Qu'est-ce que Ravicti - Glycerol Phenylbutyrate et dans quel cas est-il utilisé ?
Ravicti est un médicament utilisé pour le traitement à long terme des troubles du cycle de l'urée chez les adultes et les enfants âgés de deux mois et plus lorsque la maladie ne peut pas être prise en charge par des changements alimentaires seuls. du corps en raison d'un manque de certaines enzymes hépatiques. Dans l'organisme, l'azote résiduaire est transformé en ammoniac, ce qui est nocif s'il s'accumule. Ravicti est utilisé chez les patients présentant un déficit en une ou plusieurs des enzymes suivantes : carbamoyl phosphate synthase I, ornithine carbamyltransférase, argininosuccinate synthetase, argininosuccinate lyase, arginase I et ornithine translocase.
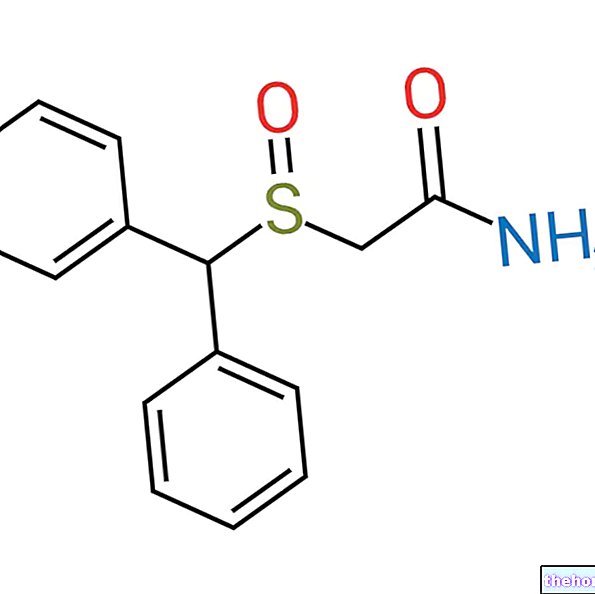
Ravicti contient le principe actif phénylbutyrate de glycérol.
Étant donné que le nombre de patients atteints de troubles du cycle de l'urée est faible, ces maladies sont considérées comme « rares », et Ravicti a été désigné « médicament orphelin » (un médicament utilisé dans les maladies rares) le 10 juin 2010.
Comment Ravicti - Glycerol Phenylbutyrate est-il utilisé ?
Ravicti est disponible sous forme de liquide (1,1 g/ml) à prendre par voie orale ou par un tube qui passe par le nez et dans l'estomac ou qui passe par le ventre jusqu'à l'estomac. Il n'est délivré que sur ordonnance et doit être prescrit par un médecin expérimenté dans le traitement des patients présentant des troubles du cycle de l'urée.
Les protéines sont une source d'azote, c'est pourquoi Ravicti doit être utilisé en association avec un régime spécial pauvre en protéines pour réduire l'apport en azote et, dans certains cas, des compléments alimentaires (basés sur l'apport quotidien en protéines nécessaire à la croissance et au développement).
La dose de Ravicti dépend de l'alimentation, de la taille et du poids du patient.Pendant le traitement, des analyses de sang doivent être effectuées régulièrement pour ajuster la dose quotidienne.La dose quotidienne de Ravicti doit être divisée en quantités égales et administrée à chaque repas. Pour plus d'informations, voir le Résumé des Caractéristiques du Produit (également inclus dans l'EPAR).
Un traitement Ravicti peut être nécessaire tout au long de la vie, à moins que le patient ne reçoive une greffe du foie réussie
Comment Ravicti - Glycerol Phenylbutyrate agit-il ?
Le principe actif de Ravicti, le phénylbutyrate de glycérol, est converti en phénylacétate dans l'organisme. Le phénylacétate se combine avec l'acide aminé glutamine présent dans les protéines, qui contient de l'azote, pour former une substance qui peut être éliminée de l'organisme par les reins. niveaux dans le corps, réduisant la quantité d'ammoniac produite.
Quel bénéfice Ravicti - Glycerol Phenylbutyrate a-t-il montré au cours des études ?
Une étude portant sur 88 adultes atteints de troubles du cycle de l'urée a comparé Ravicti au phénylbutyrate de sodium (un autre médicament utilisé pour traiter les troubles du cycle de l'urée). Le principal critère d'évaluation de l'efficacité était la variation des taux d'ammoniac dans le sang après 4 semaines de traitement. L'étude a révélé que Ravicti est au moins aussi efficace que le médicament de comparaison pour contrôler le niveau d'ammoniac dans le sang : le niveau moyen estimé d'ammoniac était d'environ 866 micromoles par litre chez les patients traités par Ravicti et de 977 micromoles par litre chez les patients traités par sodium. phénylbutyrate. Des données supplémentaires issues d'études complémentaires ont montré des effets similaires de Ravicti chez les enfants âgés de 2 mois et plus.
Quel est le risque associé à Ravicti - Glycerol Phenylbutyrate ?
Les événements indésirables les plus fréquents de Ravicti (pouvant affecter plus de 5 personnes sur 100) sont la diarrhée, les flatulences (passage de gaz), les maux de tête, la diminution de l'appétit, les vomissements, la fatigue, la sensation de malaise et une odeur anormale de la peau.
Ravicti ne doit pas être utilisé pour traiter l'hyperammoniémie aiguë (augmentation soudaine des taux d'ammoniac dans le sang).Pour la liste complète des effets indésirables rapportés avec Ravicti et leurs limites, voir la notice.
Pourquoi Ravicti - Glycerol Phenylbutyrate a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) de l'Agence a décidé que les avantages de Ravicti sont supérieurs à ses risques et a recommandé qu'il soit approuvé pour une utilisation dans l'UE.
Ravicti s'est avéré efficace pour réduire le taux d'ammoniac dans le sang chez les patients présentant des troubles du cycle de l'urée. Ravicti est un médicament à libération prolongée, ce qui signifie que la substance active est libérée régulièrement tout au long de la journée. Par conséquent, également l'élimination des déchets l'azote se produit constamment et cela produit un meilleur contrôle des niveaux d'ammoniac dans le sang pendant toute la journée. Pour la même raison, Ravicti ne doit pas être utilisé pour traiter l'hyperammoniémie aiguë, pour laquelle des traitements d'action plus rapide sont nécessaires.
De plus, étant donné qu'il est disponible sous forme liquide, le comité a estimé que Ravicti peut être plus acceptable, en particulier pour les enfants, que d'autres médicaments granulés à ajouter aux aliments ; la formulation liquide facilite également son administration par tube chez les patients incapables d'avaler en raison de problèmes neurologiques.
Les effets secondaires de Ravicti se sont principalement produits dans l'intestin et ont été considérés comme gérables.Cependant, des données supplémentaires sur la sécurité à long terme de Ravicti sont attendues.
Quelles mesures sont prises pour garantir l'utilisation sûre et efficace de Ravicti - Glycerol Phenylbutyrate ?
Un plan de gestion des risques a été élaboré pour garantir que Ravicti est utilisé de la manière la plus sûre possible. Sur la base de ce plan, des informations de sécurité ont été incluses dans le résumé des caractéristiques du produit et la notice de Ravicti, y compris les précautions appropriées à suivre par les professionnels de santé et les patients.
De plus, la société qui commercialise Ravicti mettra en place un registre de patients pour obtenir plus d'informations sur les avantages à long terme et la sécurité du médicament.
Plus d'informations sur Ravicti - Glycérol Phenylbutyrate
Pour plus d'informations sur le traitement par Ravicti, veuillez lire la notice (incluse dans l'EPAR) ou contacter votre médecin ou votre pharmacien.
Les informations sur Ravicti - Glycerol Phenylbutyrate publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.