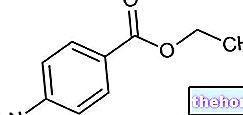
SOLARAZE ® est un médicament à base de diclofénac sodique
GROUPE THÉRAPEUTIQUE : Autres préparations dermatologiques

Indications SOLARAZE ® Diclofénac
SOLARAZE ® est utilisé dans le traitement de la kératose actinique.
Mécanisme d'action SOLARAZE ® Diclofénac
La kératose actinique, également appelée kératose solaire, est une pathologie dermatologique caractérisée par la présence de plaques pouvant atteindre un cm, squameuses, jaunâtres, rugueuses et parfois entourées de télangiectasies.
La pathogenèse, encore mal connue, voit cependant les rayons ultraviolets comme principaux coupables, à tel point que les sites dans lesquels ces lésions sont observées sont ceux chroniquement exposés au soleil, tels que le visage, le cuir chevelu, les joues, le visage, les mains. et les oreilles.
L'une des applications thérapeutiques potentielles de cette pathologie précancéreuse, bien qu'encore non étayée par des mécanismes moléculaires clairs et définis, implique l'utilisation topique d'anti-inflammatoires non stéroïdiens et plus précisément de diclofénac à 3 %, qui s'est avéré efficace pour réduire la progression et incidence des complications. .
Le principal mécanisme thérapeutique du diclofénac, également dans ce cas, est déterminé par l'action inhibitrice exercée sur les cyclooxygénases, et par la réduction relative des concentrations de prostaglandines, impliquées dans la pathogenèse de la kératose actinique.
Études réalisées et efficacité clinique
1 LES MÉCANISMES MOLÉCULAIRES DU DICLOFÉNAC DANS LA KÉRATOSE ACTINIQUE
Frère J Dermatol. Mai 2007 ; 156 Suppl 3 : 25-33.
Le rôle de l'apoptose dans le traitement et la prophylaxie des tumeurs épithéliales par les anti-inflammatoires non stéroïdiens (AINS).
Fecker LF, Stockfleth E, Nindl I, Ulrich C, Forschner T, Eberle J.
Etude visant à élucider les mécanismes moléculaires sous-tendant l'activité thérapeutique des anti-inflammatoires non stéroïdiens dans le traitement de la kératose actinique.En plus de l'action anti-inflammatoire, ce principe actif semble exercer une action apoptotique directe contre les kératinocytes hyperplasiques .
2. DICLOFÉNAC ET PHOTOTOXICITÉ "
Eur J Dermatol. juillet-août 2006, 16 : 385-90.
Le diclofénac à 3 % dans l'acide hyaluronique à 2,5 % (Solaraze) n'induit pas de photosensibilité ou de phototoxicité seul ou en association avec des écrans solaires.
Ortonne JP, Queille-Roussel C, Duteil L.
Ce travail montre comment l'application de diclofénac 3% dans l'hyaluronate en même temps que celle de crèmes solaires n'induit pas de réactions phototoxiques ou photosensibilisantes.Ces données sont très importantes compte tenu du rôle des UV dans la pathogenèse de la kératose actinique.
3. Dermatite de contact SOLARAZE ® E
Contact Dermatite. Octobre 2004 ; 51 : 215-6.
Dermatite de contact de 3 allergènes différents dans le gel Solaraze.
Kleyn CE, Bharati A, Roi CM.
Etude qui démontre le pouvoir allergène de certains excipients présents dans SOLARAZE. Malheureusement, cette réaction, caractérisée par la présence d'une dermatite de contact, limite considérablement l'utilisation topique du diclofénac, réduisant son efficacité thérapeutique.
Mode d'emploi et posologie
SOLARAZE®
Gel à usage topique avec 3% de diclofénac sodique (soit 30 mg de principe actif pour chaque gramme de gel)
Le traitement thérapeutique des kératoses solaires consiste à prendre 3 % de diclofénac deux fois par jour pendant au moins 30 jours, en appliquant une quantité de gel suffisante pour couvrir la zone touchée.
Pour faciliter l'absorption, il est nécessaire de masser doucement la pièce, en évitant d'abîmer les écailles.
Le traitement doit être supervisé par votre médecin
Mises en garde SOLARAZE ® Diclofénac
Le traitement par SOLARAZE ® doit être supervisé par votre médecin, afin que ce dernier puisse évaluer l'efficacité et l'innocuité de la thérapie mise en œuvre.
Le patient doit éviter tout contact du médicament avec les yeux, les muqueuses et toute zone endommagée, afin de minimiser l'absorption systémique de l'ingrédient actif et l'apparition d'effets secondaires.
Il est également utile pour le patient de ne pas s'exposer au soleil pendant le traitement.
La présence dans SOLARAZE ® d'excipients tels que l'alcool benzylique et l'hyaluronate de sodium pourrait augmenter le risque de réactions allergiques surtout chez les patients atopiques.
LA GROSSESSE ET L'ALLAITEMENT
L'absence d'études relatives aux effets secondaires du diclofénac topique sur la santé fœtale et celles connues associées à la prise orale, étendent les contre-indications également à la grossesse et à la période ultérieure d'allaitement.
Interactions
L'absorption systémique réduite qui est observée suite à l'application topique de diclofénac, permet de limiter considérablement les interactions médicamenteuses possibles avec d'autres ingrédients actifs, rendant l'utilisation de SOLARAZE ® aussi sûre que possible.
Contre-indications SOLARAZE ® Diclofénac
L'utilisation de SOLARAZE® est contre-indiquée chez les patients hypersensibles à la substance active ou à l'un de ses excipients, ainsi qu'aux salicylates et à tout autre type d'anti-inflammatoire non stéroïdien.
Effets indésirables - Effets secondaires
L'administration topique de SOLARAZE ® pourrait être associée à l'apparition de réactions locales telles que l'urticaire, les éruptions cutanées, la conjonctivite et la dermatite de contact.
Des effets indésirables cliniquement significatifs n'ont été que rarement observés, pour lesquels il a été nécessaire d'arrêter le traitement.
Noter
SOLARAZE ® ne peut être vendu que sur prescription médicale.
Les informations sur SOLARAZE ® Diclofénac publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.