VECTAVIR ® est un médicament à base de Penciclovir
GROUPE THÉRAPEUTIQUE : Antiviraux à usage topique

Indications VECTAVIR ® Penciclovir
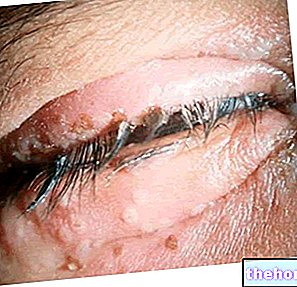
VECTAVIR ® est indiqué pour le traitement des boutons de fièvre seulement.
Mécanisme d'action VECTAVIR ® Penciclovir
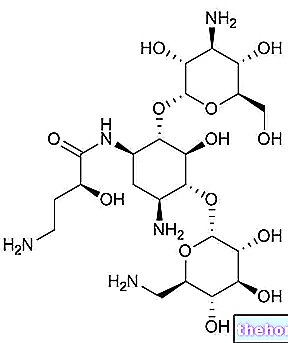
VECTAVIR ® est un médicament destiné au traitement des boutons de fièvre à base de Penciclovir, principe actif à forte activité antivirale, particulièrement efficace contre le Virus Herpes Simplex.
Appliqué à la zone affectée par le processus infectieux, le penciclovir pénètre facilement les membranes des cellules infectées, étant phosphorylé en peniciclovir triphosphate d'abord par des enzymes virales spécifiques et ensuite par des kinases cellulaires.
Le médicament triphosphorylé, analogue nucléosidique de la guanosine, est incorporé dans la chaîne naissante bloquant ainsi l'activité de l'enzyme virale ADN polymérase, et inhibant ainsi tout processus de prolifération virale responsable des symptômes en cours.
Malgré la bonne efficacité du médicament, il est possible de décrire l'apparition de mécanismes de résistance de nature à frustrer l'aide thérapeutique par :
- L'expression réduite de l'enzyme virale thymidine kinase, initiateur de la réaction enzymatique ;
- L'altération de l'affinité de liaison entre le Penciclovir et la thymidine kinase virale ;
- L'altération de l'affinité de liaison entre le Penciclovir et l'ADN polymérase virale.
Les mécanismes ci-dessus sont généralement responsables d'une aggravation du tableau clinique.
Études réalisées et efficacité clinique
CAPACITÉ DE DIFFUSION DU PENCICLOVIR TOPIQUEMENT
BMC Dermatol. 2 avril 2009 ; 9 : 3.
Évaluation de la perméation cutanée in vitro des médicaments antiviraux de la crème penciclovir 1% et de la crème aciclovir 5% utilisés pour traiter l'infection par le virus de l'herpès simplex.
Hasler-Nguyen N, Shelton D, Ponard G, Bader M, Schaffrik M, Mallefet P.
Étude moléculaire qui démontre comment l'application de crèmes Penciclvor à 1% peut être efficace dans le traitement de l'herpès labial en se propageant facilement de la couche cornée aux couches plus profondes de l'épiderme jusqu'à la couche basale.
L'EFFICACITÉ DES CRÈMES À BASE DE PENCICLOVIR DANS LE TRAITEMENT DE L'HERPÈS LABIAL
Peau Pharmacol Physiol. 2004 sept.-oct. 17 : 214-8.
Crème de penciclovir - traitement topique amélioré pour les infections à herpès simplex.
Schmid-Wendtner MH, Korting HC.
Etude clinique suggérant l'utilisation topique de crèmes à base de Penciclovir 1% dans le traitement des boutons de fièvre, compte tenu des excellents résultats obtenus quelle que soit la phase d'éruption.
RÉSISTANCE AU PENCICLOVIR
Arche Virol. Septembre 2003 ; 148 : 1757-69.
Profil de la sensibilité au penciclovir et de la prévalence de la résistance des isolats du virus de l'herpès simplex à travers onze essais cliniques.
Sarisky RT, Bacon TH, Boon RJ, Duffy KE, Esser KM, Leary J, Locke LA, Nguyen TT, Quail MR, Saltzman R.
travail intéressant qui évalue les résultats de 11 études cliniques présentées partout dans le monde, en se concentrant sur la faible tendance du HSV à développer des mécanismes de résistance au Penciclovir, utilisé par voie topique.
Mode d'emploi et posologie
VECTAVIR®
Crème de penciclovir à 1 % pour usage topique.
Afin de bloquer précocement la propagation du virus et l'aggravation du tableau clinique, il conviendrait d'appliquer la crème sur la région touchée par le processus infectieux le plus tôt possible en prenant soin de renouveler cette application à intervalles de 2 4 heures.
Mises en garde VECTAVIR® Penciclovir
VECTAVIR ® est destiné à un usage labial, il doit donc être appliqué sur les lésions infectieuses des lèvres ou du visage, en prenant soin d'éviter tout contact direct avec les muqueuses et les yeux.
Malgré la simplicité d'utilisation, il serait toujours conseillé de consulter votre médecin avant d'utiliser ce médicament.
La présence d'alcool et de propylène glycol dans VECTAVIR ® pourrait entraîner une irritation de la peau et des réactions locales.
LA GROSSESSE ET L'ALLAITEMENT
La faible absorption systémique, presque nulle, du Penciclovir, qui survient à la suite de l'utilisation topique de VECTAVIR ®, minimise les risques potentiels associés à l'exposition accidentelle du fœtus au médicament susmentionné.
Dans tous les cas, avant d'utiliser VECTAVIR ® pendant la grossesse et pendant la période d'allaitement ultérieure, il est nécessaire de consulter votre gynécologue.
Interactions
Aucune interaction médicamenteuse cliniquement pertinente n'est actuellement connue.
Contre-indications VECTAVIR ® Penciclovir
VECTAVIR ® est contre-indiqué chez les patients hypersensibles à la substance active ou à l'un de ses excipients.
Effets indésirables - Effets secondaires
L'utilisation de VECTAVIR ® pourrait entraîner la survenue d'effets indésirables transitoires et locaux, cliniquement non pertinents tels que brûlures, rougeurs, picotements, urticaire, démangeaisons et gonflements.
Heureusement, ces réactions régressent spontanément une fois le traitement en cours suspendu.
Noter
VECTAVIR ® est un médicament en vente libre.
Les informations sur VECTAVIR ® Penciclovir publiées sur cette page peuvent être périmées ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.