courbe concentration-temps (ASC) augmentent de manière proportionnelle à la dose. La biodisponibilité absolue de la nitroglycérine après la prise de comprimés enrobés de Trinitrine est d'environ 40 % et a tendance à varier en fonction des facteurs qui influencent l'absorption du médicament, tels que l'hydratation sublinguale et le métabolisme au niveau de la muqueuse buccale.
Table
Distribution:
Le volume de distribution (Vd) de la nitroglycérine après administration intraveineuse est de 3,3 L/kg.
A des concentrations plasmatiques comprises entre 50 et 500 ng/ml, la liaison de la nitroglycérine aux protéines plasmatiques est d'environ 60 %, tandis que celle de la 1,2- et 1,3-dinitroglycérine (les deux principaux métabolites de la nitroglycérine) est de 60 % et 30 %. %, respectivement.
Métabolisme:
Le principe actif est rapidement métabolisé dans le foie par une nitrate réductase organique dépendante du glutathion. Aussi, vous étudiezin vitro avec les érythrocytes humains, ils ont montré que le globule rouge est également le site de biotransformation de la nitroglycérine au moyen d'un processus enzymatique sulfhydryle-dépendant et d'une interaction avec une hémoglobine réduite.
La quantité d'hémoglobine réduite présente dans les érythrocytes humains semble jouer un rôle primordial dans leur activité métabolique, par conséquent, des précautions doivent être prises lors de la prise de nitroglycérine chez des patients anémiques (voir rubrique « Mises en garde spéciales et précautions d'emploi appropriées »). Il a été montré que les tissus vasculaires extrahépatiques (veine fémorale, veine cave inférieure, aorte) jouent également un rôle important dans le métabolisme de la nitroglycérine, ce qui explique la clairance systémique élevée des nitrates. in vitro que la biotransformation de la nitroglycérine se produit en même temps que la relaxation du muscle vasculaire lisse ; cette observation est cohérente avec l'hypothèse que la biotransformation de la nitroglycérine (en NO) est le mécanisme de la vasodilatation induite par le médicament.
Élimination:
Les concentrations plasmatiques de nitroglycérine chutent rapidement après l'administration, avec une demi-vie d'élimination moyenne de 2 à 3 minutes.La clairance (13,8 L/min) dépasse de loin le débit sanguin hépatique.
05.3 Données de sécurité précliniques
Les données non cliniques ne révèlent aucun risque pour l'homme sur la base des études conventionnelles de pharmacologie de sécurité, de toxicité à doses répétées, de génotoxicité, de potentiel cancérigène, de toxicité pour la reproduction.
Toxicité à dose unique : La DL50 de la nitroglycérine orale est de 105 mg/kg chez le rat et de 15 mg/kg chez la souris.
Toxicité à doses répétées : Dans des études de toxicité chronique chez des rats albinos en croissance, une dose de 300 mg/kg administrée pendant 6 mois par voie épicutanée n'a entraîné aucune modification significative de la croissance corporelle, de la numération globulaire ou d'autres paramètres biologiques. organes examinés a été observée. A une dose de 825 mg/kg administrée par voie intrapéritonéale chez le rat pendant 33 jours, une diminution du poids corporel a été observée.
Genèse muette : Bien que la nitroglycérine ait été faiblement mutagène dans le test d'Ames, il n'y avait aucun signe de génotoxicité dans les tests. in vivo (essai létal dominant) chez les rats mâles traités avec des doses orales allant jusqu'à 363 mg/kg, ou dans des tests cytogénétiques ex vivo dans les tissus murins et canins.
Carcinogenèse : Des rats traités par de très fortes doses de nitroglycérine (363 mg/kg/jour chez les mâles et 434 mg/kg/jour chez les femelles) pendant 2 ans ont montré l'apparition de carcinomes hépatocellulaires et de tumeurs des cellules interstitielles des testicules. Des souris traitées avec 1022 (mâles ) ou 1058 (femelles) mg/kg/jour et les rats traités par 31,5 (mâles) ou 38,1 (femelles) mg/kg/jour pendant la même période n'ont présenté aucune tumeur liée au traitement tout au long du cycle de vie de 1058 mg/kg / jour de nitroglycérine n'a pas entraîné d'augmentation de l'incidence des néoplasmes.
Toxicité pour la reproduction: Aucune étude de toxicité pour la reproduction et de tératogénicité n'a été menée avec la nitroglycérine administrée par voie sublinguale. Cependant, les études tératogènes menées chez le rat et le lapin avec de la nitroglycérine topique à des doses allant jusqu'à 80 mg/kg/jour et 240 mg/kg/jour, respectivement, n'ont révélé aucun effet nocif sur le fœtus.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Saccharose, amidon de maïs, lactose monohydraté, mannitol, talc, cellulose microcristalline, silice, dioxyde de titane, glucose anhydre, gomme arabique, amidon de pomme de terre, carbonate de magnésium, stéarate de magnésium, stéarate de calcium, gélatine, alcool polyvinylique, vanilline, p-méthyl hydroxybenzoate.
06.2 Incompatibilité
Non pertinent.
06.3 Durée de validité
2 ans.
06.4 Précautions particulières de conservation
Conserver à une température ne dépassant pas 25°C.
06.5 Nature du conditionnement primaire et contenu de l'emballage
PVC / PVDC 60 / Blister aluminium
2 plaquettes thermoformées de 35 comprimés.
06.6 Instructions d'utilisation et de manipulation
Pas d'instructions particulières.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
ACARPIA Farmaceutici srl
via Vivaio, 17
20122 MILAN (ITALIE)
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
006035063
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
Renouvellement:
06/2010
10.0 DATE DE RÉVISION DU TEXTE
06/2010