DERMOLIN ® est un médicament à base d'acétonide de fluocinolone
GROUPE THÉRAPEUTIQUE : Corticoïdes non associés, préparations dermatologiques

Indications DERMOLIN ® Fluocinolone
DERMOLIN ® est indiqué dans le traitement de toutes les pathologies inflammatoires locales sensibles au traitement par corticoïdes.
Dermatite, eczéma, lésions lupéiques, neurodermes, psoriasis, alopécie, dermatite de contact, piqûres d'insectes, brûlures, ulcères traumatiques et érythème ne sont que quelques-unes des indications thérapeutiques potentielles de l'acétonide de fluocinolone.
Mécanisme d'action DERMOLIN ® Fluocinolone
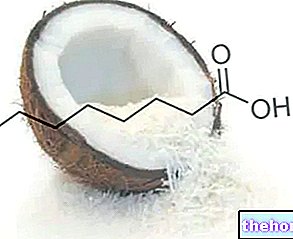
DERMOLIN ® est une spécialité médicinale à base d'acétonide de fluocinolone, un stéroïde dérivé de l'hydrocortinsone et fluoré afin d'optimiser son efficacité thérapeutique.
Comme les autres corticoïdes, la fluocinolone doit également son activité anti-inflammatoire, vasoconstrictrice et anti-allergique à sa capacité à pénétrer facilement les membranes cellulaires, induisant l'expression de la lipocortine, une protéine capable d'inhiber l'enzyme Phosdolipase A2, bloquant la cascade en amont de événements inflammatoires conduisant à la formation de médiateurs de l'inflammation.
Tout cela se traduit par un contrôle optimal des symptômes incriminés et l'amélioration progressive des conditions cliniques du patient.
Tout cela est également facilité par les bonnes propriétés pharmacocinétiques de l'acétonide de fluocinolone appliqué par voie topique, qui en limitant l'absorption systémique du principe actif garantissent une réduction significative des effets secondaires potentiels.
Études réalisées et efficacité clinique
FLUOCINOLONE ET RISQUE D'ATROPHIE CUTANÉE
Suis J Dermatopathol. 31 décembre 2009 : 794-8. doi: 10.1097 / DAD.0b013e3181a9070d.
Un examen histologique pour une atrophie cutanée après 6 mois de traitement avec de l'acétonide de fluocinolone à 0,01 %, de l'hydroquinone à 4 % et de la crème à la trétinoïne à 0,05 %.
Bhawan J, Grimes P, Pandya AG, Keady M, Byers HR, Guevara IL, Colón LE, Johnson LA, Gottschalk R.
Travail qui tente de caractériser le risque d'atrophie cutanée suite à l'application topique de Fluocinolone.Heureusement toutes les données semblent concorder sur le faible risque de cet effet même après des traitements prolongés jusqu'à 24 semaines.
FLUOCINOLONE EN DENTISTERIE
J Endod. Fév 2011 ; 37 : 181-4. doi: 10.1016 / j.joen.2010.10.013.
Effet de l'acétonide de fluocinolone sur les cellules pulpaires dentaires humaines : cytotoxicité, prolifération et formation de matrice extracellulaire.
Muincharern W, Louwakul P, Pavasant P, Lertchirakarn V.
Étude dentaire qui démontre comment de faibles concentrations de FLuocinolone peuvent avoir une efficacité clinique particulière pour favoriser la guérison de la pulpe dentaire enflammée.
FLUOCINOLONE EN CLINIQUE OCULAIRE
Rétine. Mars 2012, 32 : 537-42. doi: 10.1097 / IAE.0b013e31822058bb.
Traitement de l'uvéite postérieure réfractaire chez les patients pédiatriques avec l'implant intravitréen d'acétonide de fluocinolone (Retisert).
Patel CC, Mandava N, Oliver SC, Braverman R, Quiroz-Mercado H, Olson JL.
Étude très récente qui démontre comment les implants à base d'acétonide de fluocinolone peuvent être efficaces pour contrôler l'inflammation postérieure chez les petits patients souffrant de cataracte et de glaucome.
Mode d'emploi et posologie
DERMOLIN®
Onguent d'acétonide de fluocinolone à 0,25%.
Dans la plupart des cas, l'application de la quantité appropriée de pommade directement sur la région touchée par le processus inflammatoire 2 à 3 fois par jour garantit une rémission rapide des symptômes incriminés.
Dans d'autres cas, toujours selon les indications médicales, le traitement pourrait être renforcé par l'utilisation du pansement occlusif, en prenant soin de remplacer le pansement toutes les 12 heures, jusqu'à cicatrisation.
Mises en garde DERMOLIN ® Fluocinolone
Le traitement par DERMOLIN ® doit être précédé d'un examen médical minutieux visant à préciser l'origine des lésions incriminées et l'éventuelle adéquation de la prescription.
Le patient doit également :
- éviter d'appliquer le médicament sur les yeux et les muqueuses;
- éviter d'utiliser le médicament pendant des périodes particulièrement longues;
- éviter l'application du médicament sur des régions cutanées particulièrement larges;
- nettoyez-vous les mains après chaque application;
- contactez votre médecin immédiatement après l'apparition d'effets secondaires.
LA GROSSESSE ET L'ALLAITEMENT
L'utilisation de DERMOLIN ® pendant la grossesse et pendant la période d'allaitement subséquente est généralement contre-indiquée.
Toute utilisation, justifiée par un rapport coût-bénéfice résolument en faveur des bénéfices, doit obligatoirement être encadrée par du personnel médical spécialisé.
Interactions
Il n'y a actuellement aucune interaction médicamenteuse connue digne de mention clinique.
Contre-indications DERMOLIN ® Fluocinolone
L'utilisation de DERMOLIN ® est contre-indiquée chez les patients hypersensibles à la substance active ou à l'un de ses excipients.
Effets indésirables - Effets secondaires
L'utilisation de DERMOLIN ® pourrait déterminer l'apparition d'effets secondaires locaux tels qu'œdème, brûlure, irritation, chute de cheveux, hypertrichose, atrophie cutanée, sécheresse cutanée et dermatite.
Heureusement, tous les effets secondaires systémiques potentiels sont plus rares.
Noter
DERMOLIN ® est un médicament soumis à prescription médicale obligatoire.
Les informations sur DERMOLIN ® Fluocinolone publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.