XELEVIA ® un médicament à base de sitagliptine
GROUPE THÉRAPEUTIQUE : Agents hypoglycémiants oraux - Inhibiteurs de la DPP-4

Indications XELEVIA ® - Sitagliptine
XELEVIA ® est indiqué dans le traitement du diabète sucré de type II lorsque les mesures non pharmacologiques ou la monothérapie par metformine, sulfamides hypoglycémiants et agonistes PPAR gamma ne donnent pas les résultats attendus.
La thérapie combinée entre la Sitagliptine et les principes actifs précités garantit donc un meilleur contrôle glycémique que les monothérapies.
Mécanisme d'action XELEVIA ® - Sitagliptine
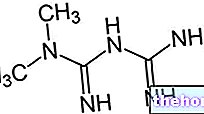
La sitagliptine, principe actif contenu dans XELEVIA® appartient aux inhibiteurs de la DPP-4, une nouvelle famille pharmacologique caractérisée par l'effet hypoglycémiant.
Pris par voie orale, en fait, il est absorbé dans l'intestin, atteignant la concentration plasmatique maximale dans les 4 premières heures suivant la prise avec une biodisponibilité absolue de 87 %.
Réversiblement lié aux protéines plasmatiques pour environ 40 %, il exerce son action thérapeutique en inhibant l'enzyme DPP-4 (Dipepetidil Peptidase 4), responsable de l'hydrolyse irréversible des hormones appelées incrétines.
Ces derniers, à savoir le GLP1 (Glucagon-like peptide 1) et le GIP (Glucose-dependent insulinotropic peptide), peuvent agir au niveau pancréatique, par des voies intracellulaires, favorisant la production et la sécrétion d'insuline et inhibant celle de glucagon.
Cette action importante se traduit par une implication métabolique évidente, c'est-à-dire par un effet hypoglycémiant soutenu à la fois par la plus grande disponibilité de l'insuline et par la synthèse réduite du glucose endogène.
Une fois son action terminée, la sitagliptine est éliminée sous forme inchangée principalement par le rein, alors que seule une faible partie de la dose prise subit un métabolisme hépatique.
Études réalisées et efficacité clinique
1. SITAGLIPTINE ET ALBUMINURIE
Endocr J. 30 janvier 2011 ; 58 : 69-73. Publication en ligne du 28 décembre 2010.
La sitagliptine réduit l'albuminurie chez les patients atteints de diabète de type 2.
Hattori S.
Étude intéressante qui a montré comment l'administration de sitagliptine chez les patients diabétiques du deuxième type, peut réduire l'albuminurie, non pas en agissant sur le taux de filtration glomérulaire, mais en améliorant cette série d'aspects métaboliques connexes tels que l'hyperglycémie, la pression artérielle et le niveau d'inflammation , particulièrement évident chez ces patients.
2. SITAGLIPTINE : ESSAI CLINIQUE
Diabète Metab Syndr Obes. 29 mars 2010, 3 : 31-41.
Utilisation des inhibiteurs de la DPP-4 dans le diabète de type 2 : focus sur la sitagliptine.
Ahrén B.
L'essai clinique de la sitagliptine au cours des dernières années a montré une efficacité particulière du médicament dans l'amélioration du profil glycémique, soutenant la fonctionnalité des cellules bêta pancréatiques, avec un profil de sécurité élevé capable de minimiser le risque d'hypoglycémie, de prise de poids et de troubles gastro-intestinaux.
3.SITAGLIPTINE ET METFORMINE
Wien Klin Wochenschr. 31 mars 2011.
Efficacité et tolérance de la sitagliptine chez les patients diabétiques de type 2 insuffisamment contrôlés par la metformine. Une étude observationnelle prospective dans les soins primaires autrichiens.
Ludvik B, Daniela L.
Essai clinique mené sur environ 2300 patients diabétiques, préalablement traités par des doses maximales de metformine sans atteindre un contrôle glycémique adéquat, dans lequel la prise concomitante de sitagliptine a permis une nette amélioration du profil glycémique, avec une diminution de l'hémoglobine glycosylée et de la glycémie à jeun et postprandiale. .
Mode d'emploi et posologie
XELEVIA® comprimés de sitagliptine 25 mg : en pratique clinique normale, la posologie de XELEVIA ® la plus utilisée est celle de 100 mg pris en une seule prise quotidienne.
Cependant, il est d'une importance fondamentale que la posologie correcte et les modalités relatives de prise en charge soient établies par son médecin en fonction de la présence éventuelle d'autres thérapies en cours, des conditions physiopathologiques du patient et de son état métabolique.
XELEVIA ® Avertissements - Sitagliptine
Il convient de rappeler que la prise en charge des maladies diabétiques de second type doit inclure parmi ses outils thérapeutiques non seulement des mesures pharmacologiques mais également des actions quotidiennes telles qu'une alimentation saine et un mode de vie correct.
Pendant le traitement par XELEVIA ®, il est nécessaire de surveiller périodiquement les taux glycémiques, en particulier en association avec des sulfonylurées, pour éviter le risque d'hypoglycémie et compenser de manière à améliorer les déséquilibres métaboliques.
Le risque d'hypoglycémie pourrait en effet réduire les capacités de perception du patient, rendant dangereux l'utilisation de machines et la conduite automobile.
LA GROSSESSE ET L'ALLAITEMENT
XELEVIA® est contre-indiqué pendant la grossesse et l'allaitement, compte tenu de l'absence de données cliniques sur le profil de tolérance de ce médicament sur la santé fœtale, et compte tenu de la disponibilité sur le marché d'antidiabétiques dont l'activité est mieux caractérisée.
Interactions
Les interactions possibles entre la sitagliptine et d'autres principes actifs sont particulièrement rares, compte tenu du faible métabolisme hépatique auquel le médicament est soumis.
De manière générale, la prise de cyclosporine et de digoxine concomitamment à celle de sitagliptine, pourrait déterminer l'apparition d'effets secondaires suite à la modification du profil pharmacocinétique.
Des études ont montré que chez les patients présentant une insuffisance rénale, il est possible de décrire des interactions, souvent cliniquement insignifiantes, entre de puissants inhibiteurs du CYP3A4 et la sitagliptine.
Cependant, étant donné l'utilisation de la sitagliptine en association avec d'autres hypoglycémiants oraux, les interactions possibles et les ajustements posologiques doivent être pris en compte.
Contre-indications XELEVIA ® - Sitagliptine
XELEVIA® est contre-indiqué en cas d'hypersensibilité à la substance active ou à l'un de ses excipients, chez les patientes présentant une insuffisance rénale, un diabète de type I et une acidocétose diabétique, et pendant la grossesse et l'allaitement.
Effets indésirables - Effets secondaires
L'essai clinique, qui représente aujourd'hui la principale source de données sur le profil de sécurité de la sitagliptine, a montré que l'administration de XELEVIA® est bien tolérée et dans certains cas (thérapie combinée) peut réduire l'incidence des effets secondaires observés ultérieurement. dose en monothérapie.
En général, les effets secondaires les plus décrits étaient des réactions gastro-intestinales telles que nausées, vomissements, diarrhée et flatulences, maux de tête et vertiges et œdème périphérique.
Des effets indésirables et des réactions d'hypersensibilité cliniquement plus significatifs ont été observés au contraire rarement et concernaient principalement la fonction cardiaque et osseuse ainsi que le profil hématologique.
Noter
XELEVIA ® ne peut être commercialisé que sur prescription médicale stricte.
Les informations sur XELEVIA ® - Sitagliptine publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.