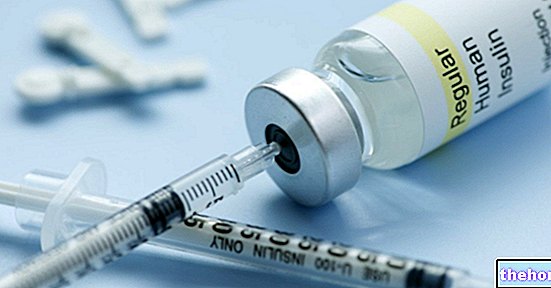
NOVORAPID ® un médicament à base d'insuline asparte.
GROUPE THÉRAPEUTIQUE : Insuline asparte pour usage injectable - insulines et analogues

Indications NOVORAPID ® - Insuline asparte
NOVORAPID ® est un médicament utilisé dans le contrôle du métabolisme du glucose chez les patients atteints de diabète sucré.
Mécanisme d'action NOVORAPID ® - Insuline aspart
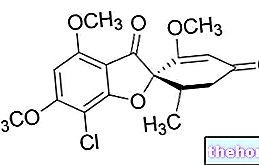
NOVORAPID ® est un médicament hypoglycémiant à base d'insuline asparte, définie ainsi comme ayant une substitution d'acide aminé (Proline avec Aspartate) par rapport à la séquence normale de cette hormone.
Cette variation d'un seul acide aminé influence de manière significative les propriétés pharmacocinétiques de l'hormone qui, en réduisant la tendance normale à former des hexamères, est rapidement absorbée après administration sous-cutanée, provoquant l'apparition de l'effet hypoglycémiant après environ 10 à 20 minutes, l'optimum entre le premier et troisième heures et une persistance de l'effet pendant environ 4 heures.
Du point de vue pharmacodynamique, cependant, le mécanisme d'action reste inchangé, permettant à l'insuline d'interagir avec son récepteur spécifique exprimé par les cellules des tissus sensibles à l'insuline et d'augmenter l'absorption et l'utilisation du glucose, réduisant ainsi les concentrations sanguines.
L'effet anabolisant important de cette hormone d'une part (inducteur de la synthèse du glycogène, de la synthèse des protéines et de la synthèse des lipides) et la capacité d'agir en inhibant les voies métaboliques utiles à la production de glucose, permet à l'insuline d'être l'hormone hypoglycémiante par excellence.
Études réalisées et efficacité clinique
1. ASPART INSULINE ET DIABÈTE GESTATIONNEL
Santé des femmes (Lond Engl). Mars 2008, 4 : 119-24.
L'insuline asparte dans la grossesse diabétique : état de l'art.
Mathiesen ER.
Étude importante montrant comment le traitement du diabète gestationnel par l'insuline asparte peut assurer un bon contrôle glycémique post-prandial, réduisant la fréquence des événements hypoglycémiques et réduisant le nombre de fausses couches prématurées.
2. INSULINE ET ANALOGUES D'INSULINE
Diabétologie. Août 2010 ; 53 : 1743-53. Publication en ligne du 28 avril 2010.
Les analogues de l'insuline activent différemment les isoformes des récepteurs de l'insuline et la signalisation post-récepteur.
Sciacca L, Cassarino MF, Genua M, Pandini G, Le Moli R, Squatrito S, Vigneri R.
Il est connu que les analogues de l'insuline diffèrent de l'insuline humaine en raison des différentes cinétiques d'action. Cette étude entièrement italienne démontre également comment ces différences peuvent également affecter la dynamique moléculaire avec des mécanismes intracellulaires activés de manière différente selon le type d'insuline utilisé. Ces études pourraient partiellement clarifier certaines variations importantes sur les différentes efficacités des analogues de l'insuline.
3. ASPART INSULINE ET ACTION ANTI-INFLAMMATOIRE
Basic Clin Pharmacol Toxicol. 23 avril 2011.
Effets de l'insuline asparte au repas et de l'insuline NPH au coucher sur l'inflammation postprandiale et la fonction des cellules endothéliales chez les patients atteints de diabète de type 2.
Bladbjerg EM, Henriksen JE, Akram S, Gram J.
Bien que l'insuline asparte ait un mode d'action plus rapide et garantisse une baisse significative de la glycémie postprandiale par rapport à l'insuline ordinaire, les effets sur la modulation des marqueurs inflammatoires et sur la santé endothéliale des patients diabétiques de second type sont quasiment identiques.
Mode d'emploi et posologie
NOVORAPID® 100 UI/ml d'insuline : 5 cartouches de 3 ml ou stylo prérempli de 3 ml : la posologie de l'insuline asparte à utiliser dans le traitement de la pathologie diabétique dépend strictement de l'état physique du patient et de son tableau clinique.
En général, la posologie utile doit être comprise entre 0,5 et 1 UI / kg de poids corporel et l'administration de NOVORAPID ® doit être effectuée immédiatement avant un repas, compte tenu des temps d'action particulièrement rapides.
Il ne faut pas oublier que la formulation posologique et tout autre ajustement doivent être déterminés par votre médecin.
Mises en garde NOVORAPID ® - Insuline asparte
Pour assurer un bon contrôle glycémique, il est nécessaire pour le patient diabétique de subir une évaluation périodique des concentrations sériques de glucose avant et pendant la période de traitement, et éventuellement d'adapter l'insulinothérapie relative.
Il est également nécessaire que le médecin informe le patient de la bonne méthode de préparation, d'administration et de conservation du médicament et des risques potentiels, afin de pouvoir reconnaître rapidement les signes d'hypoglycémie et intervenir avant que la crise hypoglycémique ne se produise.
L'insuline asparte, compte tenu de son mode d'action plus rapide, peut nécessiter une administration plus fréquente que l'insuline humaine soluble.
Tout ajustement posologique, modification du médicament utilisé ou arrêt du traitement doit être supervisé par le personnel médical.
En cas d'insuffisance rénale, il peut être nécessaire de réduire la dose du médicament utilisé, contrairement aux infections qui peuvent nécessiter une augmentation des concentrations normalement utilisées.
L'apparition possible d'une hypoglycémie pourrait réduire les capacités de perception du patient, rendant l'utilisation de machines et la conduite de voitures dangereuses.
LA GROSSESSE ET L'ALLAITEMENT
Les études menées sur l'utilisation de l'insuline asparte chez la femme enceinte n'ont rapporté aucun effet secondaire sur le fœtus ou la femme enceinte.
Par conséquent, NOVORAPID ® pourrait être utilisé efficacement dans le traitement du diabète gestationnel, en ajustant la dose utilisée aux conditions physiopathologiques de la patiente et à la période de gestation.
Interactions
Comme l'insuline classique, l'insuline asparte est également capable d'interagir avec de nombreux principes actifs.
Plus précisément, l'interaction avec les hypoglycémiants oraux, l'octréotide, les anti-MAO, les bêta-bloquants, les inhibiteurs de l'ECA, les salicylates, l'alcool et les stéroïdes anabolisants pourrait renforcer les effets hypoglycémiants du médicament en réduisant le besoin d'insuline elle-même, tandis que la prise simultanée de les contraceptifs oraux, les thiazidiques, les glucocorticoïdes, les hormones thyroïdiennes et les sympathomimétiques pourraient réduire l'effet thérapeutique de NOVORAPID ®
Contre-indications NOVORAPID ® - Insuline aspart
NOVORAPID ® est contre-indiqué en cas d'hypoglycémie et d'hypersensibilité à l'insuline humaine ou à ses excipients.
Effets indésirables - Effets secondaires
Le traitement par NOVORAPID ® pourrait, comme les autres traitements hypoglycémiants, être associé à des états hypoglycémiques reconnaissables par des signes classiques tels que sueurs froides, pâleur de la peau, nervosité, tremblements, anxiété, fatigue, faiblesse, confusion, difficultés de concentration, maux de tête, nausées, palpitations, troubles visuels et dans les cas les plus graves, la perte de connaissance et la mort.
De plus, des manifestations telles que des rougeurs, des démangeaisons et des douleurs peuvent survenir au site d'injection, tandis que l'application de l'injection au même site pendant des périodes prolongées peut être responsable d'une lipoatrophie localisée.
Des réactions d'hypersensibilité telles qu'éruption cutanée, dyspnée et difficultés respiratoires, œdème et chute brutale de la pression artérielle ont été rarement rapportées.
Noter
NOVORAPID ® vendu uniquement sur prescription médicale.
NOVORAPID ® appartient à la classe de dopage : Hormones et substances apparentées (interdites en et hors compétition).
Les informations sur NOVORAPID ® - Insulin aspart publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.








.jpg)






.jpg)



.jpg)


