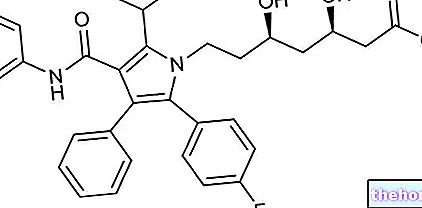
STRIANT ® est un médicament à base de testostérone
GROUPE THÉRAPEUTIQUE : Androgènes, dérivés du 3-oxoandrostène

Indications STRIANT ® - Testostérone
STRIANT ® est un médicament à base de testostérone en comprimés buccaux adhésifs mucus, utilisé dans le traitement des symptômes de déficit constitutif de cette hormone présents dans différentes affections telles que l'hypogonadisme et le retard de puberté, diagnostiqués par des tests cliniques et biochimiques.
Mécanisme d'action de STRIANT ® - Testostérone
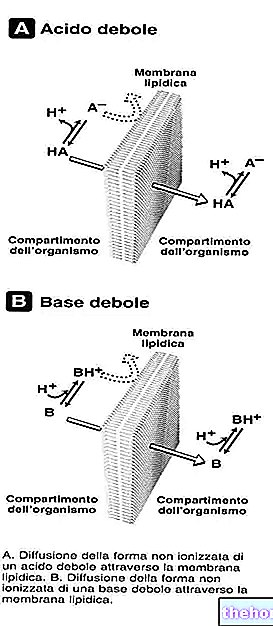
STRIANT ® présente une méthode innovante de prise de testostérone, représentée par des comprimés adhésifs de mucus buccal. Grâce à ces compresses appliquées sur la gencive ou sur les surfaces internes des joues, la testostérone est absorbée par le plexus veineux sous-jacent, et dirigée vers la veine cave supérieure, pénètre dans le système circulatoire sans subir l'effet du premier passage hépatique.
Ce système d'administration particulier permet donc d'atteindre des concentrations plasmatiques maximales en 10 à 12 heures environ, qui tendent à diminuer progressivement 4 heures après le retrait du comprimé.
Liée aux protéines plasmatiques, la testostérone atteint divers tissus, où elle est en partie métabolisée en estradiol et en partie en DHT, qui, lorsqu'elle est liée aux récepteurs des hormones stéroïdes, module le profil d'expression génique de la cellule cible.
L'action thérapeutique de la testostérone trouve donc son origine dans son rôle clé d'assurer la bonne maturation des caractères secondaires masculins, de contrôler l'activité sexuelle, d'induire une phase anabolique globale tant au niveau musculaire, osseux qu'hématopoïétique.
Études réalisées et efficacité clinique
1. COMPARAISON DES SYSTÈMES DE LIVRAISON
J Clin Endocrinol Metab. Mai 2004 ; 89 : 2039-2043.
Une comparaison d'un nouveau système buccal bioadhésif de testostérone, striant, avec un patch adhésif de testostérone chez les hommes hypogonadiques.
Korbonits M, Slawik M, Cullen D, Ross RJ, Stalla G, Schneider H, Reincke M, Bouloux PM, Grossman AB.
Un travail qui démontre comment la prise de testostérone sous forme de comprimés buccaux adhésifs mucus peut restaurer les concentrations physiologiques de testostérone plus rapidement et plus efficacement que d'autres méthodes d'administration telles que les patchs transdermiques.
2. STRIANT, UNE ALTERNATIVE PHARMACOCINÉTIQUEMENT EFFICACE
J Clin Endocrinol Metab. Août 2004 ; 89 : 3821-9.
Le nouveau système buccal de testostérone (Striant) fournit des niveaux physiologiques de testostérone : étude de pharmacocinétique chez des hommes hypogonadiques.
Wang C, Swerdloff R, Kipnes M, Matsumoto AM, Dobs AS, Cunningham G, Katznelson L, Weber TJ, Friedman TC, Snyder P, Levine HL.
Étude pharmacocinétique pure qui démontre comment l'utilisation de Striant, donc l'hypothèse de la testostérone via le mucus buccal, peut garantir une alternative efficace et sûre aux thérapies de remplacement conventionnelles.
3. STRIANT DANS LE « HYPOGONADISME »
Int J Clin Pract. Novembre 2004 ; 58 : 1073-80.
Striant SR : une nouvelle thérapie à la testostérone efficace et pratique pour l'hypogonadisme masculin.
Korbonits M, Kipnes M, Grossman AB.
Nouvelle étude pharmacocinétique réalisée sur des patients souffrant d'hypogonadisme qui démontre comment la prise de Striant peut, en seulement 24 heures, contribuer à l'obtention de concentrations sériques physiologiques de testostérone, garantissant des résultats encore meilleurs que les crèmes ou patchs à base de testostérone.
Mode d'emploi et posologie
STRIANT®
Comprimés buccaux adhésifs de testostérone 30 mg de mucus :
le comprimé buccal adhésif muqueux doit être appliqué sur la région des gencives ou sur la surface interne des joues, en veillant à ce qu'il adhère bien, afin de ne pas être ingéré avec de la nourriture.
La posologie et le moment de la prise doivent nécessairement être définis par le médecin, qui doit en même temps préciser l'importance de la bonne application du patch.
Chez les patients âgés et les adultes, la dose recommandée est généralement de deux comprimés par jour à appliquer à 12 heures d'intervalle.
STRIANT ® Avertissements - Testostérone
L'utilisation de STRIANT ® est subordonnée au diagnostic clinique et biochimique du déficit en testostérone et à un examen médical minutieux, utile pour évaluer la pertinence prescriptive et la compatibilité thérapeutique.
En effet, les patients souffrant d'hypertension, d'épilepsie, d'antécédents familiaux ou d'antécédents de maladies néoplasiques, cardiovasculaires, hépatiques, rénales et métaboliques doivent soigneusement évaluer avec leur médecin le rapport coût/bénéfice associé à cette thérapie, en prévoyant la nécessité éventuelle de contrôles périodiques. nécessaire pour vérifier l'efficacité thérapeutique et l'état de santé psycho-physique.
En fait, le médecin doit immédiatement suspendre la thérapie, dès l'apparition des premiers signes de déséquilibre à la fois organique-fonctionnel et comportemental.
Dans les cas susmentionnés, des contrôles périodiques seraient indiqués, utiles pour évaluer la santé physique et mentale du patient, prévoyant la suspension immédiate du traitement dès l'apparition des premiers effets secondaires.
L'utilisation à l'âge prépubère doit plutôt se faire avec une attention particulière, compte tenu des effets délétères possibles sur la croissance osseuse et sur le développement trop rapide des organes génitaux masculins.
Ce médicament contient du lactose par conséquent, les patients présentant des problèmes héréditaires rares d'intolérance au galactose, de déficit en lactase ou de malabsorption du glucose-galactose ne doivent pas prendre ce médicament.
LA GROSSESSE ET L'ALLAITEMENT
STRIANT ® est un médicament réservé au traitement des maladies du sexe masculin.
Son utilisation chez la femme, en plus d'induire une virilisation, si elle est effectuée pendant la grossesse, pourrait altérer le processus normal de développement fœtal, compromettant certaines fonctions telles que la fonction reproductive.
Interactions
Les différents essais cliniques orientés vers l'étude des propriétés pharmacocinétiques de la testostérone, ont observé trois interactions possibles cliniquement pertinentes, qui sont apparues suite à la prise concomitante de :
- Anticoagulants : responsables d'un risque hémorragique accru ;
- Agents hypoglycémiants : capables d'induire une hypoglycémie chez les patients sous traitement ;
- Corticoïdes : responsables de l'augmentation de la rétention hydrosaline avec œdème et hypertension.
Il est également important de se rappeler que l'apport d'androgènes pourrait modifier certains paramètres de laboratoire, en particulier ceux liés à la fonction hépatique, rénale, lipidique et thyroïdienne.
Contre-indications de STRIANT ® - Testostérone
L'hypothèse de la testostérone est contre-indiquée en cas d'hypersensibilité à la substance active ou à l'un de ses excipients, cancer du sein, néoplasmes œstrogène-dépendants, cancer et hypertrophie de la prostate, antécédent d'hépatocarcinome, hypercalcémie, syndrome néphrotique, grossesse et allaitement.
Effets indésirables - Effets secondaires
Différents essais cliniques et une surveillance post-commercialisation attentive ont le mieux caractérisé les effets secondaires d'un traitement hormonal substitutif à base de testostérone.
Ces effets, qui surviennent avec une fréquence et une gravité clinique accrues lorsqu'ils sont utilisés en dehors des indications médicales (voir dopage), impliquent différents organes et systèmes avec :
- prise de poids, rétention d'eau, œdème et hypertension dans les cas les plus sévères ;
- crampes musculaires et myalgie;
- acné, hirsutisme, alopécie ;
- Nervosité et symptômes psychiatriques ;
- Troubles de la sphère sexuelle dus à l'hyperstimulation ;
- Hypertrophie prostatique;
- Gynécomastie ;
- Oliogoazoospermie avec atrophie testiculaire dans les cas graves.
Aux effets secondaires mentionnés ci-dessus, typiques de la thérapie androgénique, s'ajoutent ceux liés au mode particulier de prise tels que l'irritation au site d'application, la fatigue, la gingivite et les altérations du goût.
Noter
STRIANT ® ne peut être vendu que sur prescription médicale.
STRIANT ® est un agent anabolisant, son utilisation, en dehors des prescriptions médicales à titre pathologique, est donc interdite en et hors compétition.
Les informations sur STRIANT ® - Testostérone publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.