Ingrédients actifs : Ézétimibe
ZETIA 10 mg comprimés
Pourquoi Zetia est-il utilisé ? Pourquoi est-ce?
ZETIA est un médicament destiné à réduire les taux élevés de cholestérol.
ZETIA diminue les taux de cholestérol total, le « mauvais » cholestérol (cholestérol LDL) et les graisses appelées triglycérides dans le sang. ZETIA augmente également les taux de « bon » cholestérol (cholestérol HDL).
L'ézétimibe, l'ingrédient actif de ZETIA, agit en diminuant l'absorption du cholestérol par l'intestin.
La diminution du taux de cholestérol causée par ZETIA s'ajoute à celle causée par les statines (un groupe de médicaments qui agissent en réduisant la production de cholestérol par l'organisme).
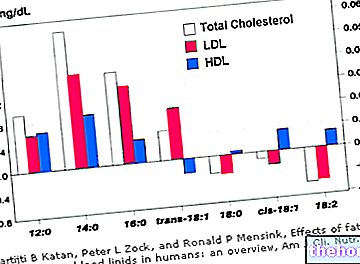
Le cholestérol est l'une des nombreuses substances grasses présentes dans le sang. Le cholestérol total est principalement composé de cholestérol LDL et de cholestérol HDL.
Le cholestérol LDL est souvent appelé « mauvais » cholestérol car il peut s'accumuler dans les parois des artères et former des plaques. Au fil du temps, cette accumulation de plaque peut entraîner un rétrécissement des artères. Ce rétrécissement peut ralentir ou bloquer le flux sanguin vers les organes vitaux tels que le cœur et le cerveau. Ce blocage du flux sanguin peut provoquer une crise cardiaque ou un accident vasculaire cérébral.
Le cholestérol HDL est souvent appelé « bon » cholestérol car il aide à empêcher le mauvais cholestérol de s'accumuler dans les artères et protège contre les maladies cardiaques.
Les triglycérides sont une autre forme de graisse dans le sang qui peut augmenter le risque de maladie cardiaque.
Il est utilisé pour les patients qui ne peuvent pas contrôler leur taux de cholestérol en réduisant leur taux de cholestérol avec un régime seul. Vous devez suivre un régime hypocholestérolémiant pendant que vous prenez ce médicament.
ZETIA est utilisé en complément de votre régime hypocholestérolémiant si vous avez :
- une augmentation du taux de cholestérol sanguin (hypercholestérolémie primaire [hétérozygote familial et non familial])
- avec une statine, lorsque votre cholestérol n'est pas suffisamment contrôlé avec une statine seule
- seul, lorsque le traitement par statine est inapproprié ou non toléré
- une maladie héréditaire (hypercholestérolémie familiale homozygote) qui augmente le taux de cholestérol dans le sang. Une statine vous sera également prescrite et un autre traitement pourra vous être prescrit
- une maladie héréditaire (sitostérolémie homozygote, également connue sous le nom de phytostérolémie) - qui augmente les niveaux de stérols végétaux dans le sang.
ZETIA ne vous aide pas à perdre du poids.
Contre-indications Quand Zetia ne doit pas être utilisé
Si ZETIA vous est prescrit avec une statine, veuillez lire la notice du médicament spécifique que vous prescrivez.
Ne prenez pas ZETIA si :
- vous êtes allergique (hypersensible) à l'ézétimibe ou à l'un des autres composants contenus dans ce médicament (voir rubrique Contenu de l'emballage et autres informations).
Ne prenez pas ZETIA avec une statine si :
- vous avez actuellement des problèmes de foie;
- êtes enceinte ou allaitez.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Zetia
- informez votre médecin de toutes les conditions que vous avez, y compris les allergies;
- votre médecin doit prescrire une analyse de sang avant de commencer un traitement par ZETIA en association avec une statine. C'est pour vérifier comment votre foie fonctionne;
- Votre médecin peut également vous demander de subir des analyses de sang pour vérifier le fonctionnement de votre foie après avoir commencé à prendre ZETIA en association avec une statine.
Si vous avez des problèmes hépatiques modérés ou sévères, l'utilisation de ZETIA n'est pas recommandée.
La sécurité et l'efficacité de ZETIA administré en association avec certains médicaments hypocholestérolémiants, les fibrates, n'ont pas été établies.
Enfants
ZETIA n'est pas recommandé pour les enfants de moins de 10 ans.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Zetia
Informez votre médecin ou pharmacien si vous prenez ou avez pris récemment tout autre médicament, même ceux obtenus sans ordonnance.
En particulier, informez votre médecin si vous prenez des médicaments contenant l'une des substances actives suivantes :
- cyclosporine (souvent utilisé chez les patients transplantés d'organes)
- médicaments contenant une substance active qui empêche la coagulation du sang, tels que la warfarine, la phenprocoumone, l'acénocoumarol ou la fluindione (anticoagulants)
- la cholestyramine (également utilisée pour abaisser le cholestérol), car elle altère l'efficacité de ZETIA
- fibrates (également utilisés pour abaisser le cholestérol).
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Ne prenez pas ZETIA avec une statine si vous êtes enceinte, essayez de le devenir ou si vous pensez être enceinte. Si vous tombez enceinte pendant que vous prenez ZETIA avec une statine, vous devez arrêter les deux traitements immédiatement et contacter votre médecin. Il n'y a pas d'expérience avec l'utilisation de ZETIA pendant la grossesse sans statine. Si vous êtes enceinte, demandez à votre médecin les recommandations appropriées avant d'utiliser ZETIA.
Ne prenez pas ZETIA avec une statine si vous allaitez, car on ne sait pas si les deux médicaments sont excrétés dans le lait maternel. ZETIA, même pris seul sans statine, ne doit pas être utilisé pendant l'allaitement.Demandez à votre médecin les recommandations appropriées.
Demandez conseil à votre médecin ou à votre pharmacien avant de prendre tout médicament.
Conduire et utiliser des machines
Aucun effet de ZETIA sur l'aptitude à conduire des véhicules ou à utiliser des machines n'est attendu. Cependant, il faut tenir compte du fait que certaines personnes peuvent ressentir des vertiges après avoir pris ZETIA.
ZETIA contient du lactose
Les comprimés ZETIA contiennent un sucre appelé lactose. Si votre médecin vous a dit que vous aviez une intolérance à certains sucres, contactez votre médecin avant de prendre ce médicament.
Dose, mode et heure d'administration Comment utiliser Zetia : Posologie
Prenez toujours ZETIA en suivant exactement les indications de votre médecin. Continuez à prendre vos autres médicaments hypocholestérolémiants, à moins que votre médecin ne vous dise d'arrêter de les utiliser. En cas de doute, consultez votre médecin ou votre pharmacien.
- Avant de commencer à prendre ZETIA, vous devez suivre un régime hypocholestérolémiant.
- Vous devez continuer à suivre ce régime hypocholestérolémiant pendant que vous prenez ZETIA.
Adultes, adolescents et enfants (10 à 17 ans) : la dose est d'un comprimé de 10 mg de ZETIA une fois par jour, à prendre par voie orale.
Vous pouvez prendre ZETIA à tout moment de la journée et indépendamment des repas.
Si votre médecin vous a prescrit ZETIA en association avec une statine, les deux médicaments peuvent être pris en même temps. Dans ce cas, veuillez également vous référer aux instructions de dosage spécifiques contenues dans la notice de l'autre médicament prescrit.
Si votre médecin vous a prescrit ZETIA en association avec un autre médicament hypocholestérolémiant contenant le principe actif cholestyramine ou tout autre médicament contenant des chélateurs biliaires, vous devez prendre ZETIA au moins 2 heures avant ou 4 heures après la prise du chélateur biliaire.
Surdosage Que faire si vous avez pris trop de Zetia
Si vous avez pris plus de ZETIA que vous n'auriez dû
Contactez votre médecin ou votre pharmacien.
Si vous oubliez de prendre ZETIA
Ne prenez pas une double dose; reprenez simplement votre dose habituelle à l'heure habituelle le lendemain.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Effets secondaires Quels sont les effets secondaires de Zetia
Comme tous les médicaments, ZETIA est susceptible d'avoir des effets indésirables, bien que tout le monde n'y soit pas sujet.
Les termes suivants sont utilisés pour décrire la fréquence à laquelle des effets secondaires ont été signalés :
- Très fréquent (peut affecter plus de 1 patient sur 10)
- Fréquent (peut affecter jusqu'à 1 patient sur 10)
- Peu fréquent (peut affecter jusqu'à 1 patient sur 100)
- Rare (peut affecter jusqu'à 1 patient sur 1 000)
- Très rare (peut affecter jusqu'à 1 patient sur 10 000, y compris les cas isolés)
Contactez immédiatement votre médecin si vous ressentez des douleurs musculaires inexpliquées, une sensibilité ou une faiblesse musculaire. En effet, en de rares occasions, les problèmes musculaires, y compris les lésions des tissus musculaires entraînant des lésions rénales, peuvent être graves et potentiellement mettre la vie en danger.
Des réactions allergiques ont été rapportées avec l'utilisation courante du médicament, notamment un gonflement du visage, des lèvres, de la langue et/ou de la gorge pouvant entraîner des difficultés à respirer ou à avaler (ce qui nécessite un traitement immédiat).
Lorsqu'il est utilisé seul, les effets indésirables suivants ont été rapportés : Fréquent : douleur abdominale ; la diarrhée; flatulence; se sentir fatigué.
Peu fréquent : augmentations de certains tests de laboratoire hépatiques (transaminases) ou de la fonction musculaire (CK) ; la toux; indigestion; maux d'estomac; la nausée; douleur articulaire; spasmes musculaires; la douleur du cou; diminution de l'appétit ; douleur ; douleur thoracique ; bouffées de chaleur ; hypertension artérielle.
De plus, les effets indésirables suivants ont été rapportés lorsque le médicament a été utilisé avec une statine :
Fréquent : augmentation de certains tests de laboratoire de la fonction hépatique (transaminases) ; mal de tête; douleurs musculaires; sensibilité ou faiblesse musculaire.
Peu fréquent : sensation de picotement ; bouche sèche; démanger; éruption; urticaire; mal au dos; faiblesse musculaire; douleur dans les bras et les jambes; fatigue ou faiblesse inhabituelle; gonflement surtout des mains et des pieds. Lorsqu'il est utilisé avec le fénofibrate, l'effet indésirable fréquent suivant a été rapporté : douleur abdominale.
De plus, les effets indésirables suivants ont été rapportés avec l'utilisation générale du médicament :
vertiges douleurs musculaires; problèmes de foie; réactions allergiques, y compris éruption cutanée et urticaire; éruption rouge surélevée, parfois avec des lésions en forme de cible (érythème polymorphe); douleur musculaire, sensibilité ou faiblesse musculaire; lésion du tissu musculaire; calculs de la vésicule biliaire ou inflammation de la vésicule biliaire (pouvant provoquer des douleurs abdominales, des nausées, des vomissements); inflammation du pancréas souvent accompagnée de douleurs abdominales sévères; constipation; réduction du nombre de cellules sanguines, ce qui peut provoquer des ecchymoses / saignements (thrombocytopénie); sensation de picotement; dépression; fatigue ou faiblesse inhabituelle; essoufflement. Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice.
Expiration et conservation
- Gardez ce médicament hors de la vue et de la portée des enfants.
- N'utilisez pas ce médicament après la date de péremption indiquée sur la boîte ou le récipient après « EXP ». La date d'expiration fait référence au dernier jour de ce mois.
- Ne pas conserver ZETIA au-dessus de 30°C.
Blisters : Conserver dans le contenant d'origine.
Flacons : garder le flacon bien fermé. Ces mesures aideront à protéger le médicament de l'humidité.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Ce que contient ZETIA
- La substance active est l'ézétimibe. Chaque comprimé contient 10 mg d'ézétimibe.
- Les autres composants sont : lactose monohydraté, cellulose microcristalline, povidone, croscarmellose sodique, laurylsulfate de sodium, stéarate de magnésium.
Description de l'apparence de ZETIA et contenu de l'emballage extérieur
Les comprimés ZETIA sont des comprimés blancs à blanc cassé, en forme de gélule, portant l'inscription « 414 » gravée sur une face.
Présentations : 7, 10, 14, 20, 28, 30, 50, 98, 100 ou 300 comprimés dans des plaquettes thermoformées à ouverture par pression ou des plaquettes thermoformées à dose unitaire avec lamina lift ;
84 ou 90 comprimés sous plaquettes thermoformées ;
50, 100 ou 300 comprimés sous plaquettes thermoformées par dose unitaire ; 100 comprimés en flacons.
Toutes les présentations peuvent ne pas être commercialisées
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
ZETIA 10 MG COMPRIMÉS
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Chaque comprimé contient 10 mg d'ézétimibe.
Excipient(s) : Chaque comprimé contient 55 mg de lactose monohydraté.
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Tablette.
Comprimés en forme de gélule blancs à blanc cassé portant l'inscription « 414 » gravée sur une face.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Hypercholestérolémie primaire
ZETIA, administré avec un inhibiteur de l'HMG-CoA réductase (statine), est indiqué en complément d'un régime alimentaire chez les patients atteints d'hypercholestérolémie primaire (familiale hétérozygote et non familiale) qui ne sont pas suffisamment contrôlés par les statines seules.
La monothérapie par ZETIA est indiquée en complément d'un régime alimentaire chez les patients atteints d'hypercholestérolémie primaire (familiale hétérozygote et non familiale) pour lesquels les statines sont considérées comme inappropriées ou ne sont pas tolérées.
Hypercholestérolémie familiale homozygote (FI homozygote)
ZETIA administré avec une statine est indiqué comme traitement d'appoint au régime alimentaire chez les patients atteints d'hypercholestérolémie familiale homozygote. Le patient peut également être soumis à d'autres mesures thérapeutiques (par exemple, l'aphérèse des LDL).
Sitostérolémie familiale homozygote (Phytostérolémie)
ZETIA est indiqué en complément d'un régime alimentaire chez les patients atteints de sitostérolémie familiale homozygote.
Un effet bénéfique de ZETIA sur la morbi-mortalité cardiovasculaire n'a pas encore été démontré.
04.2 Posologie et mode d'administration
Le patient doit suivre un régime hypocholestérolémiant adéquat et doit continuer le régime pendant le traitement par ZETIA.
Le médicament doit être administré par voie orale. La posologie recommandée est d'un comprimé de ZETIA à 10 mg une fois par jour. ZETIA peut être administré à tout moment de la journée, indépendamment des repas.
Lors de l'ajout de ZETIA à une statine, le traitement doit être poursuivi à la posologie initiale commune indiquée pour la statine spécifique ou la posologie plus élevée précédemment prescrite doit continuer à être utilisée. Dans ce cas, la fiche technique de cette statine particulière doit être consultée.
Administration concomitante avec des chélateurs des acides biliaires
L'administration de ZETIA doit avoir lieu ≥ 2 heures avant ou ≥ 4 heures après l'administration d'un agent séquestrant des acides biliaires.
Utilisation chez les personnes âgées
Aucun ajustement posologique n'est nécessaire chez les patients âgés (voir rubrique 5.2).
Utilisation chez les patients pédiatriques
La mise en route du traitement doit être effectuée sous la surveillance d'un spécialiste.
Adolescentes ≥ 10 ans (statut pubertaire : garçons de stade Tanner II et plus et filles en post-ménarche depuis au moins un an) : aucun ajustement posologique n'est nécessaire (voir rubrique 5.2). Cependant, l'expérience clinique chez les patients pédiatriques et adolescents (10 à 17 ans) est limitée.
Lorsque ZETIA est administré avec la simvastatine, les instructions de dosage de la simvastatine chez les adolescents doivent être consultées.
Enfants
Utilisation en cas d'insuffisance hépatique
Aucune adaptation posologique n'est nécessaire en cas d'insuffisance hépatique légère (score de Child-Pugh 5 à 6).Le traitement par ZETIA n'est pas recommandé chez les patients présentant une insuffisance hépatique modérée (score de Child-Pugh 7 à 9) ou sévère (score de Child-Pugh > 9), ( voir rubriques 4.4 et 5.2).
Utilisation en cas d'insuffisance rénale
Aucun ajustement posologique n'est nécessaire en cas d'insuffisance rénale (voir rubrique 5.2).
04.3 Contre-indications
Hypersensibilité à la substance active ou à l'un des excipients.
Lorsque ZETIA est administré en association avec une statine, veuillez vous référer à la fiche technique du médicament.
Le traitement par ZETIA associé à une statine est contre-indiqué pendant la grossesse et l'allaitement.
ZETIA administré en association avec une statine est contre-indiqué chez les patients présentant une maladie hépatique active ou des valeurs élevées de transaminases sériques persistantes et indéterminées.
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Lorsque ZETIA est administré en association avec une statine, veuillez vous référer à la fiche technique du médicament.
Des enzymes hépatiques
Dans les essais cliniques contrôlés au cours desquels des patients ont été traités par ZETIA et une statine, des élévations consécutives des transaminases (≥ 3 fois la limite supérieure de la normale [LSN]) ont été observées. Si ZETIA est co-administré avec une statine, des tests de la fonction hépatique doivent être réalisés au début du traitement et comme recommandé pour la statine (voir rubrique 4.8).
Muscle squelettique
Des cas de myopathie et de rhabdomyolyse ont été rapportés depuis la commercialisation de ZETIA. La plupart des patients qui ont développé une rhabdomyolyse étaient sous traitement concomitant par ZETIA et une statine. Cependant, une rhabdomyolyse a été très rarement rapportée avec ZETIA en monothérapie et très rarement avec l'ajout de ZETIA à d'autres agents connus pour être associés à un risque accru de rhabdomyolyse. Si une myopathie est suspectée sur la base de symptômes musculaires ou confirmée par des taux de créatine phosphokinase (CPK) > 10 fois la limite supérieure de la normale, la prise de ZETIA, de toute statine et de tout autre médicament de ce type que le médicament concomitant doit être interrompu immédiatement.Tous les patients commençant un traitement par ZETIA doivent être informés du risque de myopathie et doivent signaler rapidement toute douleur, sensibilité ou faiblesse musculaire inexpliquée (voir rubrique 4.8).
Insuffisance hépatique
En raison des effets inconnus de l'augmentation de l'exposition à l'ézétimibe chez les patients présentant une insuffisance hépatique modérée ou sévère, ZETIA n'est pas recommandé (voir rubrique 5.2).
Patients pédiatriques (10 à 17 ans)
La sécurité et l'efficacité de ZETIA co-administré avec la simvastatine chez des patients âgés de 10 à 17 ans atteints d'hypercholestérolémie familiale hétérozygote ont été évaluées dans une étude clinique contrôlée chez les garçons adolescents (Tanner stade II et plus) et chez les filles en post-ménarche depuis au moins une année.
Dans cette étude contrôlée limitée, il n'y avait généralement aucun effet sur la croissance ou la maturation sexuelle chez les adolescents, garçons ou filles, ni aucun effet sur la durée du cycle menstruel chez les filles. Cependant, les effets de l'ézétimibe sur une période de traitement > 33 semaines sur la croissance ou la maturation sexuelle n'ont pas été étudiés (voir rubriques 4.2 et 4.8).
La sécurité et l'efficacité de ZETIA co-administré avec des doses de simvastatine supérieures à 40 mg par jour n'ont pas été étudiées chez les patients pédiatriques âgés de 10 à 17 ans.
ZETIA n'a pas été étudié chez les patientes de moins de 10 ans ou chez les filles en pré-ménarche. (voir rubriques 4.2 et 4.8).
L'efficacité à long terme du traitement par ZETIA dans la réduction de la morbidité et de la mortalité chez l'adulte n'a pas été étudiée chez les patients de moins de 17 ans.
Liasses
L'innocuité et l'efficacité de ZETIA administré avec des fibrates n'ont pas été établies.
Si une lithiase biliaire est suspectée chez un patient traité par ZETIA et fénofibrate, des examens de la vésicule biliaire sont indiqués et le traitement doit être interrompu (voir rubriques 4.5 et 4.8).
Cyclosporine
Des précautions doivent être prises lors de l'instauration d'un traitement par ZETIA dans un cadre thérapeutique, y compris l'utilisation de la cyclosporine. Les concentrations de ciclosporine doivent être surveillées chez les patients traités par ZETIA et ciclosporine (voir rubrique 4.5).
Anticoagulants
Si ZETIA est ajouté à de la warfarine, un autre anticoagulant coumarinique ou de la fluindione, le rapport international normalisé (RIN) doit être surveillé de manière appropriée (voir rubrique 4.5).
Excipients
Les patients présentant des problèmes rares d'intolérance au galactose, de déficit en lactase de Lapp ou de malabsorption du glucose et du galactose ne doivent pas prendre ce médicament.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Les études d'interaction n'ont été menées que chez l'adulte.
Dans les études précliniques, il a été démontré que l'ézétimibe n'induit pas les enzymes du cytochrome P450 impliquées dans le métabolisme du médicament. Aucune interaction pharmacocinétique cliniquement significative n'a été observée entre l'ézétimibe et les médicaments métabolisés par les cytochromes P450 1A2, 2D6, 2C8, 2C9 et 3A4, ou la N-acétyltransférase.
Dans les études d'interactions cliniques, l'ézétimibe n'a eu aucun effet sur la pharmacocinétique de la dapsone, du dextrométhorphane, de la digoxine, des contraceptifs oraux (éthinylestradiol et lévonorgestrel), du glipizide, du tolbutamide ou du midazolam lors d'une administration concomitante. La cimétidine, co-administrée avec l'ézétimibe, n'a eu aucun effet sur la biodisponibilité de l'ézétimibe.
Antiacides : L'administration concomitante d'antiacides a diminué le taux d'absorption de l'ézétimibe mais n'a eu aucun effet sur la biodisponibilité de l'ézétimibe. Cette diminution de l'absorption n'est pas considérée comme cliniquement significative.
Cholestyramine : L'administration concomitante de cholestyramine a diminué l'aire sous la courbe moyenne (ASC) de l'ézétimibe total (ézétimibe + ézétimibe-glucuronide) d'environ 55 %. La réduction supplémentaire du cholestérol à lipoprotéines de basse densité (LDL-C) due à l'ajout de ZETIA à la cholestyramine peut être diminuée par cette interaction (voir rubrique 4.2).
Liasses : Chez les patients traités par fénofibrate et ZETIA, les médecins doivent prendre en compte le risque possible de lithiase biliaire et de maladie de la vésicule biliaire (voir rubriques 4.4 et 4.8).
Si une lithiase biliaire est suspectée chez un patient traité par ZETIA et fénofibrate, des examens de la vésicule biliaire sont indiqués et le traitement doit être interrompu (voir rubrique 4.8).
L'administration concomitante de fénofibrate ou de gemfibrozil a modérément augmenté les concentrations totales d'ézétimibe (environ 1,5 et 1,7 fois, respectivement).
L'administration concomitante de ZETIA avec d'autres fibrates n'a pas été étudiée.
Les fibrates peuvent augmenter l'excrétion du cholestérol dans la bile, conduisant à une lithiase biliaire Dans les études animales, l'ézétimibe a parfois augmenté le cholestérol dans la bile de la vésicule biliaire mais pas chez toutes les espèces (voir rubrique 5.3). ne peut être exclu.
Statines : Aucune interaction pharmacocinétique cliniquement significative n'a été observée lorsque l'ézétimibe a été co-administré avec l'atorvastatine, la simvastatine, la pravastatine, la lovastatine, la fluvastatine ou la rosuvastatine.
Ciclosporine : Dans une étude portant sur huit patients transplantés rénaux avec une clairance de la créatinine > 50 ml/min à des doses stables de cyclosporine, l'administration d'une dose unique de 10 mg de ZETIA a entraîné une augmentation de 3,4 fois (intervalle 2, 3 - 7,9 fois) de l'ASC moyenne de l'ézétimibe total par rapport à une population témoin saine d'une autre étude (n = 17) traitée par l'ézétimibe seul. Dans une autre étude, un patient transplanté rénal présentant une insuffisance rénale sévère traité par la ciclosporine et plusieurs autres ézétimibe total 12 fois plus élevé que celui des témoins relatifs traités par ézétimibe seul. Dans une étude croisée sur deux périodes chez douze individus en bonne santé, l'administration quotidienne de 20 mg d'ézétimibe pendant 8 jours avec une dose unique de 100 mg de cyclosporine au jour 7 a entraîné une augmentation moyenne de 15 % de l'ASC de la ciclosporine (intervalle de 10 % diminution à une augmentation de 51 %) par rapport à une dose unique de 100 mg de cyclosporine seule. Aucune étude contrôlée sur l'effet de l'administration concomitante d'ézétimibe sur l'exposition à la cyclosporine chez les patients transplantés rénaux n'a été menée. Des précautions doivent être prises lors de l'instauration d'un traitement par ZETIA dans le cadre de traitements incluant l'utilisation de cyclosporine. Les concentrations de ciclosporine doivent être surveillées chez les patients traités par ZETIA et ciclosporine (voir rubrique 4.4).
Anticoagulants : L'administration concomitante d'ézétimibe (10 mg une fois par jour) n'a eu aucun effet significatif sur la biodisponibilité de la warfarine et le temps de Quick dans une étude portant sur douze hommes adultes en bonne santé. Cependant, il y a eu des rapports post-commercialisation d'augmentations du rapport international normalisé (INR) chez les patients qui avaient ajouté ZETIA à la warfarine ou à la fluindione. Si ZETIA est ajouté à de la warfarine, un autre anticoagulant coumarinique ou de la fluindione, la valeur de l'INR doit être surveillée de manière adéquate (voir rubrique 4.4).
04.6 Grossesse et allaitement
L'administration concomitante de ZETIA avec une statine est contre-indiquée pendant la grossesse et l'allaitement (voir rubrique 4.3), se référer à la fiche technique spécifique de la statine.
Grossesse :
ZETIA ne doit être administré aux femmes enceintes qu'en cas de nécessité absolue. Aucune donnée clinique n'est disponible sur l'utilisation de ZETIA pendant la grossesse.Les études animales sur l'utilisation de l'ézétimibe seul n'ont mis en évidence aucun effet délétère direct ou indirect sur la grossesse, le développement embryofœtal, la naissance ou le développement postnatal (voir paragraphe 5.3).
L'heure du repas :
ZETIA ne doit pas être utilisé pendant l'allaitement. Des études chez le rat ont montré que l'ézétimibe est excrété dans le lait. On ne sait pas si l'ézétimibe est excrété dans le lait maternel.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Aucune étude sur l'aptitude à conduire des véhicules et à utiliser des machines n'a été réalisée.Cependant, lors de la conduite de véhicules ou de l'utilisation de machines, il faut tenir compte du fait que des vertiges ont été rapportés.
04.8 Effets indésirables
Etudes cliniques
Au cours d'essais cliniques d'une durée allant jusqu'à 112 semaines, ZETIA 10 mg/jour a été administré à 2 396 patients seul, avec une statine à 11 308 patients ou avec du fénofibrate à 185 patients. Les effets indésirables étaient généralement légers et transitoires. L'incidence globale des événements indésirables entre ZETIA et le placebo était similaire, de même que le taux d'arrêt pour événements indésirables était comparable entre ZETIA et le placebo.
ZETIA administré seul ou en association avec une statine :
Les effets indésirables suivants ont été observés chez les patients traités par ZETIA (N = 2396) et à une incidence plus élevée que le placebo (N = 1 159) ou chez les patients traités par ZETIA en association avec une statine (N = 11 308) et avec une incidence supérieure à avec la statine administrée seule (N = 9 361) :
Les fréquences sont définies comme : très fréquentes (≥1/10) ; commun (≥1 / 100 à
ZETIA co-administré avec le fénofibrate:
Troubles gastro-intestinaux : douleurs abdominales (fréquent).
Dans une étude clinique multicentrique, en double aveugle, contrôlée contre placebo chez des patients atteints d'hyperlipidémie mixte, 625 patients ont été traités jusqu'à 12 semaines et 576 patients jusqu'à 1 an. Dans cette étude, 172 patients traités par ZETIA et fénofibrate ont terminé 12 semaines de traitement et 230 patients traités par ZETIA et fénofibrate (dont 109 traités par ZETIA seul pendant les 12 premières semaines) ont terminé 1 an de traitement. L'étude n'a pas été conçue pour comparer les groupes de traitement pour les événements peu fréquents. Les taux d'incidence (IC à 95 %) des augmentations cliniquement pertinentes des transaminases sériques (> 3 X LSN, consécutives) étaient de 4,5 % et de 2,7 % pour le fénofibrate seul et pour ZETIA co-administré avec le fénofibrate, ajustés en fonction de l'exposition au traitement, respectivement. les taux de cholécystectomie étaient de 0,6 % et de 1,7 % pour le fénofibrate en monothérapie et pour ZETIA co-administré avec le fénofibrate, respectivement (voir rubriques 4.4 et 4.5).
Patients pédiatriques (10 à 17 ans)
Dans une étude chez des patients adolescents (10 à 17 ans) atteints d'hypercholestérolémie familiale hétérozygote (n = 248), des élévations des ALAT et/ou ASAT (≥ 3 X LSN, consécutives) ont été observées chez 3 % des patients (4 patients) dans le groupe ézétimibe/simvastatine versus 2% des patients (2 patients) dans le groupe simvastatine en monothérapie ; les pourcentages d'augmentation de CPK (≥10 X ULN) étaient de 2 % (2 patients) et de 0 %, respectivement. Aucun cas de myopathie n'a été rapporté.
Cette étude n'était pas adaptée pour comparer les effets indésirables rares des médicaments.
Enquêtes diagnostiques
Dans les essais cliniques contrôlés en monothérapie, l'incidence des élévations cliniquement importantes des transaminases sériques (ALAT et/ou ASAT 3 X LSN, valeurs consécutives) était similaire entre ZETIA (0,5 %) et le placebo (0 Dans les études de co-administration, l'incidence était 1,3 % pour les patients traités par ZETIA en association avec une statine et 0,4 % pour les patients traités par une statine seule. Les élévations étaient généralement asymptomatiques, non associées à une cholestase et sont revenues aux valeurs initiales après l'arrêt du traitement ou avec la poursuite du traitement (voir rubrique 4.4) .
Dans les essais cliniques, des valeurs CPK > 10 X LSN ont été signalées pour 4 des 1 674 (0,2 %) patients traités par ZETIA seul par rapport à 1 des 786 (0,1 %) patients ayant reçu le placebo, et pour 1 des 917 patients (0,1 %) co -administré avec ZETIA et une statine par rapport à 4 des 929 patients (0,4%) traités avec une statine seule. Il n'y avait pas de myopathie ou de rhabdomyolyse excessive associée à ZETIA par rapport à son bras contrôle (placebo ou statine seule). (voir rubrique 4.4.)
Expérience post-commercialisation
Les effets indésirables supplémentaires suivants ont été rapportés au cours de l'expérience post-commercialisation. Étant donné que ces effets indésirables ont été identifiés à partir de notifications spontanées, leurs fréquences réelles ne sont pas connues et ne peuvent être estimées.
Troubles sanguins et lymphatiques : thrombocytopénie
Troubles du système nerveux : étourdissements ; paresthésie
Troubles respiratoires, thoraciques et médiastinaux : dyspnée
Troubles gastro-intestinaux : pancréatite ; constipation
Affections de la peau et du tissu sous-cutané : érythème polymorphe
Troubles musculosquelettiques et du tissu conjonctif : myalgie ; myopathie/rhabdomyolyse (voir rubrique 4.4)
Troubles généraux et anomalies au site d'administration : asthénie
Troubles du système immunitaire : hypersensibilité, y compris éruption cutanée, urticaire, anaphylaxie et œdème de Quincke
Troubles hépatobiliaires : hépatite ; lithiase biliaire ; cholécystite
Troubles psychiatriques : Dépression
04.9 Surdosage
Dans les études cliniques, l'administration d'ézétimibe 50 mg/jour à 15 personnes en bonne santé pendant jusqu'à 14 jours, ou 40 mg/jour à 18 patients atteints d'hypercholestérolémie primaire pendant jusqu'à 56 jours a été généralement bien tolérée. Chez l'animal, aucune toxicité n'a été observée après des doses orales uniques de 5 000 mg/kg d'ézétimibe chez le rat et la souris et de 3 000 mg/kg chez le chien.
Quelques cas de surdosage avec ZETIA ont été rapportés ; la plupart d'entre eux n'étaient pas associés à des expériences indésirables. Les effets indésirables rapportés n'étaient pas graves. En cas de surdosage, des mesures symptomatiques et de soutien doivent être utilisées.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : autres substances modifiant les lipides. Code ATC : C10AX09
ZETIA fait partie d'une nouvelle classe de substances hypolipémiantes qui inhibent sélectivement l'absorption intestinale du cholestérol et des stérols végétaux apparentés. ZETIA est actif par voie orale et possède un mécanisme d'action spécifique qui diffère de celui des autres classes de substances hypocholestérolémiantes ( p. : statines, chélateurs des acides biliaires [résines], dérivés de l'acide fibrique et stanols végétaux.) La cible moléculaire de l'ézétimibe est le transporteur de stérols, Niemann-Pick C1-Like 1 (NPC1L1), responsable de l'absorption intestinale du cholestérol et des phytostérols.
L'ézétimibe est localisé sur la bordure en brosse de l'intestin grêle et inhibe l'absorption du cholestérol provoquant une diminution du passage du cholestérol intestinal dans le foie.
Les statines réduisent la synthèse du cholestérol dans le foie et ces deux mécanismes distincts produisent ensemble une réduction complémentaire du cholestérol. Dans une étude clinique de 2 semaines portant sur 18 patients hypercholestérolémiques, l'ézétimibe a inhibé l'absorption intestinale du cholestérol de 54 % par rapport au placebo.
Une série d'études précliniques a été réalisée pour déterminer la sélectivité de l'ézétimibe dans l'inhibition de l'absorption du cholestérol. L'ézétimibe a inhibé l'absorption du [14C]-cholestérol sans effet sur l'absorption des triglycérides, des acides gras, des acides biliaires, de la progestérone, de l'éthinylœstradiol ou des vitamines liposolubles A et D.
Des études épidémiologiques ont établi que la morbidité et la mortalité cardiovasculaires varient directement avec les taux de cholestérol total et de cholestérol LDL et inversement avec les taux de cholestérol HDL. Un effet bénéfique de ZETIA sur la morbi-mortalité cardiovasculaire n'a pas encore été démontré.
ETUDES CLINIQUES
Dans des essais cliniques contrôlés, ZETIA administré à la fois seul et en association avec une statine a significativement réduit le cholestérol total (C-total), le cholestérol des lipoprotéines de basse densité (LDL-C), l'apolipoprotéine B (Apo B) et les triglycérides (TG) et a augmenté cholestérol à lipoprotéines de haute densité (HDL-C) chez les patients atteints d'hypercholestérolémie.
Hypercholestérolémie primaire
Dans une étude de 8 semaines, en double aveugle, contrôlée contre placebo, 769 patients atteints d'hypercholestérolémie déjà sous statine en monothérapie et n'ayant pas atteint l'objectif de LDL-C selon le National Cholesterol Education Program (NCEP) (depuis 2, 6 à 4,1 mmol / L [100 à 160 mg / dL selon les caractéristiques initiales) ont été randomisés pour recevoir soit ZETIA 10 mg soit un placebo en plus de leur traitement par statine préexistant.
Parmi les patients traités par statine qui n'ont pas atteint l'objectif initial de LDL-C (≈ 82 %), significativement plus de patients randomisés pour recevoir ZETIA ont atteint l'objectif de LDL-C du critère d'évaluation de l'étude par rapport aux patients randomisés pour recevoir le placebo, respectivement 72 % et 19 %. en LDL-C étaient significativement différents (25 % et 4 % pour ZETIA vs placebo, respectivement). De plus, ZETIA, ajouté au traitement par statines, a significativement diminué le C total, l'Apo B, la TG et augmenté le HDL-C par rapport au placebo.
ZETIA ou un placebo ajouté au traitement par statines a réduit la protéine C-réactive médiane de 10 % ou de 0 % par rapport à la valeur initiale, respectivement.
Chez deux patients de 12 semaines, contrôlés par placebo, randomisés, en double aveugle, 1 719 patients atteints d'hypercholestérolémie primaire, ZETIA 10 mg a significativement diminué le C total (13 %), le LDL-C (19 %), l "Apo B (14 % ) et TG (8 %) et une augmentation du HDL-C (3 %) par rapport au placebo. ZETIA n'a également eu aucun effet sur les concentrations plasmatiques de vitamines liposolubles A, D et E au cours du temps de prothrombine et, comme d'autres médicaments hypolipémiants , n'a pas modifié la production d'adrénocorticoïdes.
Dans un essai clinique contrôlé multicentrique, en double aveugle (ENHANCE), 720 patients atteints d'hypercholestérolémie familiale hétérozygote ont été randomisés pour recevoir 10 mg d'ézétimibe en association avec la simvastatine 80 mg (n = 357) ou la simvastatine 80 mg (n = 363) pendant 2 années. L'objectif principal de l'étude était d'étudier l'effet de l'association thérapeutique ézétimibe/simvastatine sur l'épaisseur de la tunique intima et média (IMT) de l'artère carotide par rapport à la simvastatine seule.L'impact de ce marqueur n'est pas encore démontré. pour la morbidité et la mortalité cardiovasculaires.
Le critère d'évaluation principal, le changement moyen de l'IMT des six segments carotidiens, n'était pas significativement différent (p = 0,29) entre les deux groupes de traitement sur la base des mesures échographiques en mode B. Avec l'ézétimibe 10 mg en association avec la simvastatine 80 mg ou la simvastatine 80 mg seule, l'épaisseur de l'intima et de la tunique médiale a augmenté de 0,0111 mm et 0,0058 mm, respectivement, sur la durée de l'étude de 2 ans (au départ, la mesure moyenne de l'IMT carotidienne était 0,68 mm et 0,69 mm, respectivement).
L'ézétimibe 10 mg en association avec la simvastatine 80 mg a diminué significativement plus le LDL-C, le C total, l'Apo B et la TG que la simvastatine 80 mg. Pour les deux groupes de traitement, le pourcentage d'augmentation du C-HDL était similaire. Les effets indésirables rapportés avec l'ézétimibe 10 mg en association avec la simvastatine 80 mg étaient cohérents avec son profil de sécurité connu.
Études cliniques chez des patients pédiatriques (10 à 17 ans)
Dans une étude multicentrique, en double aveugle, contrôlée, 142 garçons (stade Tanner II et plus) et 106 filles post-ménarche, âgés de 10 à 17 ans (âge moyen 14,2 ans) atteints d'hypercholestérolémie familiale hétérozygote (IF hétérozygote) avec LDL-C à l'inclusion des valeurs comprises entre 4,1 et 10,4 mmol/L ont été randomisées pour recevoir ZETIA 10 mg co-administré avec la simvastatine (10, 20 ou 40 mg) ou la simvastatine (10, 20 ou 40 mg) seule pendant 6 semaines, ZETIA et la simvastatine 40 mg co -administré ou simvastatine 40 mg seule pendant les 27 semaines suivantes, puis ZETIA et simvastatine (10 mg, 20 mg ou 40 mg) co-administrés en ouvert pendant 20 semaines.
À la semaine 6, ZETIA co-administré avec la simvastatine (toutes doses) a significativement diminué le C total (38 % vs 26 %), le LDL-C (49 % vs 34 %), l'Apo B (39 % vs 27 %) et les non- HDL-C (47% vs 33%) versus simvastatine seule (toutes doses) Les résultats étaient similaires entre les deux groupes de traitement pour TG et HDL-C (-17% vs -12% et +7% vs +6%, respectivement .) À la semaine 33, les résultats étaient cohérents avec ceux de la semaine 6 et significativement plus de patients sous ZETIA et simvastatine 40 mg (62 %) ont atteint l'objectif thérapeutique idéal selon NCEP AAP (
La sécurité et l'efficacité de ZETIA co-administré avec des doses de simvastatine supérieures à 40 mg par jour n'ont pas été étudiées chez les patients pédiatriques de 10 à 17 ans.L'efficacité n'a pas été étudiée chez les patients de moins de 17 ans. dans la réduction de la morbidité et de la mortalité à l'âge adulte.
Hypercholestérolémie familiale homozygote (FI homozygote)
Une étude randomisée en double aveugle de 12 semaines a inclus 50 patients avec un diagnostic clinique et/ou génotypique de FI homozygote qui ont été traités par l'atorvastatine ou la simvastatine (40 mg) avec ou sans aphérèse concomitante des LDL. ZETIA administré avec de l'atorvastatine (40 ou 80 mg) ou de la simvastatine (40 ou 80 mg) a significativement réduit le LDL-C de 15 % par rapport à l'augmentation de la dose de simvastatine ou d'atorvastatine en monothérapie de 40 à 80 mg.
Sitostérolémie homozygote (phytostérolémie)
Dans une étude de 8 semaines, en double aveugle, contrôlée par placebo, 37 patients atteints de sitostérolémie homozygote ont été randomisés pour recevoir ZETIA 10 mg (n = 30) ou un placebo (n = 7). Certains patients étaient traités avec d'autres médicaments (p. ex., statines, résines). ZETIA a significativement diminué les deux principaux stérols végétaux, le sitostérol et le campestérol, de 21 % et 24 % par rapport à la ligne de base, respectivement. Les effets de la diminution des taux de sitostérol sur la morbidité et la mortalité dans cette population sont inconnus.
Sténose aortique
La simvastatine et l'ézétimibe pour le traitement de la sténose aortique (SEAS) était une étude multicentrique, en double aveugle, contrôlée par placebo d'une durée médiane de 4,4 ans chez 1 873 patients présentant une sténose aortique (SA) asymptomatique, documentée par un vitesse de pointe du flux aortique mesuré par Doppler entre 2,5 et 4,0 m/s. Seuls les patients ont été inclus pour lesquels un traitement par statine n'a pas été jugé nécessaire afin de réduire le risque de maladie cardiovasculaire athéroscléreuse. Les patients ont été randomisés dans un rapport de 1 : 1 pour recevoir un placebo ou 10 mg d'ézétimibe et 40 mg de simvastatine par jour en co-administration.
Le critère d'évaluation principal était le composite des événements cardiovasculaires majeurs (MCE) comprenant la mort cardiovasculaire, le remplacement chirurgical de la valve aortique (AVR), l'insuffisance cardiaque congestive. (ICC) résultant de la progression de la SA, d'un infarctus du myocarde non mortel, d'un pontage aortocoronarien (PAC), d'une intervention coronarienne percutanée (ICP), d'une hospitalisation pour angor instable et d'un accident vasculaire cérébral non hémorragique. Les principaux critères d'évaluation secondaires étaient composés de sous-ensembles des catégories d'événements du critère d'évaluation principal.
Par rapport au placebo, l'association ézétimibe/simvastatine 10/40 mg n'a pas réduit significativement le risque de MCE. Le critère de jugement principal est survenu chez 333 patients (35,3 %) dans le groupe ézétimibe/simvastatine et chez 355 patients (38,2 %) dans le groupe placebo (hazard ratio dans le groupe ézétimibe/simvastatine, 0,96 ; intervalle de confiance 95 %, 0,83 à 1,12 ; p = 0,59) Le remplacement valvulaire aortique a été réalisé chez 267 patients (28,3 %) dans le groupe ézétimibe/simvastatine et chez 278 patients (29,9 %) dans le groupe placebo (hazard ratio, 1,00 ; IC à 95 %, 0,84 à 1,18 ; p = 0,97) Moins de patients ont eu des événements cardiovasculaires ischémiques dans le groupe ézétimibe/simvastatine (n = 148) par rapport au groupe placebo (n = 187) (hazard ratio, 0,78 ; IC à 95 %, 0,63 à 0,97 ; p = 0,02), principalement dus au plus petit nombre de patients qu'ils avaient subi un pontage aorto-coronarien.
Le cancer est survenu plus fréquemment dans le groupe ézétimibe/simvastatine (105 versus 70, p = 0,01). La pertinence clinique de cette observation est incertaine. Dans une méta-analyse qui incluait les résultats intermédiaires de deux grandes études en cours à long terme sur l'association ézétimibe/simvastatine (n = 10 319 traités activement, 10 298 traités avec contrôle ; patients-années = 18 246 traités activement, 18 255 traités avec contrôle) pas d'augmentation du taux d'incidence du cancer (313 traitement actif, 326 témoins) ; risque relatif, 0,96 ; intervalle de confiance à 95 %, 0,82 à 1,12 ; p = 0,61) .
05.2 "Propriétés pharmacocinétiques
Absorption : Après administration orale, l'ézétimibe est rapidement absorbé et largement conjugué au glucuronide phénolique pharmacologiquement actif (ézétimibe-glucuronide). Les pics plasmatiques moyens (Cmax) sont observés en 1 à 2 heures pour l'ézétimibe-glucuronide et en 4 à 12 heures pour l'ézétimibe. la biodisponibilité absolue de l'ézétimibe ne peut pas être déterminée car le composé est pratiquement insoluble dans un milieu aqueux adapté à l'injection.
L'administration concomitante d'aliments (repas riches en matières grasses ou sans matières grasses) n'a eu aucun effet sur la biodisponibilité orale de l'ézétimibe lorsqu'il est administré sous forme de comprimés de ZETIA à 10 mg. ZETIA peut être administré avec ou sans repas.
Distribution : L'ézétimibe et l'ézétimibe-glucuronide sont liés respectivement à 99,7 % et 88 à 92 % aux protéines plasmatiques humaines.
Biotransformation : L'ézétimibe est principalement métabolisé dans l'intestin grêle et le foie par conjugaison au glucuronide (une réaction de phase II) avec excrétion biliaire ultérieure. Un métabolisme oxydatif minimal (une réaction de phase I) a été observé chez toutes les espèces évaluées. L'ézétimibe et le glucuronide d'ézétimibe sont les principaux composés dérivés du médicament trouvés dans le plasma, représentant environ 10 à 20 % et 80 à 90 % du total du médicament présent. dans le plasma, respectivement. L'ézétimibe et l'ézétimibe-glucuronide sont tous deux lentement éliminés du plasma avec des signes de cycles entérohépatiques importants. La demi-vie de l'ézétimibe et de l'ézétimibe-glucuronide est d'environ 22 heures.
Élimination : Après administration orale d'ézétimibe 14C (20 mg) chez l'homme, l'ézétimibe total représentait environ 93 % de la radioactivité plasmatique totale. Environ 78 % et 11 % de la radioactivité administrée ont été récupérés dans les selles et l'urine, respectivement, sur une période de prélèvement d'échantillons de 10 jours. Après 48 heures, aucun niveau de radioactivité n'était détectable dans le plasma.
Populations particulières :
Patients pédiatriques
L'absorption et le métabolisme de l'ézétimibe sont similaires chez les enfants et les adolescents (10 à 18 ans) et les adultes.Il n'y a pas de différences pharmacocinétiques entre les adolescents et les adultes sur la base de l'ézétimibe total. Données pharmacocinétiques dans la population pédiatrique
Patients gériatriques
Les concentrations plasmatiques d'ézétimibe total sont environ deux fois plus élevées chez les personnes âgées (≥ 65 ans) que chez les jeunes (18-45 ans). La réduction du LDL-C et le profil de sécurité sont comparables entre les personnes âgées et les personnes plus jeunes traitées par ZETIA Par conséquent, aucune posologie un ajustement est nécessaire chez les personnes âgées.
Insuffisance hépatique
Après l'administration d'une dose unique de 10 mg d'ézétimibe, l'ASC moyenne de l'ézétimibe total a augmenté d'environ 1,7 fois chez les patients présentant une insuffisance hépatique légère (score de Child Pugh 5 ou 6), par rapport aux sujets sains. Dans une étude à doses multiples de 14 jours (10 mg/jour) chez des patients atteints d'insuffisance hépatique modérée (score de Child Pugh de 7 à 9), l'ASC moyenne de l'ézétimibe total a augmenté d'environ 4 fois par jour 1 et au jour 14 par rapport aux sujets sains. Aucun ajustement posologique n'est nécessaire chez les patients présentant une insuffisance hépatique légère. En raison des effets inconnus de l'augmentation de l'exposition à l'ézétimibe chez les patients présentant une insuffisance hépatique modérée ou sévère (score de Child Pugh > 9), ZETIA n'est pas recommandé chez ces patients (voir rubrique 4.4).
Insuffisance rénale
Après une dose unique de 10 mg d'ézétimibe chez des patients atteints d'insuffisance rénale sévère (n = 8 ; ClCr moyenne ≤ 30 ml/min/1,73 m2), l'ASC moyenne de l'ézétimibe total a augmenté d'environ 1,5 fois par rapport aux sujets sains (n = 9). Ce résultat n'est pas considéré comme cliniquement significatif. Aucun ajustement posologique n'est nécessaire chez les patients insuffisants rénaux.
Un patient supplémentaire dans cette étude (après une greffe de rein et traité par une polychimiothérapie comprenant de la cyclosporine) avait une « exposition » à 12 fois supérieure à l'ézétimibe total.
Sexe
Les concentrations plasmatiques d'ézétimibe total sont légèrement plus élevées (environ 20%) chez les femmes que chez les hommes. La réduction du LDL-C et le profil de tolérance sont comparables entre les hommes et les femmes traités par ZETIA. Aucune adaptation posologique n'est donc nécessaire en fonction du sexe.
05.3 Données de sécurité précliniques
Les études de toxicité chronique de l'ézétimibe chez les animaux n'ont pas identifié d'organes cibles pour les effets toxiques. Chez les chiens traités pendant quatre semaines par l'ézétimibe (≥0,03 mg/kg/jour) la concentration de cholestérol dans la bile kystique a augmenté d'un facteur 2,5 à 3,5. Dans une étude d'un an chez des chiens traités avec des doses allant jusqu'à 300 mg/kg/jour, cependant, aucune augmentation de l'incidence de la lithiase biliaire ou d'autres effets hépatobiliaires n'a été observée. La signification de ces données pour l'homme est inconnue. . Un risque d'effet lithogène lié à l'usage thérapeutique de ZETIA ne peut être exclu.
Dans les études d'administration concomitante d'ézétimibe et de statines, les effets toxiques étaient essentiellement ceux typiquement associés aux statines. Certains des effets toxiques étaient plus prononcés que ceux observés avec le traitement aux statines seules. Ceci est attribué aux interactions pharmacocinétiques et pharmacodynamiques lors d'une administration concomitante. Des interactions de ce type ne se sont pas produites dans les essais cliniques. Des épisodes de myopathie sont survenus chez le rat uniquement après une exposition à des doses plusieurs fois supérieures à la dose thérapeutique chez l'homme (environ 20 fois l'ASC des statines et 500 à 2000 fois l'ASC des métabolites actifs).
Dans une série d'essais in vivo et in vitro L'ézétimibe, administré seul ou co-administré avec des statines, n'a montré aucun potentiel génotoxique.Les tests de cancérogénicité à long terme sur l'ézétimibe ont été négatifs.
L'ézétimibe n'a eu aucun effet sur la fertilité des rats mâles ou femelles, n'était pas tératogène chez les rats ou les lapins, ni n'a affecté le développement prénatal ou postnatal. L'ézétimibe a traversé la barrière placentaire chez les rates gravides et les lapines traitées. avec des doses multiples de 1000 mg/kg / journée. L'administration concomitante d'ézétimibe et de statines n'a pas été tératogène chez le rat. Chez les lapines gravides, un petit nombre de déformations squelettiques ont été observées (fusion des vertèbres thoraciques et caudales, nombre réduit de vertèbres caudales). Il a été démontré que l'administration d'ézétimibe en association avec la lovastatine a des effets mortels sur l'embryon.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Croscarmellose sodique
Lactose monohydraté
Stéarate de magnésium
La cellulose microcristalline
Povidone (K29-32)
Laurylsulfate de sodium
06.2 Incompatibilité
Non pertinent.
06.3 Durée de validité
3 années.
06.4 Précautions particulières de conservation
Ne pas conserver au dessus de 30°C.
Plaquettes thermoformées : à conserver dans l'emballage d'origine pour protéger le médicament de l'humidité.
Flacons : Gardez le flacon bien fermé pour protéger le médicament de l'humidité.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Blister transparent unidose en polychlorotrifluoroéthylène/PVC soudé à un enduit aluminium/papier/polyester à l'aide de résine vinylique. Les comprimés peuvent être extraits en soulevant la feuille d'aluminium / papier / polyester
Boîtes de 7, 10, 14, 20, 28, 30, 50, 98, 100 ou 300 comprimés.
Blister de polychlorotrifluoroéthylène / PVC transparent soudé à un revêtement en aluminium à l'aide de résine vinylique. Les comprimés peuvent être extraits en appuyant sur la poche en plastique.
Boîtes de 7, 10, 14, 20, 28, 30, 50, 84, 90, 98, 100 ou 300 comprimés.
Plaquette thermoformée unidose transparente en polychlorotrifluoroéthylène/PVC enduit d'aluminium. Les comprimés peuvent être extraits en appuyant sur la poche en plastique.
Boîtes de 50, 100 ou 300 comprimés.
Flacon en PEHD avec bouchon en polypropylène, contenant 100 comprimés.
Toutes les présentations peuvent ne pas être commercialisées.
06.6 Instructions d'utilisation et de manipulation
Pas d'instructions particulières.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Neopharmed Gentili S.r.l.
Via San Giuseppe Cottolengo, 15 - 20143 Milan
Italie
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
7 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017010 / M
10 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017022 / M
14 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017034 / M
20 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017251 / M
28 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017046 / M
30 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017059 / M
50 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017061 / M
98 comprimés sous blister unidose PCTFE / PVC / AL / PAPIER AIC n.036017073 / M
100 comprimés en plaquettes unidoses PCTFE / PVC / AL / PAPIER AIC n.036017085 / M
300 comprimés sous blister unidose PCTFE / PVC / AL / PAPIER AIC n.036017097 / M
7 comprimés sous blister PCTFE/PVC/AL AIC n.036017109/M
10 comprimés sous blister PCTFE/PVC/AL AIC n.036017111/M
14 comprimés sous blister PCTFE/PVC/AL AIC n.036017123/M
20 comprimés sous blister PCTFE / PVC / AL AIC n.036017263 / M
28 comprimés sous blister PCTFE/PVC/AL AIC n°036017135/M
30 comprimés sous blister PCTFE/PVC/AL AIC n.036017147/M
50 comprimés sous blister PCTFE/PVC/AL AIC n.036017150/M
84 comprimés sous blister PCTFE/PVC/AL AIC n.036017236/M
90 comprimés sous blister PCTFE/PVC/AL AIC n°036017224/M
98 comprimés sous blister PCTFE/PVC/AL AIC n.036017162/M
100 comprimés sous blister PCTFE/PVC/AL AIC n.036017174/M
300 comprimés sous blister PCTFE/PVC/AL AIC n.036017186/M
50 comprimés sous blister unidose PCTFE/PVC/AL AIC n.036017198/M
100 comprimés sous blister unidose PCTFE / PVC / AL AIC n.036017200 / M
300 comprimés en plaquettes unidoses PCTFE / PVC / AL AIC n.036017212 / M
100 comprimés en flacon PEHD AIC n.036017248/M
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
Février 2007
10.0 DATE DE RÉVISION DU TEXTE
juin 2011









--dimetindene.jpg)