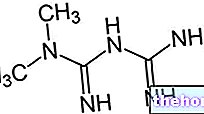
Ingrédients actifs : Énanthate de testostérone
Testoviron 250 mg/ml solution injectable à libération prolongée pour voie intramusculaire
Pourquoi le testoviron est-il utilisé ? Pourquoi est-ce?
Le testoviron est utilisé pour remplacer la testostérone chez les hommes adultes, pour traiter divers problèmes de santé dus à une carence en testostérone (hypogonadisme masculin). Cette condition doit être confirmée par deux mesures distinctes de testostérone dans le sang et doit inclure des symptômes cliniques tels que :
- impuissance
- infertilité
- faible désir sexuel
- fatigue
- humeur dépressive
- perte osseuse due à de faibles niveaux d'hormones
Contre-indications Quand Testoviron ne doit pas être utilisé
Ne pas utiliser Testoviron
- si vous êtes allergique à l'énanthate de testostérone ou à l'un des autres composants contenus dans ce médicament
- si vous avez un cancer androgéno-dépendant (cancer) de la prostate ou de la glande mammaire masculine ;
- si vous avez une hypercalcémie (excès de calcium dans le sang) secondaire à des tumeurs malignes ;
- si vous avez ou avez déjà eu un cancer du foie;
- si vous souffrez d'une maladie rénale ;
- si vous souffrez d'hypertension artérielle;
- si vous souffrez d'un dysfonctionnement hépatique sévère.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Testoviron
Adressez-vous à votre médecin ou pharmacien avant d'utiliser Testoviron.
Par précaution, subir périodiquement un examen andrologique avec exploration rectale de la prostate.
Avant et pendant le traitement, le médecin doit effectuer les analyses sanguines suivantes : taux de testostérone dans le sang, formule sanguine complète.
Si vous avez une prédisposition à l'œdème (gonflement), votre médecin devra vous surveiller attentivement, car le traitement par androgènes peut augmenter le sodium et par conséquent la rétention d'eau.
Après l'utilisation de Testoviron, des tumeurs hépatiques bénignes et malignes, pouvant provoquer des saignements intra-abdominaux potentiellement mortels, ont été observées.
Si vous souffrez d'une maladie grave du cœur, du foie ou des reins, le traitement par Testoviron peut entraîner de graves complications sous la forme d'une accumulation d'eau dans le corps, parfois accompagnée d'une insuffisance cardiaque (congestive).
Informez votre médecin si vous souffrez d'hypertension artérielle ou si vous êtes traité pour une hypertension artérielle, car la testostérone peut faire augmenter votre tension artérielle.
Les patients plus âgés traités avec des androgènes peuvent avoir un risque accru de développer une hyperplasie de la prostate (hypertrophie de la prostate). Bien qu'il n'y ait aucune indication claire que les androgènes causent le cancer de la prostate (cancer de la prostate), il est possible qu'ils favorisent sa croissance s'il est déjà présent. Par conséquent, avant de prescrire un traitement avec des préparations de testostérone, votre médecin devra exclure la présence d'un cancer de la prostate.
Le testoviron peut provoquer une « stimulation sexuelle excessive. Si cela se produit, veuillez contacter votre médecin.
Si vous êtes âgé, votre médecin veillera attentivement à ce que ce médicament ne vous provoque pas de stimulation physique, nerveuse et mentale excessive et indésirable.
Comme toutes les solutions huileuses, Testoviron ne doit être injecté que par voie intramusculaire et très lentement. La microembolie pulmonaire de solutions huileuses (petites gouttelettes huileuses dans les artères des poumons) peut entraîner des signes et des symptômes tels que toux, difficultés respiratoires et douleurs thoraciques. D'autres signes et symptômes peuvent également apparaître, notamment des malaises, des sueurs, des étourdissements, des paresthésies (sensation altérée, par exemple, des picotements) ou des évanouissements. Ces réactions peuvent survenir pendant ou peu après l'injection et sont réversibles.Le traitement consiste en des mesures de soutien, telles que l'administration d'oxygène.
Si vous souffrez d'apnée du sommeil (épisodes d'arrêt de la respiration pendant le sommeil), cela peut être accentué par l'utilisation de Testoviron.
Le testoviron n'est pas indiqué pour augmenter le développement musculaire chez les individus en bonne santé ou pour améliorer les performances physiques.
Testoviron ne doit pas être utilisé chez les femmes car il peut provoquer une jaunisse (jaunissement de la peau et du blanc des yeux) et des signes de virilisation, tels que l'acné, une croissance anormale des poils dans les zones masculines typiques du corps), des changements de voix.
Pour ceux qui pratiquent des activités sportives
L'utilisation du médicament sans nécessité thérapeutique constitue un dopage et peut en tout état de cause déterminer des tests antidopage positifs.
Enfants et adolescents
L'utilisation de Testoviron n'est pas indiquée chez les enfants et les adolescents.Chez les enfants, la testostérone, en plus d'avoir des effets masculinisants, peut avoir un effet sur la maturation osseuse, entraînant une diminution de la croissance en taille.
Patients présentant une insuffisance hépatique
L'utilisation de Testoviron est contre-indiquée chez les patients qui ont ou ont eu des tumeurs du foie (voir « N'utilisez pas Testoviron »).
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Testoviron
Informez votre médecin ou pharmacien si vous prenez, avez récemment pris ou pourriez prendre tout autre médicament.
En particulier, informez votre médecin si vous prenez les médicaments suivants :
- phénobarbital (médicament antiépileptique), car il peut réduire l'efficacité de Testoviron.
- dérivés de la coumarine (anticoagulants, utilisés pour fluidifier le sang), car le médecin surveillera de près les paramètres de coagulation.
Testoviron peut également modifier le test de tolérance au glucose :
si vous êtes diabétique, demandez conseil à votre médecin avant de prendre Testoviron, car la dose de votre insuline ou de vos médicaments antidiabétiques oraux peut devoir être ajustée.
Avertissements Il est important de savoir que :
La fertilité
La thérapie de remplacement de la testostérone peut réduire de manière réversible la spermatogenèse (la production de spermatozoïdes).
Conduire et utiliser des machines
Le testoviron n'affecte pas l'aptitude à conduire des véhicules et à utiliser des machines.
Testoviron contient de l'huile de ricin
Il peut provoquer de graves réactions allergiques.
Posologie et mode d'utilisation Comment utiliser Testoviron : Posologie
Utilisez toujours ce médicament en suivant exactement les indications de votre médecin ou pharmacien. En cas de doute, consultez votre médecin ou votre pharmacien.
Ce médicament est à usage intramusculaire uniquement.
La solution huileuse doit être injectée très lentement (voir « Avertissements et précautions » et « Effets indésirables éventuels »), immédiatement après avoir été aspirée dans la seringue.
La dose recommandée est :
- pour le développement et la stimulation des organes cibles androgéno-dépendants et le traitement initial des symptômes de carence : un flacon de 250 mg toutes les 2 à 3 semaines.
- pour le maintien d'un effet androgène adéquat : un flacon de 250 mg toutes les 3 à 4 semaines.
En fonction des besoins individuels, le médecin peut décider de réduire les intervalles entre les injections, alors que dans de nombreux cas, des intervalles plus longs, jusqu'à 6 semaines, sont suffisants.
Avant de commencer le traitement, et de temps en temps, à la fin d'un intervalle entre les injections, votre médecin mesurera votre taux de testostérone dans votre sang. Des niveaux inférieurs à la normale indiquent la nécessité de raccourcir l'intervalle entre les injections, alors qu'il devra être prolongé en cas de niveaux élevés.
Utilisation chez les personnes âgées
Aucun ajustement posologique n'est nécessaire chez les patients âgés (voir « Avertissements et précautions »).
Surdosage Que faire si vous avez pris trop de Testoviron
En cas d'ingestion/prise accidentelle d'un surdosage de Testoviron, prévenez immédiatement votre médecin ou rendez-vous à l'hôpital le plus proche. Aucune mesure thérapeutique spéciale n'est nécessaire; votre médecin décidera d'arrêter le traitement ou de réduire la dose.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Effets secondaires Quels sont les effets secondaires de Testoviron
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Concernant les effets indésirables liés à l'utilisation d'androgènes, veuillez également vous référer à la rubrique "Avertissements et précautions".
Les effets indésirables les plus fréquents avec Testoviron sont la douleur au site d'injection, l'érythème (rougeur) au site d'injection, la toux et/ou la dyspnée (difficulté à respirer) pendant ou peu après l'injection.
Les effets secondaires courants sont les suivants : augmentation du nombre de globules rouges, de l'hématocrite (pourcentage de globules rouges dans le sang) et de l'hémoglobine (le composant des globules rouges qui transporte l'oxygène), mis en évidence par des tests sanguins périodiques.
Les autres effets indésirables, dont la fréquence ne peut être estimée, sont listés ci-dessous :
- tumeurs hépatiques bénignes et malignes,
- polyglobulie (augmentation du nombre de globules rouges dans le sang),
- allergie,
- tests anormaux de la fonction hépatique,
- jaunisse (jaunissement de la peau et du blanc des yeux),
- acné,
- alopécie (chute des cheveux),
- éruption,
- urticaire (démangeaisons et petites taches sur la peau),
- démanger,
- différents types de réactions au site d'injection (douleur, rougeur, dureté, gonflement, inflammation),
- augmentation de l'antigène prostatique spécifique (PSA),
- augmentation ou diminution du désir sexuel,
- gynécomastie (croissance des seins chez les hommes).
Les injections de solutions huileuses telles que le Testoviron ont été associées à des réactions généralisées : toux, difficultés respiratoires et douleurs thoraciques. D'autres signes et symptômes peuvent également apparaître, tels qu'un malaise, des sueurs, des vertiges, des paresthésies (sensation altérée, par exemple, des picotements) ou des évanouissements.
L'administration de testostérone à fortes doses ou pour des traitements prolongés peut induire des états d'anxiété, d'hypercalcémie (augmentation de la quantité de calcium dans le sang), de rétention d'eau (accumulation de liquide dans les tissus) et d'œdème (gonflement). une tendance et en particulier si vous avez de graves problèmes rénaux ou cardiaques, souffrez d'asthme, souffrez d'épilepsie.
Des doses élevées de testostérone ou de ses dérivés inhibent la sécrétion de gonadotrophines (les hormones qui stimulent les testicules) entraînant une atrophie testiculaire.
Un traitement prolongé et à haute dose avec Testoviron inhibe la spermatogenèse (le processus de maturation des cellules germinales mâles, qui se termine par la formation de spermatozoïdes).
Contactez votre médecin si vous avez des érections fréquentes ou persistantes. Votre médecin décidera s'il faut réduire la posologie ou arrêter le traitement, pour éviter de blesser le pénis.
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice. Vous pouvez également signaler les effets indésirables directement via le système national de notification à l'adresse www.agenziafarmaco.gov.it/it/responsabili. En signalant les effets indésirables, vous pouvez contribuer à fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants.
N'utilisez pas ce médicament après la date de péremption indiquée sur la boîte après péremption. La date de péremption fait référence au dernier jour de ce mois La date de péremption fait référence au produit dans un emballage intact, correctement stocké.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Ce que contient Testoviron
- L'ingrédient actif est l'énanthate de testostérone. Un flacon contient 250 mg d'énanthate de testostérone (correspondant à 180 mg de testostérone).
- Les autres composants sont : benzoate de benzyle, huile de ricin pour préparations injectables.
A quoi ressemble Testoviron et contenu de l'emballage extérieur
Le testoviron est une solution huileuse limpide et jaunâtre. Il est disponible en conditionnements (boîtes) contenant 1 ampoule de 250 mg/1 ml
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus à jour, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
TESTOVIRON 250 MG/ML SOLUTION INJECTABLE À LIBÉRATION PROLONGÉE POUR USAGE INTRAMUSCULAIRE
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Un flacon de 1 ml contient :
principe actif : 250 mg d'énanthate de testostérone (correspondant à environ 180 mg de testostérone)
excipients à effet notoire : huile de ricin.
Pour la liste complète des excipients, voir la section 6.1.
03.0 FORME PHARMACEUTIQUE
Solution injectable à libération prolongée.
Solution huileuse limpide et jaunâtre.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Thérapie de remplacement de la testostérone pour l'hypogonadisme masculin, lorsque le déficit en testostérone a été confirmé par le tableau clinique et les analyses biochimiques.
04.2 Posologie et mode d'administration
Utilisation intramusculaire.
La solution huileuse doit être injectée très lentement (voir rubriques 4.4 et 4.8), immédiatement après avoir été aspirée dans la seringue.
Dosage
Pour le développement et la stimulation des organes cibles androgéno-dépendants et pour le traitement initial des symptômes de carence : 250 mg toutes les 2 à 3 semaines.
Pour maintenir un effet androgène adéquat : 250 mg toutes les 3 à 4 semaines. Selon les besoins individuels, des intervalles plus courts entre les injections peuvent être nécessaires, tandis que des intervalles plus longs allant jusqu'à 6 semaines sont suffisants dans de nombreux cas.
Les taux sériques de testostérone doivent être mesurés avant le début du traitement et occasionnellement à la fin d'un « intervalle entre les injections. Des taux sériques inférieurs à la normale indiquent la nécessité de raccourcir l'intervalle entre les injections, alors que cela devra être prolongé en cas de taux sérique élevé. niveaux.
Informations supplémentaires pour des catégories particulières de patients
Enfants et adolescents
L'utilisation de Testoviron n'est pas indiquée chez les enfants et les adolescents (voir rubrique 4.4).
Patients âgés
Les données limitées disponibles ne suggèrent pas la nécessité d'un ajustement de la dose chez les patients âgés (voir rubrique 4.4).
Patients présentant une insuffisance hépatique
Aucune étude formelle n'a été menée chez des patients présentant une insuffisance hépatique. L'utilisation de Testoviron est contre-indiquée chez les patients présentant des tumeurs hépatiques antérieures ou actuelles (voir rubrique 4.3).
Patients atteints d'insuffisance rénale
Aucune étude formelle n'a été menée chez des patients présentant une insuffisance rénale.
04.3 Contre-indications
Hypersensibilité à la substance active ou à l'un des excipients mentionnés à la rubrique 6.1.
Cancer androgéno-dépendant de la prostate ou de la glande mammaire masculine.
Hypercalcémie secondaire à des tumeurs malignes.
Tumeurs hépatiques antérieures ou actuelles.
Maladie rénale, hypertension, dysfonctionnement hépatique sévère.
04.4 Mises en garde spéciales et précautions d'emploi appropriées
L'expérience sur la sécurité et l'efficacité de l'utilisation de Testoviron chez les patients de plus de 65 ans est limitée.Il n'y a pas actuellement d'accord sur les valeurs de référence de testostérone spécifiques à l'âge, mais il faut considérer que les niveaux de testostérone sérique diminuent physiologiquement avec l'âge.
Après utilisation de Testoviron, des tumeurs hépatiques bénignes et malignes ont été observées, pouvant provoquer une hémorragie intra-abdominale potentiellement mortelle. tumeur hépatique dans le diagnostic différentiel.
Les patients plus âgés traités avec des androgènes peuvent avoir un risque accru de développer une hyperplasie de la prostate. Bien qu'il n'y ait aucune indication claire que les androgènes causent le cancer de la prostate, il est possible qu'ils favorisent sa croissance s'il est déjà présent. Par conséquent, avant de commencer un traitement avec des préparations de testostérone, il est nécessaire d'exclure le cancer de la prostate.
Par mesure de précaution, des examens rectaux périodiques de la prostate sont recommandés.
Les taux de testostérone doivent être surveillés au départ et à intervalles réguliers pendant le traitement. Les médecins doivent ajuster la dose sur une base individuelle d'un patient à l'autre pour assurer le maintien des taux de testostérone eugonadique.
Chez les patients recevant un traitement androgénique à long terme, les paramètres de laboratoire suivants doivent également être surveillés à intervalles réguliers : hémoglobine et hématocrite, paramètres de la fonction hépatique et profil lipidique (voir rubrique 4.8).
Chez les patients présentant une insuffisance cardiaque, hépatique ou rénale sévère ou une cardiopathie ischémique, le traitement par la testostérone peut induire des complications sévères caractérisées par un œdème avec ou sans insuffisance cardiaque congestive. Si cela se produit, le traitement doit être arrêté immédiatement.La prudence est de mise chez les patients ayant une prédisposition aux œdèmes, car le traitement par androgènes peut augmenter la rétention sodée (voir rubrique 4.8).
La testostérone peut provoquer une augmentation de la pression artérielle et Testoviron doit être utilisé avec prudence chez les personnes hypertendues.
Si des symptômes d'hyperstimulation sexuelle apparaissent, le traitement doit être interrompu. Chez les sujets âgés, il doit être soigneusement observé que le traitement ne provoque pas de stimulations nerveuses, mentales et physiques excessives indésirables.
Chez l'enfant, la testostérone, en plus d'avoir des effets masculinisants, peut accélérer la croissance et la maturation osseuse, provoquant une fermeture prématurée des épiphyses et réduisant ainsi la taille finale.
Testoviron ne doit pas être utilisé chez les femmes en raison de la jaunisse et des signes de virilisation, tels que l'acné, l'hirsutisme, des modifications de la voix, peuvent survenir en fonction de la sensibilité individuelle aux impulsions androgènes.
L'apnée du sommeil préexistante peut être potentialisée.
Les androgènes ne sont pas indiqués pour augmenter le développement musculaire chez les individus en bonne santé ou pour augmenter la capacité physique.
Comme toutes les solutions huileuses, Testoviron ne doit être injecté que par voie intramusculaire et très lentement. La microembolie pulmonaire des solutions huileuses peut entraîner des signes et des symptômes tels que la toux, une respiration sifflante et des douleurs thoraciques. D'autres signes et symptômes peuvent également apparaître, notamment des réactions vasovagales telles que malaise, hyperhidrose, vertiges, paresthésie ou syncope. Ces réactions peuvent survenir pendant ou peu après l'injection et sont réversibles.Le traitement consiste en des mesures de soutien, telles que l'administration d'oxygène.
Informations importantes sur certains ingrédients
Testoviron contient de l'huile de ricin qui peut provoquer de graves réactions allergiques.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Le phénobarbital augmente le métabolisme hépatique des stéroïdes, réduisant éventuellement leur efficacité.
Lorsque Testoviron est administré en concomitance avec des dérivés de la coumarine, les paramètres de coagulation doivent être étroitement surveillés.
Les androgènes peuvent modifier le test de tolérance au glucose ; par conséquent, leur utilisation chez les sujets diabétiques chez lesquels il peut être nécessaire d'ajuster la posologie d'insuline et d'agents antidiabétiques oraux nécessite une attention particulière.
04.6 Grossesse et allaitement
La grossesse et l'allaitement
Testoviron est destiné à être utilisé chez les hommes uniquement.Le produit n'est pas indiqué pendant la grossesse et l'allaitement (voir rubrique 5.3).
La fertilité
La thérapie de remplacement de la testostérone peut réduire de manière réversible la spermatogenèse (voir rubriques 4.8 et 5.3).
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Le testoviron n'affecte pas l'aptitude à conduire des véhicules et à utiliser des machines.
04.8 Effets indésirables
Concernant les effets indésirables liés à l'utilisation d'androgènes, veuillez également vous référer à la rubrique 4.4.
Les effets indésirables les plus fréquents avec Testoviron sont la douleur au site d'injection, l'érythème au site d'injection, la toux et/ou la dyspnée pendant ou peu après l'injection.
Les effets indésirables fréquents sont : augmentation de l'hématocrite, augmentation du nombre de globules rouges, augmentation de l'hémoglobine.
Liste tabulée des effets indésirables
Le tableau suivant rapporte les effets indésirables issus des notifications spontanées et de la littérature scientifique, dont il n'est pas possible d'estimer la fréquence sur la base des données disponibles.
§ Le terme préféré de MedDRA est utilisé pour décrire un effet indésirable particulier, ses synonymes et les conditions associées.
* Douleur au site d'injection, érythème au site d'injection, induration au site d'injection, gonflement au site d'injection, inflammation au site d'injection.
Description de certains effets indésirables
Les injections de solutions huileuses telles que le Testoviron ont été associées à des réactions systémiques : toux, respiration sifflante et douleurs thoraciques. D'autres signes et symptômes peuvent également apparaître, notamment des réactions vasovagales telles que malaise, hyperhidrose, vertiges, paresthésie ou syncope.
L'administration de testostérone, dont le Testoviron, à fortes doses ou pour des traitements prolongés peut induire des états d'anxiété, d'hypercalcémie et augmente la tendance à la rétention d'eau et aux œdèmes.
Par conséquent, il est conseillé de faire preuve de prudence en soumettant à un traitement à la testostérone les patients qui présentent une tendance similaire et en particulier ceux présentant une insuffisance rénale ou cardiovasculaire, les asthmatiques et ceux souffrant d'épilepsie.
Des doses élevées de testostérone ou de ses dérivés suppriment la sécrétion de gonadotrophines avec pour conséquence une atrophie du tissu interstitiel et des tubules séminifères du testicule.
Un traitement prolongé à haute dose avec Testoviron inhibe la spermatogenèse.
Si des érections fréquentes ou persistantes surviennent dans des cas individuels, la posologie doit être réduite ou le traitement interrompu pour éviter des lésions péniennes.
Déclaration des effets indésirables suspectés
La déclaration des effets indésirables suspectés survenant après autorisation du médicament est importante car elle permet un suivi continu du rapport bénéfice/risque du médicament. Les professionnels de santé sont invités à déclarer tout effet indésirable suspecté via le système national de déclaration. agenziafarmaco.gov.it/it/responsabili.
04.9 Surdosage
En cas de surdosage, aucune mesure thérapeutique particulière n'est requise, autre que l'interruption du traitement ou la réduction de la dose.
Des études de toxicité aiguë montrent cependant que l'énanthate de testostérone, l'ester contenu dans Testoviron, doit être classé comme non toxique après une seule administration.Même en cas d'administration unique d'une dose plusieurs fois supérieure à celle nécessaire à l'objectif thérapeutique aucun risque de toxicité est prévue.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : Hormones sexuelles et modulateurs de l'appareil génital, androgènes, dérivés 3-oxoandrostène
Code ATC : G03BA03
Le testoviron contient, en tant qu'ingrédient actif, de l'énanthate de testostérone, un dérivé de la testostérone, une hormone sexuelle masculine naturelle, et est donc capable de contrer les symptômes de la carence en androgènes.La forme active, la testostérone, est formée en divisant la chaîne latérale.
05.2 Propriétés pharmacocinétiques
Absorption
Après administration intramusculaire, la disponibilité systémique de l'énanthate de testostérone est complète. Le composé est progressivement libéré du dépôt avec une demi-vie d'environ 4,5 jours et décomposé en testostérone et acide énanthique.Avec une dose de 250 mg d'énanthate de testostérone, les patients reçoivent une dose totale de testostérone de 180 mg.Après environ 4 semaines après l'administration du médicament, la libération de testostérone du dépôt est complète.
Distribution
Des concentrations maximales de testostérone de 20 ng/mL ont été mesurées entre 1,5 et 3 jours après administration IM. de 250 mg d'énanthate de testostérone aux jeunes hommes. Par la suite, les taux plasmatiques de testostérone ont diminué avec une demi-vie d'environ 4,5 jours, ce qui correspond à la vitesse de libération du dépôt. Les concentrations de testostérone ≥ 2 ng/ml ont été maintenues pendant 20 jours, tandis que les concentrations ≥ 1 ng/ml ont été maintenues pendant 26 jours.
La liaison de la testostérone aux protéines sériques, en particulier à l'albumine et à la globuline liant les hormones sexuelles, est élevée.
Métabolisme
La testostérone générée par l'hydrolyse de l'énanthate de testostérone est métabolisée et excrétée comme la testostérone endogène. La biodisponibilité absolue de la testostérone à partir de l'ester est presque complète, indiquant une "hydrolyse de l'ester" rapide et efficace.L'acide énanthique est métabolisé par -oxydation comme les autres acides carboxyliques aliphatiques.
Élimination
La clairance métabolique de la testostérone est de 16 ± 7 ml/min/kg et est liée au métabolisme hépatique et extrahépatique. Les métabolites de la testostérone sont éliminés avec une demi-vie de 7,8 jours, environ 90 % par le rein et 10 % par la bile.
Régime permanent
L'injection de 250 mg d'énanthate de testostérone toutes les 3 à 4 semaines n'entraîne pas d'accumulation cliniquement significative de testostérone dans le sérum.
05.3 Données de sécurité précliniques
Toxicité aiguë
Comme pour les hormones stéroïdes en général, la toxicité aiguë de la testostérone est très faible.
Toxicité chronique
Des investigations sur la tolérabilité systémique après administration répétée, aucune donnée n'a émergé qui déconseillerait l'utilisation de l'ingrédient actif aux doses nécessaires pour le traitement.
Potentiel mutagène et cancérigène
Études de mutagénicité in vitro avec la testostérone libérée de l'ester, ils n'ont donné aucune indication d'un potentiel mutagène.Aucune étude n'a été menée pour évaluer un éventuel effet cancérogène du testoviron après administration répétée. Ces études n'ont pas été jugées nécessaires, car aucune indication d'effet cancérigène n'est ressortie des études de tolérance systémique pour des doses répétées pendant 6 mois chez le rat et le chien.
Toxicité pour la reproduction
Aucune étude de fertilité préclinique sur les dommages possibles aux cellules germinales n'a été menée. Les études précliniques montrent une « inhibition centrale de la spermatogenèse et de l'ovogenèse.
Tolérance locale
Des études de tolérance locale après administration intramusculaire ont montré que l'énanthate de testostérone n'augmente pas l'effet irritant causé par le solvant seul. Les investigations menées avec le solvant huileux de Testoviron n'ont pas révélé d'effet sensibilisant. Aucune autre investigation sur le solvant n'a été réalisée. énanthate de testostérone. Aucun effet sensibilisant évident n'a été démontré.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Benzoate de benzyle, huile de ricin pour préparations injectables.
06.2 Incompatibilité
Rien.
06.3 Durée de validité
5 années.
06.4 Précautions particulières de conservation
Ce médicament ne nécessite aucune condition particulière de conservation.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Chaque emballage de Testoviron contient un flacon en verre, classe hydrolytique I sec. F.U., 1 ml.
06.6 Instructions pour l'utilisation et la manipulation
Pas d'instructions particulières.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Bayer S.p.A. - Viale Certosa, 130 - 20156 Milan
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
A.I.C. n.m. 002922060
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
10 juin 1953/01 juin 2010
10.0 DATE DE RÉVISION DU TEXTE
Détermination AIFA du 04/2015