Ingrédients actifs : Lisinopril, Hydrochlorothiazide
Comprimés ZESTORETIC 20 mg + 12,5 mg
Pourquoi Zestoretic est-il utilisé ? Pourquoi est-ce?
CATÉGORIE PHARMACOTHERAPEUTIQUE
Inhibiteur de l'ECA (inhibiteur de l'enzyme de conversion de l'angiotensine) et diurétique, en association.
INDICATIONS THÉRAPEUTIQUES
Zestoretic est indiqué pour le traitement de l'hypertension artérielle essentielle chez les patients pour lesquels un traitement combiné est approprié.
Contre-indications Quand Zestoretic ne doit pas être utilisé
Hypersensibilité au lisinopril, à l'un des excipients ou à d'autres inhibiteurs de l'enzyme de conversion de l'angiotensine (ECA
Hypersensibilité à l'hydrochlorothiazide et à d'autres médicaments dérivés des sulfamides.
Anurie.
Antécédents d'œdème de Quincke liés à un traitement antérieur par des inhibiteurs de l'enzyme de conversion de l'angiotensine
Angiœdème héréditaire ou idiopathique.
Deuxième et troisième trimestre de la grossesse (voir Mises en garde spéciales).
Insuffisance rénale sévère (clairance de la créatinine < 30 ml/min).
Insuffisance hépatique sévère.
L'utilisation concomitante de Zestoretic avec des produits contenant de l'aliskirène est contre-indiquée chez les patients atteints de diabète sucré ou d'insuffisance rénale (DFG < 60 ml/min/1,73 m2)
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Zestoretic
Avant de commencer le traitement, informez votre médecin si :
- vous avez une sténose de la valve aortique ou mitrale / une cardiomyopathie hypertrophique ou une sténose de l'artère rénale ;
- vous avez des problèmes tels que diarrhée ou vomissements, goutte, problèmes de foie ou de rein, si vous subissez une dialyse ou une transplantation rénale ou un régime pauvre en sel ou si vous suivez des traitements de désensibilisation pour certaines allergies, par exemple pour les piqûres d'insectes ;
- vous avez eu des réactions allergiques, éventuellement avec gonflement des mains, des pieds ou des chevilles, du visage, des lèvres, de la langue et/ou de la gorge avec difficulté à respirer. Informez votre médecin si une réaction similaire s'est déjà produite chez un membre de votre famille pour quelque raison que ce soit.
- un traitement de séparation du sang (aphérèse) est nécessaire
- vous prenez l'un des médicaments suivants utilisés pour traiter l'hypertension artérielle :
- un antagoniste des récepteurs de l'angiotensine II (AIIRA) (également connu sous le nom de sartans, par exemple valsartan, telmisartan, irbésartan), en particulier si vous avez des problèmes rénaux liés au diabète.
- aliskiren.
Votre médecin peut vérifier votre fonction rénale, votre tension artérielle et la quantité d'électrolytes (tels que le potassium) dans votre sang à intervalles réguliers. Voir aussi ce qui est rapporté sous la rubrique « Contre-indications ».
Signalez également à votre médecin si vous souffrez d'hypotension (pression artérielle basse pouvant se manifester par une sensation de faiblesse ou des étourdissements).
Ethnicité
Les inhibiteurs de l'enzyme de conversion (inhibiteurs de l'ECA) provoquent un angio-œdème plus fréquemment chez les patients noirs que chez les patients non noirs. à une prévalence plus élevée de faibles concentrations de rénine dans la population hypertendue noire.
Lithium
L'association d'inhibiteurs de l'ECA et de lithium et de lisinopril n'est généralement pas recommandée (voir Interactions).
Un déséquilibre électrolytique
Comme pour tout patient sous traitement diurétique, une détermination périodique des électrolytes sériques doit être effectuée à des intervalles appropriés.
Les thiazidiques, y compris l'hydrochlorothiazide, peuvent provoquer un déséquilibre hydrique et électrolytique (hypokaliémie, hyponatrémie et alcalose hypochlorémique).Les signes indicateurs d'un déséquilibre hydrique ou électrolytique sont bouche sèche, soif, faiblesse, léthargie, somnolence, crampes ou douleurs musculaires, fatigue musculaire, hypotension, oligurie , tachycardie et troubles gastro-intestinaux tels que nausées et vomissements.
Une hyponatrémie de dilution peut survenir chez les patients œdémateux par temps chaud. Une carence en chlorure est généralement bénigne et ne nécessite pas de traitement. Il a été démontré que les thiazidiques augmentent l'excrétion urinaire de magnésium, ce qui peut entraîner une hypomagnésémie.
Les thiazidiques peuvent diminuer l'excrétion urinaire du calcium et provoquer des augmentations légères et intermittentes du calcium. Une hypercalcémie marquée peut révéler une hyperparathyroïdie asymptomatique. Le traitement thiazidique doit être interrompu avant que des tests de la fonction parathyroïdienne ne soient effectués.
Hyperkaliémie
Des élévations de la kaliémie ont été observées chez certains patients traités par des inhibiteurs de l'ECA, y compris le lisinopril. Les patients à risque de développer une hyperkaliémie comprennent les patients atteints d'insuffisance rénale, de diabète sucré ou ceux recevant un traitement concomitant par des diurétiques d'épargne potassique, des suppléments potassiques ou des substituts de sel contenant du potassium, ou les patients sous d'autres médicaments avec lesquels une augmentation de la kaliémie (par ex. héparine). Si l'utilisation concomitante des agents ci-dessus est jugée nécessaire, une surveillance régulière de la kaliémie est recommandée (voir Interactions).
Patients diabétiques
Chez les patients diabétiques traités par des antidiabétiques oraux ou de l'insuline, le contrôle glycémique doit être étroitement surveillé pendant le premier mois de traitement par un inhibiteur de l'ECA (voir Interactions).
Neutropénie et agranulocytose
Une neutropénie/agranulocytose, une thrombocytopénie et une anémie ont été rapportées chez des patients recevant des inhibiteurs de l'ECA. Chez les patients ayant une fonction rénale normale et en l'absence d'autres facteurs de complication, une neutropénie survient rarement. La neutropénie et l'agranulocytose disparaissent après l'arrêt du traitement par IEC.
Le lisinopril doit être administré avec une extrême prudence aux patients présentant une maladie du collagène, ceux sous traitement immunosuppresseur, allopurinol ou procaïnamide, ou qui présentent une combinaison de ces facteurs de complication, en particulier en cas d'insuffisance rénale antérieure. Certains de ces patients ont développé des infections sévères qui, dans quelques cas, n'ont pas répondu à une antibiothérapie intensive. Si ces patients sont traités par le lisinopril, une surveillance périodique de la numération des globules blancs est recommandée et les patients doivent être informés de la nécessité de signaler tout épisode d'infection.
La toux
Des cas de toux ont été rapportés lors de l'utilisation d'inhibiteurs de l'ECA.Ceci est typiquement non productif, persistant et disparaît à l'arrêt du traitement.
La toux induite par les IEC doit être prise en compte dans le diagnostic différentiel de la toux.
La dose initiale peut provoquer une chute de la pression artérielle supérieure à celle qui se produit par la suite avec la poursuite du traitement. Une sensation de faiblesse ou de vertige peut survenir ; dans ces cas, il peut être utile de s'allonger. Si les symptômes persistent, consultez votre médecin.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Zestoretic
Informez votre médecin ou pharmacien si vous prenez ou avez pris récemment tout autre médicament, même sans ordonnance.
En particulier, avant de commencer le traitement, informez votre médecin si :
- vous prenez d'autres médicaments, en particulier des diurétiques (y compris des épargneurs de potassium), d'autres médicaments antihypertenseurs (pour le traitement de l'hypertension artérielle), y compris ceux contenant de l'aliskiren, qui, lorsqu'ils sont administrés simultanément, peuvent augmenter l'effet antihypertenseur, de l'indométacine (ou d'autres médicaments pour le traitement). de l'arthrite ou des douleurs musculaires), le lithium (pour certains problèmes psychiatriques), l'or injectable (pour le traitement de la polyarthrite rhumatoïde), les AINS (anti-inflammatoires non stéroïdiens) qui, lorsqu'ils sont administrés en association avec des IEC, peuvent diminuer l'effet antihypertenseur et les patients présentant une insuffisance rénale peuvent entraîner une « réduction supplémentaire de la fonction rénale ; allopurinol, agents cytostatiques, immunosuppresseurs et procaïnamide qui, lorsqu'ils sont administrés avec des inhibiteurs de l'ECA, peuvent augmenter le risque de leucopénie ;
- vous souffrez de diabète, car les diurétiques thiazidiques peuvent entraîner la nécessité d'ajuster la dose d'antidiabétiques, y compris l'insuline ;
- vous prenez des médicaments contre l'hypertension contenant de l'aliskiren et vous souffrez de diabète sucré ;
- vous prenez des suppléments de potassium ou des substituts de sel contenant du potassium ;
- vous prenez des médicaments contre l'hypertension contenant de l'aliskirène et avez des problèmes rénaux ;
- prenez des médicaments induisant des torsades de pointes (certains antiarythmiques, certains antipsychotiques),
- vous prenez de la nitroglycérine et d'autres nitrates ou d'autres vasodilatateurs ;
- prenez des relaxants musculaires (par exemple, du chlorure de tubocurarine)
- vous prenez des antidépresseurs tricycliques, des antipsychotiques et des anesthésiques,
- prenez des sympathomimétiques;
- vous prenez de l'amphotéricine B (par voie parentérale), de la carbénoxolone ou des laxatifs stimulants ;
- vous prenez des sels de calcium ;
- prenez des glycosides cardiaques;
- vous prenez de la cholestyramine et du colestipol ;
- vous prenez du triméthoprime ;
- vous prenez du sotalol ;
- vous prenez de la cyclosporine.
Lorsqu'ils sont administrés ensemble, les médicaments suivants peuvent interagir avec les diurétiques thiazidiques : Alcool - Barbituriques - Narcotiques : Une potentialisation de la chute de pression en position debout peut se produire.
Corticoïdes, ACTH : déplétion électrolytique intensifiée notamment hypokaliémie.
Amines pressives (par exemple adrénaline) : une diminution de la réponse aux amines pressives est possible, mais pas de manière à exclure leur utilisation.
Votre médecin devra peut-être modifier votre dose et/ou prendre d'autres précautions :
- Si vous prenez un antagoniste des récepteurs de l'angiotensine II (AIIRA) ou de l'aliskiren (voir également les informations sous Contre-indications et "Précautions d'emploi").
Avertissements Il est important de savoir que :
En cas d'hospitalisation, informer le personnel médical et notamment l'anesthésiste, en cas d'intervention chirurgicale, du traitement en cours par Zestoretic. Il sera également conseillé d'informer votre dentiste si une anesthésie dentaire est administrée.
L'innocuité et l'efficacité de Zestoretic chez les enfants n'ont pas été établies, par conséquent, le médicament ne doit pas être administré aux enfants.
Le médicament est destiné à un usage personnel uniquement et ne doit jamais être pris par d'autres.
Fertilité, grossesse et allaitement
Demandez conseil à votre médecin ou à votre pharmacien avant de prendre tout médicament.
Informez votre médecin si vous êtes enceinte ou souhaitez le devenir ou si vous allaitez ou souhaitez allaiter.
Grossesse
Inhibiteurs de l'ECA :
L'utilisation d'inhibiteurs de l'ECA n'est pas recommandée pendant le premier trimestre de la grossesse.L'utilisation des inhibiteurs de l'ECA est contre-indiquée pendant les deuxième et troisième trimestres de la grossesse (voir Contre-indications).
Le traitement par inhibiteur de l'ECA ne doit pas être instauré pendant la grossesse.
Les preuves épidémiologiques sur le risque de tératogénicité suite à une exposition aux inhibiteurs de l'ECA au cours du premier trimestre de la grossesse n'ont pas été concluantes ; cependant, une légère augmentation du risque ne peut être exclue, en particulier pour les systèmes cardiovasculaire et nerveux central. Si le lisinopril est utilisé pendant le premier trimestre de la grossesse, les patientes doivent être informées des risques potentiels pour le fœtus.
Pour les patientes qui envisagent une grossesse, le médecin doit être informé immédiatement car des traitements antihypertenseurs alternatifs avec un profil d'innocuité prouvé pour une utilisation pendant la grossesse doivent être utilisés, à moins que la poursuite du traitement avec un médicament ne soit considérée comme essentielle.
Lorsqu'une grossesse est diagnostiquée, le médecin doit être informé immédiatement car le traitement par les inhibiteurs de l'ECA doit être arrêté immédiatement et, le cas échéant, un traitement alternatif doit être instauré. Si une exposition à un IEC a eu lieu à partir du deuxième trimestre de la grossesse, un contrôle échographique de la fonction rénale et du crâne est recommandé.Les nouveau-nés dont la mère a reçu des IEC doivent être étroitement surveillés pour détecter une hypotension, une oligurie et une hyperkaliémie (voir Contre-indications).
Hydrochlorothiazide :
L'expérience avec l'hydrochlorothiazide pendant la grossesse est limitée, en particulier au cours du premier trimestre. Les études chez l'animal sont insuffisantes.
L'hydrochlorothiazide traverse le placenta. En raison de son mécanisme d'action, l'utilisation de l'hydrochlorothiazide au cours des deuxième et troisième trimestres de la grossesse peut compromettre la perfusion placentaire fœtale et provoquer des effets fœtaux et néonatals tels qu'un ictère, des troubles de l'équilibre électrolytique et une thrombocytopénie.
L'hydrochlorothiazide ne doit pas être utilisé pour le traitement de l'œdème gestationnel, de l'hypertension gestationnelle ou de la prééclampsie en raison du risque de diminution du volume plasmatique et d'hypoperfusion placentaire sans effet bénéfique sur l'évolution de la maladie.
L'hydrochlorothiazide ne doit pas être utilisé pour le traitement de l'hypertension artérielle essentielle chez la femme enceinte, sauf dans de rares situations où aucun autre traitement ne peut être utilisé.
L'heure du repas
Inhibiteurs de l'ECA :
Etant donné qu'aucune donnée n'est disponible concernant l'utilisation de lisinopril/hydrochlorothiazide pendant l'allaitement, le lisinopril/hydrochlorothiazide n'est pas recommandé et des traitements alternatifs avec un profil de sécurité prouvé pour une utilisation pendant l'allaitement sont préférés, notamment en cas d'allaitement du nouveau-né ou du prématuré.
Hydrochlorothiazide :
L'hydrochlorothiazide est excrété dans le lait maternel en petites quantités. Les diurétiques thiazidiques à forte dose provoquent une diurèse intense qui peut inhiber la production de lait. L'utilisation de Zestoretic pendant l'allaitement n'est pas recommandée. Si Zestoretic est pris pendant l'allaitement, les doses doivent être maintenues aussi faibles que possible.En raison des réactions graves possibles causées par l'hydrochlorothiazide chez les bébés allaités, une décision doit être prise s'il est plus approprié d'arrêter l'allaitement ou Zestoretic en tenant compte de la l'importance du médicament pour la mère.
Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Comme avec les autres antihypertenseurs, l'association lisinopril/hydrochlorothiazide peut avoir un effet léger à modéré sur l'aptitude à conduire des véhicules et à utiliser des machines.
Lors de la conduite de véhicules ou de machines, il faut tenir compte du fait que des étourdissements ou de la fatigue peuvent survenir.
Cependant, il est préférable de ne pas effectuer ces activités, qui nécessitent une attention particulière, jusqu'à ce que l'on sache comment le médicament est toléré.
Pour ceux qui pratiquent des activités sportives L'utilisation du médicament sans nécessité thérapeutique constitue un dopage et peut en tout état de cause déterminer des tests antidopage positifs.
Dose, méthode et moment d'administration Comment utiliser Zestoretic : Posologie
Vous devez suivre les instructions de votre médecin concernant la manière et la fréquence de prise des comprimés.
Prenez les comprimés avec un peu d'eau, à peu près à la même heure, de préférence tôt le matin.
L'amélioration de la santé ne doit pas conduire à l'arrêt du traitement, sauf demande du médecin
L'hypertension artérielle essentielle
La posologie habituelle est d'un comprimé administré une fois par jour. En général, si l'effet thérapeutique souhaité n'est pas atteint dans les 2 à 4 semaines, la posologie peut être augmentée à 2 comprimés administrés en une seule prise quotidienne.
Posologie en cas d'insuffisance rénale
Les diurétiques thiazidiques peuvent être inappropriés pour une utilisation chez les patients insuffisants rénaux et sont inefficaces à des valeurs de clairance de la créatinine de 30 ml/min ou moins (c'est-à-dire en présence d'une insuffisance rénale modérée ou sévère). Zestoretic ne doit pas être utilisé comme traitement initial. chez les patients insuffisants rénaux.
Chez les patients présentant une clairance de la créatinine > 30 et < 80 ml/min, Zestoretic ne doit être utilisé qu'après titrage des composants individuels.
Lorsqu'il est utilisé seul, la dose initiale recommandée de lisinopril dans l'insuffisance rénale légère est de 5 à 10 mg.
Traitement diurétique antérieur
Une hypotension symptomatique peut survenir après la dose initiale de Zestoretic; cela est plus susceptible de se produire chez les patients hypovolémiques et/ou déplétés en sodium à la suite d'un traitement diurétique antérieur. Le traitement diurétique doit être suspendu pendant 2 à 3 jours avant de commencer le traitement par Zestoretic. Si cela n'est pas possible, le traitement doit être instauré par le lisinopril seul à la dose de 5 mg.
Enfants
La sécurité et l'efficacité de Zestoretic chez les enfants n'ont pas été établies.
Utilisation chez les personnes âgées
L'efficacité et la tolérabilité du produit chez les personnes âgées ne diffèrent pas de celles des adultes et aucun ajustement posologique n'est donc nécessaire.
Surdosage Que faire si vous avez pris trop de Zestoretic
En cas d'ingestion/prise accidentelle d'une dose excessive de Zestoretic, contactez immédiatement votre médecin ou l'hôpital le plus proche
Que faire si vous avez oublié de prendre une ou plusieurs doses
Dans le cas où, en raison d'oublis, la prise d'une dose est omise, le traitement doit être poursuivi selon la fréquence prévue sans prendre de dose supplémentaire.
Effets secondaires Quels sont les effets secondaires de Zestoretic
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Zestoretic est généralement bien toléré.
Dans les études cliniques, les effets indésirables étaient généralement légers et transitoires ; dans la plupart des cas, il n'était pas nécessaire d'interrompre le traitement. Les effets indésirables observés étaient limités à ceux précédemment rapportés avec le lisinopril ou l'hydrochlorothiazide.
Les effets indésirables (EIM) les plus fréquemment rapportés sont les étourdissements, qui ont généralement répondu à la réduction de la dose et ont rarement nécessité l'arrêt du traitement, les maux de tête, la toux et l'hypotension, y compris l'hypotension orthostatique qui peut survenir chez 1 à 10, % des patients traités.
Encore moins fréquents étaient : diarrhée, nausées, vomissements, sécheresse de la bouche, éruption cutanée, goutte, palpitations, gêne thoracique, crampes et faiblesse musculaires, paresthésie, asthénie, impuissance, insuffisance rénale aiguë et syncope
Les effets indésirables suivants ont été observés et rapportés au cours du traitement par lisinopril/hydrochlorothiazide avec les fréquences suivantes : très fréquent (> 10 %), fréquent (> 1 %, 0,1 %, 0,01 %,
Troubles du système sanguin et lymphatique
Rares : anémie.
Très rare : aplasie médullaire, thrombocytopénie, leucopénie, agranulocytose, anémie hémolytique.
Pathologies endocriniennes
Rare : sécrétion inappropriée d'hormone antidiurétique.
Troubles du métabolisme et de la nutrition
Peu fréquent : goutte.
Rare : hyperglycémie, hypokaliémie, hyperuricémie, hyperkaliémie.
Troubles du système nerveux et troubles psychiatriques
Fréquent : vertiges, maux de tête, paresthésie.
Rare : troubles olfactifs.
Peu fréquent : symptômes dépressifs.
Pathologies cardiaques et vasculaires
Fréquent : effets orthostatiques (y compris hypotension), syncope.
Peu fréquent : palpitations.
Troubles respiratoires, thoraciques et médiastinaux
Fréquent : toux.
Problèmes gastro-intestinaux
Fréquent : diarrhée, nausées, vomissements.
Peu fréquent : bouche sèche.
Rare : pancréatite.
Très rare : œdème de Quincke intestinal.
Troubles hépatobiliaires
Très rare : hépatite à la fois hépatocellulaire et cholestatique, ictère, insuffisance hépatique. Des cas d'hépatite ont très rarement évolué vers une insuffisance hépatique chez certains patients. Les patients recevant Zestoretic qui présentent un ictère ou une élévation marquée des enzymes hépatiques doivent interrompre le traitement par Zestoretic et recevoir une surveillance médicale appropriée.
Affections de la peau et du tissu sous-cutané
Fréquent : éruption cutanée.
Peu fréquent : hypersensibilité/œdème de Quincke : œdème de Quincke du visage, des extrémités, des lèvres, de la langue, de la glotte et/ou du larynx (voir rubrique « Précautions d'emploi »).
Très rare : pseudolymphome cutané. Des symptômes complexes ont été rapportés, pouvant inclure un ou plusieurs des éléments suivants : fièvre, vascularite, myalgie, arthralgie/arthrite, positivité des anticorps antinucléaires (ANA), augmentation de la vitesse de sédimentation des érythrocytes (VS), éosinophilie et leucocytose, éruption cutanée, photosensibilité et autres symptômes dermatologiques. manifestation.
Troubles musculo-squelettiques et du tissu conjonctif
Fréquent : crampes musculaires.
Rare : faiblesse musculaire.
Maladies de l'appareil reproducteur et du sein
Fréquent : impuissance.
Troubles généraux et anomalies au site d'administration
Fréquent : fatigue, asthénie.
Peu fréquent : oppression thoracique.
Tests de laboratoire
Fréquent : augmentation de l'urée sanguine, augmentation de la créatinine sérique, augmentation des enzymes hépatiques, diminution de l'hémoglobine
Peu fréquent : diminution de l'hématocrite.
Rare : augmentation de la bilirubine sérique.
D'autres effets secondaires qui ont été rapportés avec les composants individuels et qui peuvent être des effets secondaires potentiels de Zestoretic sont :
Hydrochlorothiazide (fréquences inconnues) :
Autres effets secondaires : purpura, éruption cutanée.
Dans de très rares cas, un syndrome de Stevens-Johnson peut survenir.
Dans des cas isolés : hypercalcémie, cette dernière rendant nécessaire des tests diagnostiques pour mettre en évidence une éventuelle hyperparathyroïdie Des troubles du rythme cardiaque, une myopie aiguë et un glaucome aigu à angle fermé sont possibles.
Des troubles visuels transitoires, des douleurs oculaires sévères accompagnées de rougeurs sont possibles. Les patients souffrant de douleur aiguë et d'yeux rouges doivent consulter immédiatement un médecin ; si elle n'est pas traitée, cette condition peut entraîner une perte de vision permanente
Lisinopril
* Très rarement, il a été rapporté que chez certains patients le développement indésirable d'une hépatite a évolué vers une insuffisance hépatique. Les patients recevant du lisinopril/hydrochlorothiazide et qui développent un ictère ou une élévation marquée des enzymes hépatiques doivent arrêter le lisinopril/hydrochlorothiazide et recevoir un traitement médical approprié.
** Un complexe de symptômes a été rapporté qui peut inclure un ou plusieurs des éléments suivants : fièvre, vascularite, myalgie, arthralgie/arthrite, positivité des anticorps antinucléaires (ANA), vitesse de sédimentation élevée des globules rouges (VS), éosinophilie et leucocytose, éruption cutanée , photosensibilité ou autres manifestations dermatologiques.
Comme avec les autres inhibiteurs de l'ECA, des hallucinations visuelles et/ou auditives ont été rapportées.
Le respect des instructions contenues dans la notice réduit le risque d'effets indésirables.
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice. Les effets indésirables peuvent également être signalés directement via le système national de notification à l'adresse : https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse
En signalant les effets secondaires, vous pouvez contribuer à fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
Expiration : voir la date d'expiration imprimée sur l'emballage.
La date de péremption indiquée est destinée au produit dans un emballage intact, correctement conservé.
Avertissement : n'utilisez pas le médicament après la date de péremption indiquée sur la plaquette et la boîte.
Précautions particulières de conservation
Les comprimés doivent être conservés dans leur emballage pour les conserver à l'abri de la lumière.
Les médicaments ne doivent pas être jetés au tout à l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Gardez ce médicament hors de la portée et de la vue des enfants.
COMPOSITION
Chaque comprimé contient : principes actifs : 21,8 mg de lisinopril dihydraté (équivalent à 20 mg de lisinopril anhydre), 12,5 mg d'hydrochlorothiazide.
Excipients : mannitol, phosphate de calcium dibasique dihydraté, amidon de maïs, amidon prégélatinisé, stéarate de magnésium.
FORME PHARMACEUTIQUE ET CONTENU
Comprimés.
Paquet de 14 comprimés.
Paquet de 28 comprimés.
Paquet de 42 comprimés.
Toutes les présentations peuvent ne pas être commercialisées.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
COMPRIMÉS ZESTORETIC 20 MG + 12,5 MG
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Chaque tablette contient:
principes actifs : lisinopril dihydraté 21,8 mg (équivalent à 20 mg de lisinopril anhydre) + 12,5 mg d'hydrochlorothiazide.
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Comprimés.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Zestoretic est indiqué pour le traitement de l'hypertension artérielle primaire chez les patients pour lesquels un traitement combiné est approprié.
04.2 Posologie et mode d'administration
Hypertension artérielle primaire
La posologie habituelle est d'un comprimé administré une fois par jour. Comme avec les autres médicaments administrés une fois par jour, Zestoretic doit être pris à peu près à la même heure.
En général, si l'effet thérapeutique souhaité n'est pas atteint dans les 2 à 4 semaines, la posologie peut être augmentée à 2 comprimés administrés en une seule prise quotidienne.
Posologie en cas d'insuffisance rénale
Les diurétiques thiazidiques peuvent être inappropriés pour une utilisation chez les patients insuffisants rénaux et sont inefficaces à des valeurs de clairance de la créatinine de 30 ml/min ou moins (c'est-à-dire en présence d'une insuffisance rénale modérée ou sévère). Zestoretic ne doit pas être utilisé comme traitement initial. chez les patients insuffisants rénaux.
Chez les patients présentant une clairance de la créatinine > 30 et une titration des composants individuels.
Lorsqu'il est utilisé seul, la dose initiale recommandée de lisinopril dans l'insuffisance rénale légère est de 5 à 10 mg.
Traitement diurétique antérieur
Une hypotension symptomatique peut survenir après la dose initiale de Zestoretic; cela est plus susceptible de se produire chez les patients hypovolémiques et/ou déplétés en sodium à la suite d'un traitement diurétique antérieur. Le traitement diurétique doit être suspendu pendant 2 à 3 jours avant de commencer le traitement par Zestoretic. Si cela n'est pas possible, le traitement doit être instauré par le lisinopril seul, à la dose de 5 mg.
Population pédiatrique
La sécurité et l'efficacité de Zestoretic chez les enfants n'ont pas été établies.
Utilisation chez les personnes âgées
Dans les études cliniques, l'efficacité et la tolérabilité du lisinopril et de l'hydrochlorothiazide administrés ensemble étaient similaires chez les patients hypertendus âgés et plus jeunes.
Le lisinopril, dans une fourchette posologique quotidienne de 20 à 80 mg, était aussi efficace chez les patients hypertendus âgés (65 ans ou plus) que chez les patients hypertendus non âgés. Chez les patients hypertendus âgés, le lisinopril en monothérapie a été aussi efficace pour réduire la pression artérielle diastolique que l'hydrochlorothiazide ou l'aténolol.
Dans les études cliniques, l'âge n'a pas affecté la tolérance du lisinopril.
04.3 Contre-indications
Zestoretic est contre-indiqué
• chez les patients hypersensibles au lisinopril, à l'un des excipients mentionnés à la rubrique 6.1, ou à d'autres inhibiteurs de l'enzyme de conversion de l'angiotensine (ECA).
• chez les patients hypersensibles à l'hydrochlorothiazide ou à d'autres médicaments dérivés des sulfamides.
• chez les patients souffrant d'anurie.
• chez les patients ayant des antécédents d'œdème de Quincke liés à un traitement antérieur par des inhibiteurs de l'enzyme de conversion de l'angiotensine.
• chez les patients présentant un angio-œdème héréditaire ou idiopathique.
• au cours des deuxième et troisième trimestres de la grossesse (voir rubriques 4.4 et 4.6).
• chez les patients atteints d'insuffisance rénale sévère (clairance de la créatinine
• chez les patients présentant une insuffisance hépatique sévère.
• l'utilisation concomitante de Zestoretic avec des produits contenant de l'aliskiren est contre-indiquée chez les patients atteints de diabète sucré ou d'insuffisance rénale (GFR 2) (voir rubriques 4.5 et 5.1).
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Hypotension symptomatique
Une hypotension symptomatique a rarement été observée chez les patients souffrant d'hypertension non compliquée, mais elle est plus susceptible de se produire chez les patients qui ont présenté une diminution du volume sanguin ou une hypomagnésémie en raison d'un traitement diurétique antérieur, d'une restriction alimentaire en sel, d'une dialyse, de diarrhées ou de vomissements, ou avec une rénine sévère. hypertension dépendante (voir rubriques 4.5 et 4.8). Chez ces patients, des contrôles réguliers des électrolytes sériques doivent être effectués à des intervalles appropriés.
Chez les patients présentant un risque accru d'hypotension symptomatique, l'instauration du traitement et l'ajustement posologique doivent être étroitement surveillés.
Une attention particulière doit être accordée aux patients atteints de maladie cardiaque ou de cérébropathie ischémique, car une chute excessive de la pression artérielle pourrait provoquer un infarctus du myocarde ou un événement vasculaire cérébral.
En cas d'hypotension, le patient doit être placé en décubitus dorsal et, si nécessaire, perfusé avec une solution saline par voie intraveineuse. Une réponse hypotensive transitoire n'est pas une contre-indication à d'autres doses du médicament. En rétablissant un volume sanguin et une pression artérielle efficaces, le traitement peut être rétabli à une dose réduite ; sinon il est possible d'utiliser l'un ou l'autre membre de l'association individuellement.
Chez certains patients présentant une insuffisance cardiaque et une pression artérielle normale ou basse, une baisse supplémentaire de la pression artérielle peut survenir avec le lisinopril. Cet effet est attendu et ne constitue généralement pas un motif d'arrêt du traitement. Si l'hypotension devient symptomatique, une réduction de la posologie ou l'arrêt de Zestoretic peut être nécessaire.
Sténose de la valve aortique ou mitrale / cardiomyopathie hypertrophique
Comme avec les autres inhibiteurs de l'ECA, Zestoretic doit être administré avec prudence aux patients présentant une sténose de la valve mitrale et une obstruction de l'éjection ventriculaire gauche telle qu'une sténose aortique ou une cardiomyopathie hypertrophique.
Double blocage du système rénine-angiotensine-aldostérone (RAAS)
Il existe des preuves que l'utilisation concomitante d'inhibiteurs de l'ECA, d'inhibiteurs des récepteurs de l'angiotensine II ou d'aliskirène augmente le risque d'hypotension, d'hyperkaliémie et de diminution de la fonction rénale (y compris l'insuffisance rénale aiguë). les antagonistes ou l'aliskiren ne sont donc pas recommandés (voir rubriques 4.3 et 4.5).
Si un traitement en double bloc est considéré comme absolument nécessaire, cela ne doit être fait que sous la supervision d'un spécialiste et avec une surveillance étroite et fréquente de la fonction rénale, des électrolytes et de la pression artérielle.
Les inhibiteurs de l'ECA et les antagonistes des récepteurs de l'angiotensine II ne doivent pas être utilisés simultanément chez les patients atteints de néphropathie diabétique.
Chirurgie / anesthésie
Chez les patients subissant une intervention chirurgicale majeure ou pendant une anesthésie avec des agents provoquant une hypotension, le lisinopril peut bloquer la formation d'angiotensine II secondaire à la libération compensatrice de rénine. Si une hypotension attribuable à ce mécanisme survient, celle-ci peut être corrigée par une expansion volémique.
Fonction rénale altérée
Les diurétiques thiazidiques peuvent ne pas être les diurétiques appropriés dans le traitement des patients insuffisants rénaux et sont inefficaces à des valeurs de clairance de la créatinine inférieures ou égales à 30 ml/min (c'est-à-dire en présence d'une insuffisance rénale modérée ou sévère).
Zestoretic ne doit pas être administré aux patients présentant une insuffisance rénale (clairance de la créatinine ≤ 80 ml/min) jusqu'à ce que la titration des composants individuels ait d'abord démontré la nécessité des dosages présents dans le comprimé combiné.
Chez les patients insuffisants cardiaques, une hypotension après l'instauration d'un traitement par inhibiteur de l'ECA peut entraîner une altération supplémentaire de la fonction rénale.Une insuffisance rénale aiguë, généralement réversible, a été rapportée dans cette situation.
Chez certains patients présentant une sténose bilatérale de l'artère rénale ou une sténose monorénale de l'artère rénale, des augmentations de l'azote uréique du sang et de la créatinine habituellement réversibles après un traitement par des inhibiteurs de l'enzyme de conversion de l'angiotensine (ECA) ont été observées. Si une hypertension rénovasculaire est également présente, il existe un risque accru d'hypotension sévère et d'insuffisance rénale. Chez ces patients, le traitement doit être instauré sous surveillance médicale étroite à de faibles doses et après un ajustement posologique adéquat. Étant donné qu'un traitement diurétique peut contribuer à ce qui précède, la fonction rénale doit être surveillée pendant les premières semaines de traitement par Zestoretic.
Certains patients hypertendus sans maladie rénale vasculaire apparente ont développé des augmentations généralement légères et transitoires de l'azote uréique du sang et de la créatinine sanguine lorsque le lisinopril était administré en concomitance avec un diurétique.
Ceci est plus susceptible de se produire chez les patients présentant une insuffisance rénale préexistante. Il peut être nécessaire de réduire la posologie et/ou d'arrêter le diurétique et/ou le lisinopril.
Chez les patients atteints d'insuffisance cardiaque sévère dont la fonction rénale peut être dépendante du système rénine-angiotensine-aldostérone, le traitement par IEC peut être associé à une oligurie et/ou une azotémie progressive et, rarement, à une insuffisance rénale aiguë et/ou à la mort. Chez ces patients, le traitement par IEC doit être instauré avec une prudence particulière.
Hépatopathie
Les thiazidiques doivent être utilisés avec prudence chez les patients présentant une insuffisance hépatique ou une maladie hépatique évolutive, car des modifications minimes de l'équilibre hydrique et électrolytique peuvent précipiter un coma hépatique (voir rubrique 4.3). Rarement, les inhibiteurs de l'ECA ont été associés à un syndrome qui commence par une cholestatique. ictère ou hépatite et évolue vers une nécrose fulminante et (parfois) la mort. Le mécanisme de ce syndrome est inconnu. Les patients traités par lisinopril / hydrochlorothiazide qui développent une jaunisse ou une élévation marquée des enzymes hépatiques doivent arrêter le traitement par lisinopril / hydrochlorothiazide et subir un suivi médical approprié. en haut.
Hypersensibilité / œdème de Quincke
Un œdème de Quincke du visage, des extrémités, des lèvres, de la langue, de la glotte et/ou du larynx a été rapporté peu fréquemment chez des patients traités par des inhibiteurs de l'enzyme de conversion de l'angiotensine, y compris le lisinopril. Cela peut arriver à tout moment pendant le traitement. Dans de tels cas, l'administration de lisinopril doit être interrompue rapidement et une surveillance appropriée doit être instituée pour assurer une rémission complète des symptômes avant la sortie du patient. Même dans les cas où le gonflement n'affecte que la langue sans détresse respiratoire, les patients doivent être surveillés pendant une période prolongée car le traitement par antihistaminiques et corticostéroïdes peut ne pas être suffisant.
Des événements mortels dus à un œdème de Quincke associé à un œdème du larynx ou de la langue ont été très rarement rapportés. Une obstruction des voies respiratoires peut survenir chez les patients présentant une atteinte de la langue, de la glotte ou du larynx, en particulier chez les personnes ayant des antécédents de chirurgie des voies respiratoires. Dans ces cas, un traitement d'urgence doit être administré rapidement. Cela peut inclure l'administration d'épinéphrine et/ou des mesures pour maintenir les voies respiratoires ouvertes. Le patient doit être maintenu sous surveillance médicale étroite jusqu'à la résolution complète et persistante des symptômes. .
Les inhibiteurs de l'enzyme de conversion (inhibiteurs de l'ECA) provoquent un angio-œdème plus fréquemment chez les patients noirs que chez les patients non noirs.
Les patients ayant des antécédents d'œdème de Quincke non lié au traitement par inhibiteur de l'ECA peuvent présenter un risque accru d'œdème de Quincke lorsqu'ils prennent un inhibiteur de l'ECA (voir également rubrique 4.3).
Chez les patients prenant des thiazidiques, des réactions d'hypersensibilité peuvent survenir avec ou sans antécédents d'épisodes allergiques ou d'asthme bronchique. Une exacerbation ou une activation du lupus érythémateux disséminé a été rapportée avec l'utilisation de thiazidiques.
Effets métaboliques et endocriniens
Chez les patients diabétiques traités par antidiabétiques oraux ou par insuline, la glycémie doit être étroitement surveillée pendant le premier mois de traitement par un IEC.
La thérapie thiazidique peut altérer la tolérance au glucose; par conséquent, un ajustement de la posologie des agents antidiabétiques, y compris l'insuline, peut être nécessaire.
Des augmentations des taux de cholestérol et de triglycérides ont été associées à un traitement diurétique par thiazidiques.
Chez certains patients, le traitement par thiazidiques peut précipiter une hyperuricémie et/ou une goutte, mais le lisinopril peut induire une augmentation de l'acide urique dans les urines et par conséquent atténuer l'effet hyperuricémique de l'hydrochlorothiazide.
Désensibilisation
Les patients ayant reçu des inhibiteurs de l'ECA au cours d'un traitement désensibilisant (par exemple du venin d'hyménoptère) ont présenté des réactions anaphylactoïdes. Chez les mêmes patients, ces réactions ont été évitées lorsque l'inhibiteur de l'ECA a été temporairement retiré, mais elles sont réapparues après que le médicament a été ré-administré par inadvertance.
Réactions anaphylactoïdes chez les patients hémodialysés
L'utilisation de lisinopril-hydrochlorothiazide n'est pas indiquée chez les patients nécessitant une dialyse pour insuffisance rénale.
Des réactions anaphylactotides ont été rapportées chez des patients subissant certaines procédures d'hémodialyse (par exemple avec des membranes AN 69 à haut flux et pendant l'aphérèse des lipoprotéines de basse densité (LDL) réalisée avec des colonnes de sulfate de dextran) traités de manière concomitante avec des inhibiteurs de l'ECA. L'utilisation de différents types de membranes de dialyse ou de différents types d'agents antihypertenseurs doit être envisagée pour ces patients.
Réactions anaphylactoïdes au cours de l'aphérèse des lipoprotéines de basse densité (LDL)
En de rares occasions, des patients prenant des inhibiteurs de l'ECA au cours d'une aphérèse des lipoprotéines de basse densité (LDL) avec du sulfate de dextran ont développé des réactions anaphylactoïdes potentiellement mortelles.Ces symptômes pourraient être évités en suspendant temporairement le traitement par inhibiteurs de l'ECA avant chaque séance d'aphérèse.
Ethnicité
Les inhibiteurs de l'enzyme de conversion (inhibiteurs de l'ECA) provoquent un angio-œdème plus fréquemment chez les patients noirs que chez les patients non noirs.
Comme avec d'autres inhibiteurs de l'ECA, le lisinopril peut être moins efficace pour abaisser la tension artérielle chez les patients noirs que chez les patients non noirs, probablement en raison d'une prévalence plus élevée de faibles concentrations de rénine dans la population hypertendue noire.
La toux
Des cas de toux ont été rapportés lors de l'utilisation d'inhibiteurs de l'ECA. Celle-ci est généralement non productive, persistante et disparaît à l'arrêt du traitement. La toux induite par les IEC doit être prise en compte dans le diagnostic différentiel de la toux.
Neutropénie et agranulocytose
Une neutropénie/agranulocytose, une thrombocytopénie et une anémie ont été rapportées chez des patients recevant des inhibiteurs de l'ECA. Chez les patients ayant une fonction rénale normale et en l'absence d'autres facteurs de complication, une neutropénie survient rarement. La neutropénie et l'agranulocytose disparaissent après l'arrêt du traitement par IEC.
Le lisinopril doit être administré avec une extrême prudence aux patients présentant une maladie du collagène, ceux sous traitement immunosuppresseur, allopurinol ou procaïnamide, ou qui présentent une combinaison de ces facteurs de complication, en particulier en cas d'insuffisance rénale antérieure. Certains de ces patients ont développé des infections sévères qui, dans quelques cas, n'ont pas répondu à une antibiothérapie intensive. Si ces patients sont traités par le lisinopril, une surveillance périodique de la numération des globules blancs est recommandée et les patients doivent être informés de la nécessité de signaler tout épisode d'infection.
Grossesse
Le traitement par inhibiteur de l'ECA ne doit pas être instauré pendant la grossesse.
Pour les patientes qui envisagent une grossesse, des traitements antihypertenseurs alternatifs avec un profil d'innocuité prouvé pour une utilisation pendant la grossesse doivent être utilisés à moins que la poursuite du traitement par inhibiteur de l'ECA ne soit considérée comme essentielle. Lorsqu'une grossesse est diagnostiquée, le traitement par les inhibiteurs de l'ECA doit être arrêté immédiatement et, le cas échéant, un traitement alternatif doit être instauré (voir rubriques 4.3 et 4.6).
Traitement diurétique antérieur
Le traitement diurétique doit être suspendu pendant 2-3 jours avant de commencer le traitement par lisinopril/hydrochlorothiazide. Si cela n'est pas possible, le traitement doit être instauré par le lisinopril seul, à la dose de 5 mg.
Patients transplantés rénaux
En l'absence d'expérience avec des patients ayant récemment subi une greffe de rein, cette association ne doit pas être utilisée.
Un déséquilibre électrolytique
Comme pour tout patient sous traitement diurétique, une détermination périodique des électrolytes sériques doit être effectuée à des intervalles appropriés.
Les thiazidiques, y compris l'hydrochlorothiazide, peuvent provoquer un déséquilibre hydrique et électrolytique (hypokaliémie, hyponatrémie et alcalose hypochlorémique).Les signes indicateurs d'un déséquilibre hydrique ou électrolytique sont bouche sèche, soif, faiblesse, léthargie, somnolence, crampes ou douleurs musculaires, fatigue musculaire, hypotension, oligurie , tachycardie et troubles gastro-intestinaux tels que nausées et vomissements.
Une hyponatrémie de dilution peut survenir chez les patients œdémateux par temps chaud. Une carence en chlorure est généralement bénigne et ne nécessite pas de traitement. Il a été démontré que les thiazidiques augmentent l'excrétion urinaire de magnésium, ce qui peut entraîner une hypomagnésémie.
Les thiazidiques peuvent diminuer l'excrétion urinaire du calcium et provoquer des augmentations légères et intermittentes du calcium. Une hypercalcémie marquée peut révéler une hyperparathyroïdie asymptomatique. Le traitement thiazidique doit être interrompu avant que des tests de la fonction parathyroïdienne ne soient effectués.
Hyperkaliémie
Des élévations de la kaliémie ont été observées chez certains patients traités par des inhibiteurs de l'ECA, y compris le lisinopril. Les patients à risque de développer une hyperkaliémie comprennent les patients atteints d'insuffisance rénale, de diabète sucré ou ceux recevant un traitement concomitant par des diurétiques d'épargne potassique, des suppléments potassiques ou des substituts de sel contenant du potassium, ou les patients sous d'autres médicaments avec lesquels une augmentation de la kaliémie (par ex. héparine). Si l'utilisation concomitante des agents ci-dessus est jugée nécessaire, une surveillance régulière de la kaliémie est recommandée (voir rubrique 4.5).
Patients diabétiques
Chez les patients diabétiques traités par des antidiabétiques oraux ou de l'insuline, le contrôle glycémique doit être étroitement surveillé pendant le premier mois de traitement par un inhibiteur de l'ECA (voir rubrique 4.5).
Lithium
L'association d'inhibiteurs de l'ECA, de lithium et de lisinopril n'est généralement pas recommandée (voir rubrique 4.5).
Test de dopage
L'hydrochlorothiazide contenu dans ce médicament peut donner un résultat positif au test de dopage.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Double blocage du système rénine-angiotensine-aldostérone
Les données des essais cliniques ont montré que le double blocage du système rénine-angiotensine-aldostérone (SRAA) par l'utilisation combinée d'inhibiteurs de l'ECA, d'inhibiteurs des récepteurs de l'angiotensine II ou d'aliskiren est associé à une fréquence plus élevée d'événements indésirables tels qu'hypotension, hyperkaliémie et diminution de la fonction rénale. (y compris l'insuffisance rénale aiguë) par rapport à l'utilisation d'un seul agent actif sur le système RAAS (voir rubriques 4.3, 4.4 et 5.1) Surveiller étroitement la pression artérielle, la fonction rénale et les électrolytes chez les patients traités par le lisinopril et d'autres agents affectant le RAAS Ne pas administrer l'aliskiren avec le lisinopril chez les patients diabétiques Eviter l'utilisation de l'aliskiren avec le lisinopril chez les patients présentant une insuffisance rénale (DFG 2) (voir rubrique 4.3).
Autres antihypertenseurs
L'administration concomitante de ces médicaments peut augmenter l'effet hypotenseur du lisinopril/hydrochlorothiazide.L'administration concomitante de nitroglycérine et d'autres nitrates ou d'autres vasodilatateurs peut réduire davantage la pression artérielle.
L'association du lisinopril avec des médicaments contenant de l'aliskirène doit être évitée (voir rubriques 4.3 et 4.4).
Suppléments de potassium, diurétiques d'épargne potassique ou substituts du sel contenant du potassium et d'autres médicaments pouvant augmenter les taux sériques de potassium
La déplétion potassique induite par les diurétiques thiazidiques est généralement atténuée par l'effet d'épargne potassique du lisinopril.
L'utilisation de suppléments potassiques, d'agents d'épargne potassique ou de substituts du sel contenant du potassium et d'autres médicaments susceptibles d'augmenter les taux sériques de potassium (p. Si l'utilisation concomitante de lisinopril/hydrochlorothiazide et de l'un de ces agents est nécessaire, ils doivent être utilisés avec prudence et avec une surveillance fréquente de la kaliémie (voir rubrique 4.4).
Lithium
Des augmentations réversibles des concentrations sériques de lithium et de la toxicité ont été rapportées lors de l'administration concomitante de lithium et d'inhibiteurs de l'ECA.
Les agents diurétiques et les inhibiteurs de l'ECA réduisent la clairance rénale du lithium, entraînant un risque élevé de sa toxicité.
Par conséquent, l'utilisation de lisinopril/hydrochlorothiazide en association avec le lithium n'est pas recommandée, mais, si l'association est nécessaire, une surveillance attentive des taux sériques de lithium doit être effectuée (voir rubrique 4.4). Avant d'utiliser des produits contenant du lithium, consultez le Résumé des Caractéristiques du Produit correspondant.
Or
Des réactions nitritoïdes (symptômes de vasodilatation incluant bouffées vasomotrices, nausées, vertiges et hypotension, qui peuvent être très sévères) ont été rapportées plus fréquemment chez les patients traités par des inhibiteurs de l'ECA après l'administration d'or injectable (par exemple, aurothiomalate de sodium).
Autres thérapies concomitantes
L'indométacine peut diminuer l'efficacité antihypertensive du lisinopril et de l'hydrochlorothiazide administrés simultanément.
Un traitement concomitant avec des activateurs tissulaires du plasminogène peut augmenter le risque de développer un œdème de Quincke.
Anti-inflammatoires non stéroïdiens (AINS) dont l'acide acétylsalicylique
L'administration chronique d'AINS, y compris d'inhibiteurs sélectifs de la cyclooxygénase-2, peut réduire l'effet antihypertenseur d'un inhibiteur de l'ECA.
Les inhibiteurs de l'ECA et les AINS peuvent exercer un effet additif sur l'aggravation de la fonction rénale. Ces effets sont généralement réversibles.
Dans de rares cas, une insuffisance rénale aiguë peut survenir, en particulier chez les patients insuffisants rénaux tels que les personnes âgées ou les sujets déshydratés. L'association doit être administrée avec prudence, en particulier chez les personnes âgées. Les patients doivent être suffisamment hydratés et une surveillance de la pression artérielle doit être envisagée. fonction au début du traitement concomitant.
Myorelaxants non dépolarisants (par exemple, chlorure de tubocurarine)
L'effet de ces médicaments peut être intensifié par l'hydrochlorothiazide.
Allopurinol
L'administration concomitante d'inhibiteurs de l'ECA et d'allopurinol augmente le risque d'atteinte rénale et peut entraîner un risque accru de leucopénie.
Agents cytostatiques et immunosuppresseurs, procaïnamide, cyclophosphamide, méthotrexate
L'administration concomitante d'inhibiteurs de l'ECA peut augmenter le risque de leucopénie. Les thiazidiques peuvent, en réduisant l'excrétion rénale des médicaments cytotoxiques, potentialiser leur effet myélosuppresseur (voir rubrique 4.4).
Médicaments provoquant des torsades de pointes
En raison du risque d'hypokaliémie, l'administration d'hydrochlorothiazide et de médicaments induisant des torsades de pointes, par exemple certains antiarythmiques, certains antipsychotiques et autres médicaments connus pour induire des torsades de pointes, doit être utilisée avec prudence.
Antidépresseurs tricycliques / antipsychotiques / anesthésiques
L'administration concomitante de certains anesthésiques, antidépresseurs tricycliques et antipsychotiques avec des inhibiteurs de l'ECA peut entraîner une « baisse supplémentaire de la pression artérielle (voir rubrique 4.4).
Sympathomimétiques
Les sympathomimétiques peuvent réduire les effets antihypertenseurs des inhibiteurs de l'ECA.
Interactions médicamenteuses potentielles
Autres antihypertenseurs : des effets additifs peuvent survenir.
Lorsqu'ils sont administrés ensemble, les médicaments suivants peuvent interagir avec les diurétiques thiazidiques : Alcool - Barbituriques - Narcotiques : Une potentialisation de la chute de pression en position debout peut se produire.
Médicaments antidiabétiques
Le traitement par un diurétique thiazidique peut altérer la tolérance au glucose Des études épidémiologiques ont indiqué que l'administration concomitante d'inhibiteurs de l'ECA et d'antidiabétiques (hypoglycémiants oraux et insuline) peut entraîner une augmentation de l'effet hypoglycémiant avec risque d'hypoglycémie. chez les patients diabétiques, un ajustement de la posologie des antidiabétiques peut être nécessaire et le besoin d'autres antidiabétiques, y compris l'insuline, peut rester inchangé ou être augmenté ou diminué.
Autres diurétiques kaliurétiques, amphotéricine B (parentérale), carbénoxolone, corticoïdes, corticotropine (ACTH) ou laxatifs stimulants, dérivés de l'acide salicylique
L'effet de déplétion potassique de l'hydrochlorothiazide peut être intensifié par des médicaments provoquant une perte de potassium et une hypokaliémie. Une hypokaliémie peut se développer lors de l'utilisation concomitante de stéroïdes ou d'adrénocorticotropes (ACTH).
Sels de calcium (suppléments de calcium ou de vitamine D)
Lorsqu'ils sont administrés en concomitance avec des diurétiques thiazidiques, ils peuvent provoquer une augmentation des taux de calcium sérique entraînant une diminution de l'excrétion.
Si des suppléments de calcium ou de vitamine D sont nécessaires, les taux de calcium sérique doivent être surveillés et leur dose ajustée en conséquence.
Glycosides cardiaques
Il existe un risque accru d'intoxication digitalique associée à l'hypokaliémie induite par les thiazidiques.L'hypokaliémie peut sensibiliser ou augmenter la réponse du cœur aux effets toxiques de la digitaline (par exemple, augmentation de l'irritabilité ventriculaire).
Cholestyramine et colestipol
Ils peuvent réduire ou ralentir l'absorption de l'hydrochlorothiazide. Par conséquent, les diurétiques sulfamides doivent être pris au moins une heure avant ou 4 à 6 heures après la prise de ces médicaments.
Triméthoprime
L'administration concomitante d'inhibiteurs de l'ECA et de thiazidiques avec le triméthoprime augmente le risque d'hyperkaliémie.
Sotalol
L'hypokaliémie induite par les thiazidiques peut augmenter le risque d'arythmies induites par le sotalol.
Ciclosporine
L'administration concomitante d'inhibiteurs de l'ECA et de cyclosporine augmente le risque d'atteinte rénale, d'hyperkaliémie, d'hyperuricémie et de complications de type goutte.
Amines pressives (par exemple adrénaline)
Les thiazidiques peuvent diminuer la réponse artérielle à la noradrénaline, mais pas au point d'empêcher son utilisation en thérapie comme agent de pression.
Diazoxide
Les thiazidiques peuvent augmenter l'effet hyperglycémiant du diazoxide.
Amantadina
Les thiazidiques peuvent augmenter le risque d'effets indésirables causés par l'amantadine.
04.6 Grossesse et allaitement
Grossesse
Inhibiteurs de l'ECA :
L'utilisation d'inhibiteurs de l'ECA n'est pas recommandée pendant le premier trimestre de la grossesse (voir rubrique 4.4) L'utilisation d'inhibiteurs de l'ECA est contre-indiquée pendant les deuxième et troisième trimestres de la grossesse (voir rubriques 4.3 et 4.4).
Les preuves épidémiologiques sur le risque de tératogénicité suite à une exposition aux inhibiteurs de l'ECA au cours du premier trimestre de la grossesse n'ont pas été concluantes ; cependant, une légère augmentation du risque ne peut être exclue, en particulier pour les systèmes cardiovasculaire et nerveux central. Si le lisinopril est utilisé pendant le premier trimestre de la grossesse, les patientes doivent être informées des risques potentiels pour le fœtus.
Pour les patientes qui envisagent une grossesse, des traitements antihypertenseurs alternatifs avec un profil d'innocuité prouvé pour une utilisation pendant la grossesse doivent être utilisés à moins que la poursuite du traitement par inhibiteur de l'ECA ne soit considérée comme essentielle.
Lorsqu'une grossesse est diagnostiquée, le traitement par inhibiteurs de l'ECA doit être arrêté immédiatement et, le cas échéant, un traitement alternatif doit être instauré.
L'exposition aux IEC au cours des deuxième et troisième trimestres est connue pour induire une toxicité fœtale (diminution de la fonction rénale, oligohydramnios, retard de l'ossification du crâne) et néonatale (insuffisance rénale, hypotension, hyperkaliémie) chez la femme (voir également paragraphe 5.3).
Une oligodramniose maternelle s'est produite, représentant vraisemblablement une diminution de la fonction rénale du fœtus et pouvant entraîner une contracture des membres, des déformations craniofaciales et un développement pulmonaire hypoplasique.
En cas d'exposition à un inhibiteur de l'ECA à partir du deuxième trimestre de la grossesse, un contrôle échographique de la fonction rénale et du crâne est recommandé.
Les nouveau-nés dont les mères ont pris des inhibiteurs de l'ECA doivent être étroitement surveillés afin de détecter toute hypotension (voir rubriques 4.3 et 4.4).
Dans les rares cas où l'utilisation pendant la grossesse est considérée comme essentielle, des échographies en série doivent être effectuées pour vérifier les conditions intra-amniotiques si l'exposition à Zestoretic s'est produite au cours du deuxième ou du troisième trimestre de la grossesse. En cas de découverte d'une oligodramniose, le lisinopril doit être interrompu à moins qu'il ne soit considéré comme salvateur pour la mère.
Les médecins et les patients doivent être conscients, cependant, que l'oligodramniose ne peut également être évidente qu'après que des dommages irréversibles au fœtus ont été établis.
Les nourrissons dont les mères ont pris du lisinopril doivent également être étroitement surveillés pour l'oligurie et l'hyperkaliémie.
Le lisinopril, qui traverse le placenta, a été retiré de la circulation néonatale par dialyse intrapéritonéale avec un certain bénéfice clinique et peut théoriquement être retiré par plasmaphérèse.
Hydrochlorothiazide :
L'expérience avec l'hydrochlorothiazide pendant la grossesse est limitée, en particulier au cours du premier trimestre. Les études chez l'animal sont insuffisantes.
L'hydrochlorothiazide traverse le placenta. En raison de son mécanisme d'action, l'utilisation de l'hydrochlorothiazide au cours des deuxième et troisième trimestres de la grossesse peut altérer la perfusion placentaire fœtale et provoquer des effets fœtaux et néonatals tels qu'un ictère, des troubles électrolytiques et une thrombocytopénie.
L'utilisation continue de diurétiques chez les femmes enceintes en bonne santé n'est pas recommandée et expose la mère et le fœtus à des risques inutiles, notamment la jaunisse néonatale, la thrombocytopénie et d'autres effets indésirables qui ont été rapportés chez les adultes sont également possibles.
Il n'existe aucune expérience concernant l'élimination de l'hydrochlorothiazide, qui traverse le placenta, de la circulation néonatale.
L'hydrochlorothiazide ne doit pas être utilisé pour le traitement de l'œdème gestationnel, de l'hypertension gestationnelle ou de la prééclampsie en raison du risque de diminution du volume plasmatique et d'hypoperfusion placentaire sans effet bénéfique sur l'évolution de la maladie.
L'hydrochlorothiazide ne doit pas être utilisé pour le traitement de l'hypertension artérielle primaire chez la femme enceinte, sauf dans de rares situations où aucun autre traitement ne peut être utilisé.
L'heure du repas
Inhibiteurs de l'ECA :
On ne sait pas si le lisinopril est excrété dans le lait maternel car aucune donnée n'est disponible concernant l'utilisation de lisinopril/hydrochlorothiazide pendant l'allaitement. Lisinopril/hydrochlorothiazide n'est pas recommandé et des traitements alternatifs avec un profil de sécurité prouvé pour une utilisation pendant l'allaitement sont préférés, en particulier lors de l'allaitement d'un nouveau-né ou d'un prématuré.
Hydrochlorothiazide :
L'hydrochlorothiazide est excrété dans le lait maternel en petites quantités. Les diurétiques thiazidiques à forte dose provoquent une diurèse intense qui peut inhiber la production de lait. L'utilisation de Zestoretic pendant l'allaitement n'est pas recommandée. Si Zestoretic est pris pendant l'allaitement, les doses doivent être maintenues aussi faibles que possible.
En raison des possibles réactions graves provoquées par l'hydrochlorothiazide chez les bébés allaités, une décision doit être prise s'il est plus approprié d'interrompre l'allaitement ou Zestoretic, en tenant compte de l'importance du médicament pour la mère.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Comme avec les autres antihypertenseurs, l'association lisinopril/hydrochlorothiazide peut avoir un effet léger à modéré sur l'aptitude à conduire des véhicules et à utiliser des machines.
Lors de la conduite de véhicules ou de machines, il faut tenir compte du fait que des étourdissements ou de la fatigue peuvent survenir.
04.8 Effets indésirables
Etudes cliniques
Zestoretic est généralement bien toléré. Dans les études cliniques, les effets indésirables étaient généralement légers et transitoires ; dans la plupart des cas, il n'était pas nécessaire d'interrompre le traitement. Les effets indésirables observés étaient limités à ceux précédemment rapportés avec le lisinopril ou l'hydrochlorothiazide.
Les effets indésirables (EIM) les plus fréquemment rapportés sont des étourdissements, qui ont généralement répondu à une réduction de dose et ont rarement nécessité l'arrêt du traitement, des maux de tête, une toux et une hypotension, y compris une hypotension orthostatique qui peut survenir chez 1 à 10 % des patients traités.
Encore moins fréquents étaient : diarrhée, nausées, vomissements, bouche sèche, éruption cutanée, goutte, palpitations, gêne thoracique, crampes et faiblesse musculaires, paresthésie, asthénie, impuissance, insuffisance rénale aiguë et syncope.
Post-commercialisation
Les effets indésirables suivants ont été observés et rapportés au cours du traitement par lisinopril/hydrochlorothiazide avec les fréquences suivantes : très fréquent (≥1/10), fréquent (≥1/100,
Troubles du système sanguin et lymphatique
Rares : anémie.
Très rare : aplasie médullaire, thrombocytopénie, leucopénie, agranulocytose, anémie hémolytique.
Pathologies endocriniennes
Rare : sécrétion inappropriée d'hormone antidiurétique.
Troubles du métabolisme et de la nutrition
Peu fréquent : goutte.
Rare : hyperglycémie, hypokaliémie, hyperuricémie, hyperkaliémie.
Troubles du système nerveux et troubles psychiatriques
Fréquent : vertiges, maux de tête, paresthésie.
Rare : troubles olfactifs.
Peu fréquent : symptômes dépressifs.
Pathologies cardiaques et vasculaires
Fréquent : effets orthostatiques (y compris hypotension), syncope.
Peu fréquent : palpitations.
Troubles respiratoires, thoraciques et médiastinaux
Fréquent : toux.
Problèmes gastro-intestinaux
Fréquent : diarrhée, nausées, vomissements.
Peu fréquent : bouche sèche.
Rare : pancréatite.
Très rare : œdème de Quincke intestinal.
Troubles hépatobiliaires
Très rare : hépatite à la fois hépatocellulaire et cholestatique, ictère, insuffisance hépatique. Des cas d'hépatite ont très rarement évolué vers une insuffisance hépatique chez certains patients. Les patients recevant Zestoretic qui présentent un ictère ou une élévation marquée des enzymes hépatiques doivent interrompre le traitement par Zestoretic et recevoir une surveillance médicale appropriée.
Affections de la peau et du tissu sous-cutané
Fréquent : éruption cutanée.
Peu fréquent : hypersensibilité / œdème de Quincke : œdème de Quincke du visage, des extrémités, des lèvres, de la langue, de la glotte et/ou du larynx (voir rubrique 4.4).
Très rare : pseudolymphome cutané.
Des symptômes complexes ont été rapportés, pouvant inclure un ou plusieurs des éléments suivants : fièvre, vascularite, myalgie, arthralgie/arthrite, positivité des anticorps antinucléaires (ANA), augmentation de la vitesse de sédimentation des érythrocytes (VS), éosinophilie et leucocytose, éruption cutanée, photosensibilité et autres symptômes dermatologiques. manifestation.
Troubles musculo-squelettiques et du tissu conjonctif
Fréquent : crampes musculaires.
Rare : faiblesse musculaire.
Maladies de l'appareil reproducteur et du sein
Fréquent : impuissance.
Troubles généraux et conditions du site d'administration
Fréquent : fatigue, asthénie.
Peu fréquent : oppression thoracique.
Tests de laboratoire
Fréquent : augmentation de l'urée sanguine, augmentation de la créatinine sérique, augmentation des enzymes hépatiques, diminution de l'hémoglobine.
Peu fréquent : diminution de l'hématocrite.
Rare : augmentation de la bilirubine sérique.
D'autres effets secondaires qui ont été rapportés avec les composants individuels et qui peuvent être des effets secondaires potentiels de Zestoretic sont :
Hydrochlorothiazide (fréquences inconnues) :
Autres effets secondaires : purpura, lupus érymateux systémique, éruption cutanée.
Dans de très rares cas, un syndrome de Stevens-Johnson peut survenir.
Dans des cas isolés : hypercalcémie, cette dernière rendant nécessaire des tests diagnostiques pour mettre en évidence une éventuelle hyperparathyroïdie Des troubles du rythme cardiaque, une myopie aiguë et un glaucome aigu à angle fermé sont possibles.
Lisinopril
* Très rarement, il a été rapporté que chez certains patients le développement indésirable d'une hépatite a évolué vers une insuffisance hépatique. Les patients recevant du lisinopril/hydrochlorothiazide et qui développent un ictère ou une élévation marquée des enzymes hépatiques doivent arrêter le lisinopril/hydrochlorothiazide et recevoir un traitement médical approprié.
** Un complexe de symptômes a été rapporté qui peut inclure un ou plusieurs des éléments suivants : fièvre, vascularite, myalgie, arthralgie/arthrite, positivité des anticorps antinucléaires (ANA), vitesse de sédimentation élevée des globules rouges (VS), éosinophilie et leucocytose, éruption cutanée , photosensibilité ou autres manifestations dermatologiques.
Comme avec les autres inhibiteurs de l'ECA, des hallucinations visuelles et/ou auditives ont été rapportées.
Déclaration des effets indésirables suspectés
La déclaration des effets indésirables suspectés survenant après l'autorisation du médicament est importante car elle permet un suivi continu du rapport bénéfice/risque du médicament. Les professionnels de santé sont invités à déclarer tout effet indésirable suspecté via le système national de déclaration. » adresse https : //www.aifa.gov.it/content/segnalazioni-reazioni-avverse.
04.9 Surdosage
Les données sur le surdosage chez l'homme sont limitées.
Le traitement est symptomatique et de soutien. Le traitement par Zestoretic doit être interrompu et le patient étroitement surveillé. Les mesures thérapeutiques dépendent de la nature et de la gravité des symptômes. Des mesures doivent être prises pour empêcher l'absorption et accélérer l'élimination du médicament.
Les symptômes associés à un surdosage d'inhibiteurs de l'ECA peuvent inclure une hypotension, un choc circulatoire, des troubles électrolytiques, une insuffisance rénale, une hyperventilation, une tachycardie, des palpitations, une bradycardie, des étourdissements, de l'anxiété et de la toux. En cas d'hypotension sévère, le patient doit être placé en position de choc. Le traitement recommandé en cas de surdosage est l'administration d'une solution saline par perfusion intraveineuse. En cas d'hypotension, le patient doit être placé en décubitus dorsal.Si disponible, un traitement par perfusion d'angiotensine II et/ou injection intraveineuse de catécholamines peut également être envisagé.Si l'ingestion est récente, prendre des mesures pour éliminer le lisinopril (par exemple : vomissements, lavage gastrique, administration de médicaments absorbants et sulfate de sodium).
Le lisinopril peut être éliminé de la circulation par hémodialyse (voir rubrique 4.4). Éviter l'utilisation de membranes de dialyse en polyacrylonitrile à haut flux. Le stimulateur cardiaque est indiqué pour la bradycardie résistante au traitement. Les signes vitaux, les électrolytes sériques et la créatinine doivent être surveillés fréquemment.
Les symptômes supplémentaires d'un surdosage en hydrochlorothiazide sont une augmentation du débit urinaire, une dépression de la conscience (y compris le coma), des convulsions, une parésie, des arythmies cardiaques et une insuffisance rénale.
Si de la digitaline a également été administrée, l'hypokaliémie peut accentuer les arythmies cardiaques.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : inhibiteurs de l'ECA, associations - inhibiteurs de l'ECA et diurétiques. Code ATC : C09BA03
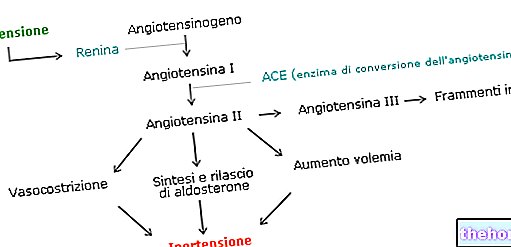
Zestoretic est l'association à dose fixe de lisinopril, un inhibiteur de l'enzyme de conversion de l'angiotensine (ECA) et d'hydrochlorothiazide, un diurétique thiazidique.Les deux composants ont des mécanismes d'action complémentaires et exercent un effet antihypertenseur additif.
Lorsqu'il est associé à d'autres agents antihypertenseurs, une diminution supplémentaire de la pression artérielle peut survenir.
Le lisinopril est un inhibiteur de la peptidyl dipeptidase qui catalyse la conversion de l'angiotensine I en peptide vasoconstricteur de l'angiotensine II.
L'angiotensine II stimule également la sécrétion d'aldostérone par le cortex surrénalien. L'inhibition de l'ECA entraîne une diminution des concentrations d'angiotensine II entraînant une diminution de l'activité vasopressive et une sécrétion d'aldostérone, une élévation de la kaliémie.
Bien que le mécanisme par lequel le lisinopril abaisse la pression artérielle semble être principalement la suppression du système rénine-angiotensine-aldostérone, le lisinopril est également efficace chez les patients souffrant d'hypertension à faible rénine. L'ECA est identique à la kininase II, une enzyme qui dégrade la bradykinine.On ne sait pas si des niveaux accrus de bradykinine, un puissant vasodilatateur, jouent un rôle dans l'effet thérapeutique du lisinopril.
L'hydrochlorothiazide est un agent diurétique et antihypertenseur.Il exerce son action antihypertensive sur le mécanisme de réabsorption électrolytique du tubule rénal distal et augmente l'excrétion des chlorures et du sodium dans une mesure équivalente. La natriurèse peut s'accompagner d'une perte de potassium et de bicarbonates. Le mécanisme de l'effet antihypertenseur des diurétiques thiazidiques n'est pas connu. Les diurétiques thiazidiques n'affectent généralement pas la tension artérielle normale. L'administration concomitante d'autres médicaments antihypertenseurs entraîne une réduction supplémentaire de la tension artérielle.
Deux grands essais contrôlés randomisés (ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) et VA Nephron-D (The Veterans Affairs Nephroopathy in Diabetes)) ont examiné l'utilisation de l'association d'un inhibiteur de l'ECA avec un antagoniste du récepteur de l'angiotensine II.
ONTARGET était une étude menée chez des patients ayant des antécédents de maladie cardiovasculaire ou cérébrovasculaire, ou de diabète sucré de type 2 associé à des signes de lésions organiques. VA NEPHRON-D était une étude menée chez des patients atteints de diabète de type 2 et de néphropathie diabétique.
Ces études n'ont pas démontré d'effet bénéfique significatif sur les résultats rénaux et/ou cardiovasculaires et la mortalité, alors qu'un risque accru d'hyperkaliémie, d'atteinte rénale aiguë et/ou d'hypotension a été observé par rapport à la monothérapie.
Ces résultats sont également pertinents pour d'autres inhibiteurs de l'ECA et antagonistes des récepteurs de l'angiotensine II, étant donné leurs propriétés pharmacodynamiques similaires.
Les inhibiteurs de l'ECA et les antagonistes des récepteurs de l'angiotensine II ne doivent donc pas être utilisés simultanément chez les patients atteints de néphropathie diabétique.
ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints) était une étude visant à vérifier l'intérêt d'ajouter l'aliskiren au traitement standard d'un inhibiteur de l'ECA ou d'un antagoniste des récepteurs de l'angiotensine II chez les patients atteints de diabète de type 2 et d'insuffisance rénale chronique. , maladie cardiovasculaire ou les deux. L'étude a été interrompue prématurément en raison d'un risque accru d'événements indésirables. Les décès d'origine cardiovasculaire et les accidents vasculaires cérébraux étaient tous deux numériquement plus fréquents dans le groupe aliskiren que dans le groupe placebo, et les événements indésirables et les événements indésirables graves d'intérêt ( hyperkaliémie, hypotension et dysfonctionnement rénal) ont été rapportés plus fréquemment dans le groupe aliskiren que dans le groupe placebo.
05.2 "Propriétés pharmacocinétiques
L'administration concomitante de lisinopril et d'hydrochlorothiazide a peu ou pas d'effet sur la biodisponibilité de l'un ou l'autre médicament. L'association préétablie est bioéquivalente aux deux médicaments administrés simultanément.
Absorption
Après administration orale de lisinopril, les concentrations plasmatiques maximales sont observées dans les 7 heures, avec un léger retard chez les patients présentant un infarctus aigu du myocarde. Sur la base de la récupération urinaire, le degré d'absorption du lisinopril est d'environ 25 %, avec une variabilité inter-patient de 6 à 60 % à toutes les doses testées (5 à 80 mg). La biodisponibilité absolue est réduite d'environ 16 % chez les patients atteints d'insuffisance cardiaque. L'absorption de Zestoretic n'est pas affectée par la nourriture.
Distribution
Le lisinopril ne semble pas être lié à d'autres protéines plasmatiques autres que l'ECA circulante.Des études chez le rat indiquent que le lisinopril traverse mal la barrière hémato-encéphalique.
Élimination
Le lisinopril n'est pas métabolisé et est excrété totalement inchangé par les reins. Après plusieurs doses, le lisinopril présente une demi-vie de stockage de 12,6 heures. La clairance du lisinopril chez le sujet sain est d'environ 50 ml/min. La diminution des concentrations sériques montre une phase terminale prolongée qui ne contribue pas à l'accumulation du médicament.Cette phase terminale représente probablement la liaison saturable au niveau de l'ECA et n'est pas proportionnelle à la dose.
Insuffisance hépatique
L'insuffisance hépatique chez les patients cirrhotiques entraîne une diminution de l'absorption du lisinopril (environ 30 % sur la base de la récupération urinaire), mais une augmentation de l'exposition (environ 50 %) par rapport aux sujets sains, en raison d'une diminution de la clairance.
Insuffisance rénale
L'insuffisance rénale réduit l'élimination du lisinopril, qui est excrété par le rein, mais cette réduction ne devient cliniquement importante que lorsque le débit de filtration glomérulaire est inférieur à 30 ml/min.
Paramètres pharmacocinétiques du lisinopril en relation avec la fonction rénale dans différents groupes de patients après administration d'un multiple de la dose de 5 mg.
Avec une clairance de la créatinine de 30 à 80 ml/min, l'AUC moyenne n'a augmenté que de 13 %, tandis qu'une augmentation de 4 à 5 fois a été observée avec une clairance de la créatinine de 5 à 30 ml/min.
Le lisinopril peut être éliminé par dialyse. Au cours d'une hémodialyse de 4 heures, les concentrations plasmatiques de lisinopril diminuent en moyenne de 60 % avec une clairance de dialyse comprise entre 40 et 55 ml/min.
Insuffisance cardiaque
Par rapport aux sujets sains, les patients insuffisants cardiaques ont une exposition plus élevée au lisinopril (une augmentation moyenne de l'ASC de 125 %), mais sur la base de la récupération urinaire du lisinopril, une réduction de l'absorption d'environ 16 % est notée.
personnes agées
Par rapport aux sujets jeunes, les patients âgés présentent des augmentations des concentrations sanguines et de l'ASC (augmentation d'environ 60 %).
Hydrochlorothiazide
Avec une surveillance des taux plasmatiques pendant au moins 24 heures, la demi-vie plasmatique variait de 5,6 à 14,8 heures.
Au moins 61 % de la dose orale sont éliminés sous forme inchangée dans les 24 heures. Après administration orale d'hydrochlorothiazide, l'effet diurétique commence dans les 2 heures, atteint son maximum en 4 heures environ et dure de 6 à 12. L'hydrochlorothiazide traverse la barrière placentaire mais pas la barrière hémato-encéphalique.
05.3 Données de sécurité précliniques
Lisinopril
L'innocuité du lisinopril a été largement étudiée chez les animaux de laboratoire. La DL orale du lisinopril était supérieure à 20 g/kg chez la souris et le rat.
Il apparaît que la toxicité du lisinopril chez le rat et le chien est principalement liée à une exaspération des effets pharmacologiques. Il existe un écart important entre la dose thérapeutique pour l'homme et les doses toxiques pour l'animal.
Le rapport de la dose non toxique pour le chien (5 mg/kg/jour) à celle recommandée pour l'homme de 40 mg/jour était 6 fois plus élevé chez cette espèce sensible.
Chez l'homme, avec une dose de 40 mg/jour, une concentration plasmatique maximale de 468 ng/ml a été atteinte, significativement inférieure au taux plasmatique de 11 370 ng/ml déterminé par une dose néphrotoxique chez le chien.
Les principaux signes de toxicité chez le chien étaient liés à une altération de la fonction rénale (taux élevés de BUN et de créatinine), parfois associés à une dégénérescence tubulaire rénale. Ce dernier n'a pas été observé chez les rats, bien que des augmentations de l'azotémie aient été notées. Ces modifications de la fonction rénale représentent probablement des modifications prérénales de l'azotémie d'origine médicamenteuse liées à l'activité pharmacologique du lisinopril. Un apport supplémentaire de solution saline améliore ou prévient la toxicité du lisinopril chez le rat ainsi que le chien, ce qui renforce encore l'hypothèse d'une toxicité basée sur le mécanisme.
Études de cancérogenèse, mutagenèse et fertilité
Il n'y a eu aucune preuve d'effets oncogènes lorsque le lisinopril a été administré à des rats mâles et femelles pendant 105 semaines à des doses allant jusqu'à 90 mg/kg/jour (environ 110 fois la dose quotidienne maximale recommandée chez l'humain).Le lisinopril a également été administré à 92 semaines chez la souris. (mâle et femelle) à des doses allant jusqu'à 135 mg/kg/jour (environ 170 fois la dose quotidienne maximale recommandée chez l'humain) et n'a montré aucun signe de cancérogénicité. Le lisinopril n'a pas montré de propriétés mutagènes dans le test de mutagénicité microbienne d'Ames avec ou sans activation métabolique. Il a été "testé négatif lors d'un test de mutation précoce" utilisant des cellules pulmonaires de hamster chinois. Le lisinopril n'a pas produit de cassures d'ADN simple brin dans un essai d'élution alcaline in vitro dans des hépatocytes de rat. Le lisinopril n'a pas non plus produit d'augmentation des aberrations chromosomiques dans un test in vitro sur des cellules d'ovaire de hamster chinois et dans une étude in vivo sur la moelle osseuse de souris Aucun effet indésirable des effets sur les performances de reproduction sont survenus chez des rats mâles et femelles traités par des doses de lisinopril allant jusqu'à 300 mg/kg/jour.
Tératogénèse
Le lisinopril n'a pas été tératogène chez les souris traitées avec des doses allant jusqu'à 1000 mg/kg/jour (1250 fois la dose quotidienne maximale recommandée chez l'humain) du 6e au 15e jour de gestation.
Il n'y a pas eu d'augmentation des résorptions fœtales avec des doses inférieures à 100 mg/kg ; à des doses de 1000 mg / kg, cela a été évité par un apport supplémentaire de sel. Il n'y a eu aucune foetotoxicité ou tératogénicité chez les rats traités avec des doses de lisinopril jusqu'à 300 mg/kg/jour (375 fois la dose maximale recommandée) du 6e au 17e jour de gestation.
Chez les rats ayant reçu du lisinopril du 15e jour de la gestation jusqu'au-delà du 21e jour du post-partum, il y a eu une augmentation de l'incidence des décès à la naissance entre le 2e et le 7e jour du post-partum. Le 21e jour du post-partum, le poids corporel moyen des naissances était plus faible. Avec un apport supplémentaire de sel à la mère, il n'y a eu ni augmentation des décès ni diminution du poids des naissances. Chez le lapin, le lisinopril n'a pas montré de tératogénicité lorsqu'il a été administré sur toute la période organogénétique à des doses allant jusqu'à 1 mg/kg/jour en présence d'un apport supplémentaire de sel.
Ce dernier a été utilisé pour éliminer les effets toxiques chez la mère et permettre une évaluation du potentiel tératogène à la dose la plus élevée possible.Le lapin s'est avéré extrêmement sensible aux inhibiteurs de l'enzyme de conversion (captopril et énalapril) montrant des effets maternels et fœtotoxiques. à des doses égales ou inférieures à la dose thérapeutique recommandée pour l'homme.
Chez le lapin, une fœtotoxicité est survenue avec une incidence accrue de résorption fœtale aux doses de 1 mg/kg/jour de lisinopril et avec une incidence accrue d'ossification incomplète à la dose testée la plus faible (0,1 mg/kg/jour). Une dose unique de 15 mg/kg/jour de lisinopril administrée par voie intraveineuse à des lapines gravides au cours des 16e, 21e et 26e jours de gestation a entraîné 88 à 100 % de morts fœtales.
Hydrochlorothiazide
Dans les études de toxicologie aiguë et chronique, l'hydrochlorothiazide s'est avéré avoir une toxicité relativement faible. Dans les études de toxicologie aiguë chez l'animal, la DL50 chez la souris était supérieure à 10 g/kg en suspension per os.
Les chiens ont toléré au moins 2 g/kg par voie orale sans aucun signe de toxicité. L'hydrochlorothiazide a été administré à des rats dans une étude sur deux portées, à des souris dans une étude sur 2 générations et à des lapines avec un test de gestation positif.Aucune de ces études n'a montré d'effets tératogènes de l'hydrochlorothiazide.
La progéniture élevée jusqu'au sevrage ou à la maturité n'a montré aucun signe d'effets liés au traitement.
Études de cancérogenèse, mutagenèse et fertilité
L'hydrochlorothiazide est actuellement à l'étude dans le cadre du programme américain d'essais de cancérogenèse. L'hydrochlorothiazide n'a pas montré de propriétés mutagènes in vitro dans le test de mutagène microbien d'Ames à des concentrations allant jusqu'à 5 mg/boîte en utilisant les souches TA98 et TA100. Les échantillons d'urine de patients traités par l'hydrochlorothiazide n'ont pas montré d'activité mutagène dans le test d'Ames. certains médicaments pour induire la non-disjonction et le croisement ont été mesurés sur Aspergillus nidulans.Un grand nombre de médicaments, dont l'hydrochlorothiazide, ont induit la non-disjonction.
Tératogénèse
Les études de reproduction chez le lapin, la souris et le rat à des doses allant jusqu'à 100 mg/kg/jour (50 fois la dose humaine maximale) n'ont montré aucun signe d'anomalies fœtales externes dues à l'hydrochlorothiazide.
L'hydrochlorothiazide administré dans une étude sur deux générations chez le rat à des doses de 4 à 6 mg/kg/jour (environ 1 à 2 fois la dose humaine maximale recommandée) n'a pas altéré la fertilité ni produit d'anomalies de la progéniture à la naissance.
Lisinopril/hydrochlorothiazide
L'administration de lisinopril avec l'hydrochlorothiazide entraîne des réponses toxiques à des doses plus faibles que celles observées avec chaque composé administré seul. Étant donné que la toxicité de chaque composant est due à son activité thérapeutique (hypotension) et qu'il existe une augmentation de l'activité pharmacologique du lisinopril lorsqu'il est administré en association avec des diurétiques (hydrochlorothiazide), l'augmentation de la toxicité avec les deux médicaments était attendue. de toxicité secondaire à la potentialisation de l'effet pharmacologique a été observée à des doses élevées, il n'y a aucune raison de prédire une réponse toxique chez l'homme aux doses thérapeutiques de l'un ou l'autre des médicaments. L'innocuité du lisinopril et de l'hydrochlorothiazide administrés en association à des posologies thérapeutiques a été démontrée dans des études cliniques.Le lisinopril en association avec l'hydrochlorothiazide n'a pas montré de propriétés mutagènes dans un test de mutagène microbien utilisant Salmonella typhimurium (test d'Ames) ou Escherichia coli avec soit sans activation soit en un test de mutation utilisant des cellules pulmonaires de hamster chinois. Le lisinopril-hydrochlorothiazide n'a pas produit de cassures monoéliques de l'ADN dans le liquide d'élution alcalin lors du test in vitro sur les hépatocytes du rat. De plus, il n'a pas produit d'augmentation des aberrations chromosomiques dans le test in vitro sur cellules ovariennes de hamster chinois ou dans l'étude in vivo sur la moelle osseuse de souris.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Mannitol, phosphate de calcium dibasique dihydraté, amidon de maïs, amidon prégélatinisé, stéarate de magnésium.
06.2 Incompatibilité
Pas connu.
06.3 Durée de validité
30 mois.
06.4 Précautions particulières de conservation
Conservez les comprimés dans la boîte afin de les protéger de la lumière.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Blisters PVC et aluminium
Paquet de 14 comprimés.
Paquet de 28 comprimés.
Paquet de 42 comprimés.
Toutes les présentations peuvent ne pas être commercialisées.
06.6 Instructions d'utilisation et de manipulation
Aucune précaution particulière n'est requise
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
AstraZeneca S.p.A.
Palais Ferraris, Via Ludovico il Moro, 6 / C - Basiglio (MI)
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
"20 mg + 12,5 mg comprimés 14 comprimés" A.I.C. 027482013
"Comprimés 20 mg + 12,5 mg 28 comprimés" A.I.C. 027482025
" Comprimés 20 mg + 12,5 mg 42 comprimés " A.I.C. 027482037
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
29.01.1992/02.2008
10.0 DATE DE RÉVISION DU TEXTE
novembre 2015