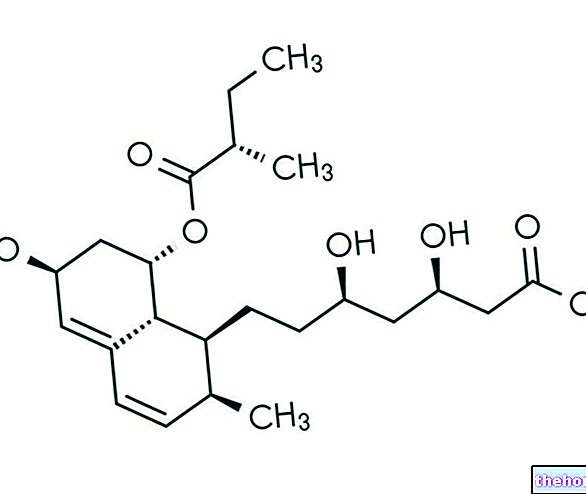
PANTORC ® est un médicament à base de Pantoprazole.
GROUPE THÉRAPEUTIQUE : Antireflux - Antiulcéreux - Inhibiteurs de la pompe acide

Indications PANTORC ® Pantoprazole
PANTORC ® est utilisé à la fois comme traitement pharmacologique préventif des ulcères gastriques et duodénaux induits par l'utilisation prolongée d'anti-inflammatoires non stéroïdiens, et comme traitement thérapeutique des symptômes associés au reflux gastro-œsophagien.
PANTORC ® Mécanisme d'action Pantoprazole
Le pantoprazole contenu dans PANTORC ® représente un principe actif appartenant à la catégorie pharmacologique des inhibiteurs de la pompe à protons, avec des caractéristiques pharmacocinétiques avantageuses par rapport à l'oméprazole plus courant.
En effet, une fois pris par voie orale, le pantoprazole est rapidement absorbé au niveau duodénal, atteignant la concentration plasmatique maximale en seulement 2 heures, avec une biodisponibilité qui est d'environ 77% de la dose totale prise, donc significativement supérieure à 30,% de l'oméprazole.
D'un point de vue pharmacodynamique, cependant, le mécanisme d'action de ce principe actif reste presque inchangé, pouvant agir sélectivement sur la pompe H + / K + ATPase, exprimée par les cellules pariétales de l'estomac, inhibant ainsi à la fois basale et sécrétion d'acide induite par des hormones, comme la gastrine ou l'histamine.
Il est donc évident que son utilisation prolongée de ce médicament s'accompagnera d'une augmentation significative des concentrations sanguines de gastrine, heureusement sans répercussion cliniquement évidente.
Une fois son action terminée, après une demi-vie très rapide, le médicament est principalement éliminé par voie rénale.
Études réalisées et efficacité clinique
1. PANTOPRAZOLE ET REFLUX GASTROOESOPHAGEEUX
MMW Fortschr Med. 8 avril 2010, 152 Suppl 1 : 21-9.
Efficacité et tolérance du pantoprazole dans le traitement du reflux gastro-œsophagien.
Gillessen A.
Le traitement par pantoprazole s'est avéré particulièrement efficace pour traiter les symptômes associés au reflux gastro-œsophagien, entraînant une réduction des cas de brûlures d'estomac, de sécrétions acides, de douleurs abdominales et de dyspepsie sans effets secondaires particuliers en une seule semaine de traitement.
2. PANTOPRAZOLE ET HEMORRAGE GASTRO-INTESTINAL
J Soins critiques. 25 janvier 2011.
Pantoprazole pour la prévention des saignements gastro-intestinaux chez les patients à haut risque atteints de syndromes coronariens aigus.
Wu H, Jing Q, Wang J, Guo X.
Les patients atteints d'un syndrome coronarien aigu ont un risque élevé de développer une hémorragie gastro-intestinale. Chez ces patients, l'administration de 40 mg de pantoprazole par jour pendant 7 jours s'est avérée utile pour réduire significativement l'incidence de cet effet indésirable, sans effets secondaires particuliers.
3. RÉACTIVITÉ CROISÉE " ENTRE LES INHIBITEURS DE LA POMPE PROTONIQUE
Clin Drug Investig. 2010 ; 30 : 559-63.
Neutropénie induite par les inhibiteurs de la pompe à protons : réactivité croisée possible entre l'oméprazole et le pantoprazole.
Gouraud A, Vochelle V, Descotes J, Vial T.
La réactivité croisée entre différents inhibiteurs de la pompe à protons a été établie dans ce cas clinique, dans lequel un homme de 60 ans a présenté une neutropénie à la fois après avoir pris de l'oméprazole et du pantoprazole après environ 1 an. Ces données sont importantes pour souligner la présence d'une réactivité croisée entre différents inhibiteurs de la pompe à protons.
Mode d'emploi et posologie
PANTORC® Comprimés gastro-résistants de 20-40 mg de pantoprazole :
tant dans le traitement des symptômes associés au reflux gastro-oesophagien que dans la prévention des ulcères et de l'oesophagite, la dose journalière efficace est de 20 mg à prendre de préférence le matin avec un verre d'eau.
En général, la rémission des symptômes s'observe en 2 à 4 semaines de traitement, au terme desquelles une prise ponctuelle pourra être utilisée suite à la réapparition des symptômes.
Des variations de posologie pourraient être prévues dans des catégories particulières de patients à risque, et toujours sous stricte indication de votre médecin.
PANTORC ® Pantoprazole Avertissements
Avant de prendre PANTORC®, le médecin doit effectuer une « évaluation clinique minutieuse du patient, visant à exclure l'origine maligne des pathologies du tractus gastro-intestinal, afin que l'action thérapeutique du médicament, capable de masquer certains symptômes particulièrement importants, ne retardez pas son diagnostic.
La diminution de la teneur en acide de l'estomac, très importante suite à une prise prolongée de pantoprazole, pourrait entraîner une diminution de l'absorption de la vitamine B12, avec l'apparition de symptômes associés.
L'administration de PANTORC ® doit être effectuée avec une prudence particulière chez les patients présentant une insuffisance rénale et hépatique.
LA GROSSESSE ET L'ALLAITEMENT
Les études actuellement présentes dans la littérature, statiquement insignifiantes, ne permettent pas de caractériser complètement le profil de tolérance du pantoprazole, lorsqu'il est pris pendant la grossesse.
Par conséquent, nous ne recommandons pas l'utilisation de PANTORC ® pendant la grossesse et la période de lactation subséquente.
Interactions
Comme on le sait, le pantoprazole est métabolisé par le système du cytochrome p450, également impliqué dans le métabolisme des principes actifs élevés tels que la carbamazépine, la caféine, le diazépam, le diclofénac, la digoxine, l'éthanol, le glibenclamide, le métoprolol, le naproxène, la nifédipine, la phénytoïne, le piroxicam, la théophylline et contraceptifs.
Au vu de ces données, d'éventuelles interactions pourraient altérer les propriétés pharmacocinétiques normales des composés ci-dessus, avec une importance particulière pour les anticoagulants, pour lesquels une surveillance continue du temps de Quick serait nécessaire.
De plus, la prise concomitante d'atazanavir et de pantoprazole pourrait entraîner une réduction significative de l'absorption de l'atazanavir, qui est obtenue en raison de la teneur en acide de l'estomac.
Contre-indications PANTORC ® Pantoprazole
PANTORC® est contre-indiqué chez les patients présentant une hypersensibilité connue à la substance active ou à l'un de ses excipients. Compte tenu de la possibilité de réactivité croisée, il serait conseillé d'éviter de prendre le médicament même en cas d'hypersensibilité à des composés analogues au pantoprazole.
Effets indésirables - Effets secondaires
Les essais cliniques et la surveillance post-commercialisation ont montré des effets secondaires particulièrement fréquents, tels que maux de tête, somnolence et vertiges, douleurs abdominales, nausées, vomissements, diarrhée, constipation et flatulence, suite à la prise de PANTORC ®
Les cas d'apparition de lésions hépatiques, rénales, hématologiques et cutanées, qui ont rapidement disparu après l'arrêt du traitement, ont été plus rares.
Noter
PANTORC ® un médicament qui ne peut être vendu que sur prescription médicale.
Les informations sur PANTORC ® Pantoprazole publiées sur cette page peuvent être périmées ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.










-e-agorafobia.jpg)