NEFLUAN ® est un médicament à base de sulfate de néomycine + acétonide de fluocinolone + lidocaïne
GROUPE THÉRAPEUTIQUE : Anesthésiques locaux

Indications NEFLUAN ® Néomycine + Fluocinolone acétonide + Lidocaïne
NEFLUAN ® est cliniquement utilisé dans toutes les manœuvres urologiques endoscopiques et instrumentales, dans lesquelles il s'est avéré efficace pour réduire la douleur associée à l'examen, l'inflammation des muqueuses concernées et toutes les séquelles infectieuses possibles.
Mécanisme d'action NEFLUAN ® Néomycine + Fluocinolone acétonide + Lidocaïne
L'utilisation de NEFLUAN ® en milieu clinique est due aux différentes activités thérapeutiques exercées par ses principes actifs, qui font de ce médicament un excellent anesthésique local aux capacités inflammatoires et antibiotiques élevées.
Plus précisément, les activités susmentionnées sont attribuables à la présence de :
- Néomycine : aminoside avec une forte activité antibiotique directe contre les bactéries Gram positives et Gram négatives, garantie par la capacité de l'ingrédient actif à se lier et à inhiber la sous-unité ribosomique 30S, conduisant à la formation de protéines aberrantes capables de compromettre à la fois les activités métaboliques normales de le microorganisme est les caractéristiques structurelles et en particulier l'intégrité membranaire.
- Lidocaïne : anesthésique local largement utilisé par voie topique, capable de contrer la dépolarisation des neurones impliqués dans la transmission nociceptive, exerçant ainsi un effet analgésique important utile dans les manœuvres chirurgicales mineures.
- Acétonide de fluocinolone : corticostéroïde de synthèse dérivé de l'hydrocortisone largement utilisé en dermatologie, capable d'inhiber la synthèse des médiateurs inflammatoires réduisant ainsi les lésions de la muqueuse concernée.
Suite à l'application topique de NEFLUAN ®, une concentration plus importante des principes actifs est observée principalement au niveau du site, garantissant ainsi une action thérapeutique locale particulièrement efficace.
Cependant, il convient de rappeler que la lidocaïne peut être absorbée par voie systémique, métabolisée dans le foie et ensuite excrétée principalement sous forme de catabolites inactifs.
Études réalisées et efficacité clinique
1. LA NEOMYCINE FAVORISE LA STABILITÉ DES VALVES CARDIAQUES
J Biomed Mater Res B Appl Biomater. .
La néomycine améliore la stabilité de la matrice extracellulaire des valves cardiaques bioprothétiques réticulées au glutaraldéhyde.
Friebe VM, Mikulis B, Kole S, Ruffing CS, Sacks MS, Vyavahare NR.
Étude intéressante qui démontre comment l'utilisation de la néomycine peut améliorer la stabilité de la matrice extracellulaire des valves cardiaques, améliorant la formation de liaisons croisées entre les molécules de glutaraldéhyde, responsables des propriétés structurelles du tissu.
2 . NEOMYCINE DANS LA PROPHYLAXIE DE LA CHIRURGIE INTESTINALE
Frère J Surg. Mai 2007, 94 : 546-54.
Essai clinique randomisé sur l'effet des symbiotiques, de la néomycine et de la préparation mécanique de l'intestin sur la fonction de la barrière intestinale chez les patients subissant une colectomie.
Reddy BS, Macfie J, Gatt M, Larsen CN, Jensen SS, Leser TD.
Travail qui démontre comment l'administration de néomycine en association avec des symbiotes avant des manœuvres intestinales invasives peut empêcher la translocation bactérienne et la colonisation par les entérobactéries.
3. HYPERSENSIBILITÉ À LA NÉOMYCINE
Thérapeutique. 1973 novembre ; 49 : 609-13.
Etude dermatologique d'un nouveau composé topique de fluocinonide et de sulfate de néomycine.
Privat Y.
Étude très datée évaluant pour la première fois l'efficacité et la tolérance de l'administration topique combinée de néomycine et d'acétonide de flucinolone pour les maladies dermatologiques.
Mode d'emploi et posologie
NEFLUAN®
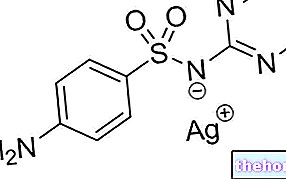
Gel hydrosoluble contenant 2,5 g de chlorhydrate de lidocaïne, 0,5 g de sulfate de néomycine et 0,025 g d'acétonide de flucinolone.
L'utilisation de NEFLUAN ® étant réservée au personnel soignant, l'application de ce gel doit avoir lieu quelques minutes avant l'évaluation instrumentale par le médecin ou le personnel soignant.
Mises en garde NEFLUAN ® Néomycine + Fluocinolone acétonide + Lidocaïne
L'application de NEFLUAN ® doit être effectuée par le personnel soignant dans les structures ambulatoires appropriées.
Il est recommandé d'éviter l'application du gel sur les muqueuses gravement endommagées compte tenu du risque éventuel d'absorption systémique des principes actifs pris.
Une utilisation prolongée et inappropriée du produit pourrait faciliter la formation de souches résistantes à l'antibiothérapie courante, aggravant le tableau clinique du patient et l'exposant à d'éventuelles réactions de sensibilisation.
LA GROSSESSE ET L'ALLAITEMENT
L'utilisation de NEFLUAN ® est généralement déconseillée pendant la grossesse et dans la période d'allaitement ultérieure, étant donné l'absence d'essais cliniques capables de caractériser le profil d'innocuité du médicament chez le fœtus exposé au médicament.
Interactions
Toutes les interactions médicamenteuses possibles capables de faire varier les caractéristiques pharmacocinétiques et pharmacodynamiques de NEFLUAN ® sont dues à la présence de lidocaïne, dont la demi-vie plasmatique pourrait être augmentée par l'administration simultanée de propanolol.
Contre-indications NEFLUAN ® Néomycine + Fluocinolone acétonide + Lidocaïne
L'utilisation de NEFLUAN ® est contre-indiquée en cas d'hypersensibilité aux principes actifs et aux excipients correspondants.
Effets indésirables - Effets secondaires
Bien que l'utilisation de NEFLUAN ® soit généralement sûre et exempte d'effets secondaires cliniquement pertinents, la présence de lidocaïne pourrait déterminer l'apparition d'effets secondaires liés avant tout à d'éventuelles réactions indésirables au principe actif.
La présence de néomycine pourrait au contraire provoquer des réactions d'hypersensibilité locales caractérisées principalement par des démangeaisons, des rougeurs et des gonflements.
Noter
NEFLUAN ® est un médicament d'ordonnance
Les informations sur NEFLUAN ® Néomycine + Acétonide de fluocinolone + Lidocaïne publiées sur cette page peuvent être périmées ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.