Ingrédients actifs : Nomegestrol (Acétate de Nomegestrol), Estradiol
Zoely 2,5 mg / 1,5 mg comprimés pelliculés
Indications Pourquoi Zoely est-il utilisé ? Pourquoi est-ce?
Zoely est une pilule contraceptive utilisée pour prévenir la grossesse.
- Les 24 comprimés pelliculés blancs sont des comprimés actifs qui contiennent une petite quantité de deux hormones féminines différentes. Ces hormones sont l'acétate de nomégestrol (un progestatif) et l'estradiol (un œstrogène).
- Les 4 comprimés jaunes sont des comprimés inactifs qui ne contiennent pas d'hormones et sont appelés comprimés placebo.
- Les pilules contraceptives qui contiennent deux hormones différentes, comme Zoely, sont appelées « pilules combinées ».
- L'œstradiol, l'œstrogène contenu dans Zoely, est identique à l'hormone produite par les ovaires pendant le cycle menstruel.
- L'acétate de nomégestrol, le progestatif contenu dans Zoely, est dérivé de l'hormone progestérone.La progestérone est produite par les ovaires pendant le cycle menstruel.
À savoir sur les contraceptifs hormonaux combinés (COC) :
- Lorsqu'elles sont utilisées correctement, elles constituent l'une des méthodes de contraception réversibles les plus fiables.
- Les COC augmentent légèrement le risque de caillot sanguin dans les veines et les artères, en particulier au cours de la première année d'utilisation ou si vous reprenez l'utilisation d'un contraceptif hormonal combiné après une interruption de 4 semaines ou plus.
- Soyez prudent et consultez votre médecin si vous pensez présenter des symptômes dus à un caillot sanguin (voir rubrique « Caillots sanguins »).
Contre-indications Quand Zoely ne doit pas être utilisé
Considération générale
Avant de commencer à utiliser Zoely, vous devez lire les informations sur les caillots sanguins (thrombose). Il est particulièrement important de lire le texte sur les symptômes liés à un caillot sanguin - voir la section "Caillots sanguins").
Avant de pouvoir commencer à prendre Zoely, votre médecin vous posera quelques questions sur vos antécédents médicaux et les antécédents médicaux de vos proches. Votre médecin mesurera également votre tension artérielle et, en fonction de votre situation personnelle, pourra également effectuer d'autres tests.
Cette notice décrit plusieurs situations dans lesquelles vous devrez arrêter de prendre la pilule, ou dans lesquelles la fiabilité de la pilule peut être réduite. Dans ces situations, vous devrez renoncer à avoir des relations sexuelles ou prendre des mesures contraceptives non hormonales supplémentaires, par ex. un préservatif ou une autre méthode de barrière. N'utilisez pas de méthodes basées sur les rythmes biologiques ou la température. Ces méthodes peuvent être inefficaces, car la pilule modifie les changements de température corporelle et de glaire cervicale qui se produisent normalement pendant le cycle menstruel.
Zoely, comme les autres contraceptifs hormonaux, ne protège pas contre l'infection par le VIH (SIDA) ou d'autres maladies sexuellement transmissibles.
Vous ne devez pas utiliser Zoely si l'une des conditions énumérées ci-dessous s'applique à vous. Si l'une des conditions énumérées ci-dessous s'applique à vous, vous devez en informer votre médecin. Votre médecin discutera avec vous d'autres méthodes contraceptives plus appropriées.
- si vous avez (ou avez déjà eu) un caillot sanguin dans un vaisseau sanguin des jambes (thrombose veineuse profonde, TVP), des poumons (embolie pulmonaire, EP) ou d'autres organes ;
- si vous savez que vous souffrez d'un trouble de la coagulation tel qu'un déficit en protéine C, en protéine S, en antithrombine III, en facteur V Leiden ou en anticorps antiphospholipides ;
- si vous devez subir une intervention chirurgicale ou vous allonger pendant une longue période (voir rubrique « Caillots sanguins »);
- si vous avez déjà eu une crise cardiaque ou un accident vasculaire cérébral;
- si vous avez (ou avez eu dans le passé) une angine de poitrine (une affection qui provoque des douleurs thoraciques sévères et peut être le premier signe d'une crise cardiaque) ou un accident ischémique transitoire (AIT - symptômes temporaires d'AVC) ;
- si vous souffrez de l'une des maladies suivantes pouvant augmenter le risque de caillots sanguins dans les artères :
- diabète sévère avec atteinte vasculaire
- très haute tension artérielle
- taux très élevés de graisses dans le sang (cholestérol ou triglycérides) ;
- une condition appelée hyperhomocystéinémie
- si vous avez (ou avez déjà eu) un type de migraine appelé « migraine avec aura » ;
- si vous avez (avez déjà eu) « une inflammation du pancréas (pancréatite) associée à des taux élevés de graisse dans le sang ;
- si vous avez (avez eu) une maladie hépatique grave et que votre foie ne fonctionne toujours pas comme il le devrait ;
- si vous avez (avez déjà eu) une tumeur bénigne ou maligne du foie ;
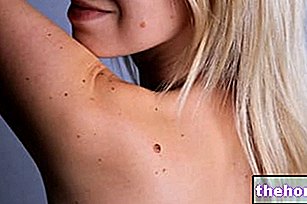
- si vous avez (avez eu) ou pourriez avoir un cancer du sein ou des organes génitaux ;
- si vous avez des saignements vaginaux d'origine inconnue.
- si vous êtes allergique à l'estradiol ou à l'acétate de nomégestrol ou à l'un des autres composants contenus dans ce médicament.
Si l'une de ces conditions apparaît pour la première fois lors de l'utilisation de Zoely, arrêtez immédiatement de le prendre et informez votre médecin. En attendant, utilisez un contraceptif non hormonal. Voir aussi "Considérations générales"
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Zoely
Quand devez-vous contacter votre médecin ? Contactez votre médecin de toute urgence
- si vous remarquez des signes possibles d'un caillot sanguin pouvant indiquer que vous avez un caillot dans la jambe (c'est-à-dire une thrombose veineuse profonde), un caillot dans les poumons (c'est-à-dire une "embolie pulmonaire), une crise cardiaque ou un accident vasculaire cérébral (voir rubrique" Caillots de sang "ci-dessous).
Pour une description des symptômes caractéristiques de ces effets secondaires graves, voir "Comment reconnaître un caillot sanguin".
- si vous constatez une modification de votre état de santé, notamment en ce qui concerne les aspects mentionnés dans cette notice (voir également rubrique « Quand vous ne devez pas utiliser Zoely » ; n'oubliez pas les modifications de l'état de santé de vos proches) ;
- si vous remarquez une grosseur dans votre sein;
- si vous présentez des symptômes d'œdème de Quincke tels que gonflement du visage, de la langue et/ou de la gorge et/ou difficulté à avaler ou urticaire avec difficulté à respirer ;
- si vous devez utiliser d'autres médicaments (voir également la rubrique « Autres médicaments et Zoely »);
- si vous devez vous allonger longtemps ou subir une intervention chirurgicale (prévenez votre médecin au moins quatre semaines à l'avance) ;
- si vous avez des saignements vaginaux inhabituels et intenses ;
- si vous avez oublié deux comprimés ou plus au cours de la première semaine de la plaquette et avez eu des relations sexuelles au cours des sept jours précédents (voir également la rubrique « Si vous oubliez de prendre Zoely »);
- en cas de diarrhée sévère ;
- si vous n'avez pas de règles et pensez être enceinte (ne commencez pas la bandelette suivante sans consulter votre médecin, voir également la rubrique « Si vous n'avez pas eu vos règles une ou plusieurs fois »).
Informez votre médecin si l'un des cas suivants s'applique à vous.
Vous devez également informer votre médecin si l'une de ces conditions apparaît ou s'aggrave lors de l'utilisation de Zoely.
- si vous souffrez d'un angio-œdème héréditaire. Consultez immédiatement votre médecin si vous présentez des symptômes d'œdème de Quincke tels qu'un gonflement du visage, de la langue et/ou de la gorge et/ou des difficultés à avaler ou de l'urticaire avec des difficultés respiratoires. Les préparations contenant des œstrogènes peuvent induire ou aggraver les symptômes d'un « œdème de Quincke ;
- si un parent proche a ou a eu un cancer du sein ;
- si vous souffrez d'épilepsie (voir rubrique « Autres médicaments et Zoely »);
- si vous souffrez d'une maladie du foie (par exemple, la jaunisse) ou d'une maladie de la vésicule biliaire (par exemple, des calculs biliaires) ;
- si vous êtes diabétique ;
- si vous souffrez de dépression ;
- si vous avez la maladie de Crohn ou la rectocolite hémorragique (maladie inflammatoire chronique de l'intestin) ;
- si vous souffrez de lupus érythémateux disséminé (LED, une maladie qui affecte le système de défense de l'organisme) ;
- si vous souffrez d'un syndrome hémolytique et urémique (SHU, un trouble de la coagulation provoquant une insuffisance rénale) ;
- si vous souffrez d'anémie falciforme (une maladie héréditaire des globules rouges) ;
- si vous avez des taux élevés de graisses dans le sang (hypertriglycéridémie) ou des antécédents familiaux positifs de cette maladie. L'hypertriglycéridémie a été associée à un risque accru de développer une pancréatite (inflammation du pancréas);
- si vous devez subir une intervention chirurgicale ou vous allonger pendant une longue période (voir sous « Caillots sanguins »).
- si vous venez d'accoucher, le risque de caillots sanguins est plus élevé. Demandez à votre médecin combien de temps après avoir eu un bébé vous pouvez commencer à prendre Zoely.
- si vous avez « une inflammation des veines sous la peau (thrombophlébite superficielle) ;
- si vous avez des varices.
- si vous avez une maladie qui est apparue pour la première fois ou s'est aggravée pendant la grossesse ou lors d'une utilisation antérieure d'hormones sexuelles (par exemple, perte auditive, porphyrie [un trouble sanguin], herpès gestationnel [éruption avec cloques pendant la grossesse], chorée de Sydenham [une maladie nerveuse dans laquelle des mouvements corporels soudains se produisent], (voir rubrique « Quand contacter votre médecin » );
- si vous avez (ou avez eu dans le passé) un chloasma [taches pigmentées jaune-brun, appelées « taches de grossesse », en particulier sur le visage]. Si c'est le cas, évitez une exposition excessive au soleil ou aux ultraviolets
CAILLOTS SANGUINS
L'utilisation d'un contraceptif hormonal tel que Zoely augmente votre risque de développer un caillot par rapport au risque que vous auriez si vous ne l'utilisiez pas. Dans de rares cas, un caillot sanguin peut bloquer les vaisseaux sanguins et causer de graves problèmes.
Des caillots peuvent se développer
- dans les veines (appelées « thrombose veineuse », « thromboembolie veineuse » ou TEV)
- dans les artères (appelée « thrombose artérielle », « thromboembolie artérielle » ou ATE).
La récupération après un caillot sanguin n'est pas toujours complète. Rarement, des effets graves peuvent persister ou, très rarement, ils peuvent être mortels.
Il est important de se rappeler que le risque global d'un caillot sanguin nocif de Zoely est faible.
COMMENT RECONNAÎTRE UN CAILLOT DE SANG
Consultez votre médecin d'urgence si vous remarquez l'un des signes ou symptômes suivants.
- douleur ou sensibilité excessive dans la jambe, qui peut survenir uniquement en position debout ou en marchant
- chaleur excessive dans la jambe affectée
- changement de couleur de la peau sur la jambe, par exemple devenant pâle, rouge ou bleu
- respiration sifflante ou respiration rapide inexpliquée d'apparition soudaine;
- toux soudaine sans cause évidente, y compris des saignements ;
- douleur piquante dans la poitrine, qui peut augmenter avec une respiration profonde;
- étourdissements graves ou vertiges;
- rythme cardiaque rapide ou irrégulier;
- douleur à l'estomac sévère;
- perte immédiate de la vision ou
- vision floue indolore, pouvant évoluer vers une perte de vision
- douleur thoracique, gêne, pression, lourdeur
- sensation de constriction ou d'oppression dans la poitrine, le bras ou sous le sternum;
- sensation de satiété, d'indigestion ou d'étouffement ;
- gêne dans le haut du corps irradiant vers le dos, la mâchoire, la gorge, le bras, l'estomac ;
- transpiration, nausées, vomissements ou vertiges;
- faiblesse extrême, anxiété ou essoufflement;
- rythme cardiaque rapide ou irrégulier
- faiblesse ou engourdissement soudain du visage, du bras ou de la jambe, en particulier d'un côté du corps ;
- confusion soudaine, difficulté à parler ou à comprendre;
- difficulté visuelle soudaine affectant un ou les deux yeux;
- difficulté soudaine à marcher, vertiges, perte d'équilibre ou de coordination;
- maux de tête soudains, sévères ou prolongés sans cause connue ;
- perte de conscience ou évanouissement avec ou sans convulsions.
- gonflement et légère décoloration bleuâtre dans une extrémité;
- douleur à l'estomac sévère (abdomen aigu);
CAILLOTS DE SANG DANS UNE VEINE
Que peut-il se passer si un caillot sanguin se forme dans une veine ?
- L'utilisation de contraceptifs hormonaux combinés a été associée à un risque accru de caillots sanguins dans les veines (thrombose veineuse), mais ces effets secondaires sont rares et surviennent le plus souvent au cours de la première année d'utilisation d'un contraceptif hormonal combiné.
- Si un caillot de sang se forme dans une veine de la jambe ou du pied, il peut provoquer une thrombose veineuse profonde (TVP).
- Si un caillot de sang se déplace de la jambe et se loge dans le poumon, il peut provoquer une "embolie pulmonaire".
- Très rarement, un caillot peut se former dans une veine d'un autre organe, comme l'œil (thrombose veineuse rétinienne).
Quand le risque de caillot sanguin dans une veine est-il le plus élevé ?
Le risque de formation d'un caillot sanguin dans une veine est le plus élevé au cours de la première année de prise d'un contraceptif hormonal combiné. Le risque peut également être plus élevé si vous reprenez un contraceptif hormonal combiné (le même produit ou une préparation différente) après une interruption de 4 semaines ou plus. Après la première année, le risque diminue, mais est toujours légèrement supérieur au risque de ne pas utiliser de contraceptif hormonal combiné.
Lorsque vous arrêtez de prendre Zoely, le risque de formation de caillots sanguins revient à des niveaux normaux en quelques semaines.
Quel est le risque de formation d'un caillot sanguin ?
Le risque dépend de votre risque naturel de TEV et du type de contraceptif hormonal combiné que vous prenez. Le risque global de développer un caillot sanguin dans la jambe ou le poumon (TVP ou EP) avec Zoely est faible.
- Sur 10 000 femmes qui n'utilisent pas de contraceptif hormonal combiné et qui ne sont pas enceintes, environ 2 développeront un caillot en un an.
- Sur 10 000 femmes qui utilisent un contraceptif hormonal combiné contenant du lévonorgestrel, de la noréthistérone ou du norgestimate, environ 5 à 7 développeront un caillot en un an.
- Le risque de caillots sanguins associé à Zoely par rapport au risque associé à un contraceptif hormonal combiné contenant du lévonorgestrel n'est pas encore connu.
- Le risque de développer un caillot de sang varie en fonction de vos antécédents médicaux personnels (voir « Facteurs qui augmentent le risque de caillot de sang » ci-dessous).
Il y a un risque de formation d'un caillot sanguin en un an
Facteurs qui augmentent le risque de caillot sanguin dans une veine
Le risque de formation d'un caillot sanguin avec Zoely est faible, mais il augmente dans certaines conditions. Le risque est plus grand :
- si vous êtes en surpoids (indice de masse corporelle ou IMC supérieur à 30 kg/m 2 ) ;
- si un membre de votre famille immédiate a eu un caillot de sang dans la jambe, le poumon ou un autre organe à un jeune âge (par exemple, avant l'âge de 50 ans environ). Si c'est le cas, vous souffrez peut-être d'un trouble de la coagulation héréditaire;
- si vous devez subir une intervention chirurgicale ou rester au lit pendant une longue période en raison d'un accident ou d'une maladie, ou si vous avez une jambe dans le plâtre. Zoely peut devoir être arrêté plusieurs semaines avant la chirurgie ou pendant que vous êtes moins mobile. Si vous devez arrêter de prendre Zoely, demandez à votre médecin quand vous pourrez recommencer à l'utiliser.
- avec l'âge (surtout après environ 35 ans);
- si elle a accouché il y a moins de quelques semaines
Le risque d'avoir un caillot sanguin augmente au fur et à mesure que ces conditions vous affectent. Les voyages en avion (plus de 4 heures) peuvent augmenter temporairement le risque de caillot sanguin, en particulier si vous présentez l'un des autres facteurs de risque énumérés.
Il est important d'informer votre médecin si l'un de ces cas s'applique à vous, même si ce n'est pas sûr. Votre médecin peut décider que l'utilisation de Zoely doit être interrompue.
Si l'une des conditions ci-dessus change pendant l'utilisation de Zoely, par exemple si un parent proche a une thrombose sans cause connue ou si vous prenez un poids considérable, veuillez en informer votre médecin.
CAILLOTS DE SANG DANS UNE ARTÈRE
Que peut-il se passer si un caillot sanguin se forme dans une « artère ? Comme un caillot sanguin dans une veine, un caillot dans une artère » peut causer de graves problèmes. Par exemple, il peut provoquer une crise cardiaque ou un accident vasculaire cérébral.
Facteurs qui augmentent le risque de caillot sanguin dans une artère
Il est important de noter que le risque de crise cardiaque ou d'accident vasculaire cérébral lié à l'utilisation de Zoely est très faible, mais il peut augmenter :
- avec l'âge (après environ 35 ans);
- si vous fumez. Lorsque vous utilisez un contraceptif hormonal combiné tel que Zoely, vous devez arrêter de fumer. Si vous ne parvenez pas à arrêter de fumer et que vous avez plus de 35 ans, votre médecin peut vous conseiller d'utiliser un autre type de contraceptif.
- si vous êtes en surpoids ;
- si vous souffrez d'hypertension artérielle;
- si un membre de votre famille immédiate a eu une crise cardiaque ou un accident vasculaire cérébral à un jeune âge (avant environ 50 ans).Si c'est le cas, vous aussi pouvez avoir un risque plus élevé d'avoir une crise cardiaque ou un accident vasculaire cérébral;
- si vous ou un proche avez des taux élevés de graisses dans le sang (cholestérol ou triglycérides) ;
- si vous souffrez de migraines, notamment de migraines avec aura ;
- si vous avez un problème cardiaque (une maladie des valves cardiaques, un trouble du rythme appelé fibrillation auriculaire)
- si vous souffrez de diabète.
Si plusieurs de ces conditions s'appliquent à vous ou si l'une d'entre elles est particulièrement grave, le risque de formation de caillots peut être encore plus grand.
Si l'une des conditions que vous signalez change pendant l'utilisation de Zoely, par exemple si vous commencez à fumer, si un parent proche développe une thrombose sans cause connue ou si vous prenez un poids considérable, veuillez en informer votre médecin.
Cancer
Le cancer du sein a été observé un peu plus fréquemment chez les utilisatrices de pilules combinées, mais on ne sait pas s'il est causé par les pilules combinées. Par exemple, plus de cancers peuvent être diagnostiqués chez les femmes utilisant des pilules combinées parce que ces femmes sont vues plus souvent par leur médecin. Après l'arrêt de la pilule combinée, le risque accru est progressivement réduit.
Il est important de contrôler régulièrement vos seins et de contacter votre médecin si vous avez des grumeaux. Informez également votre médecin si un parent proche a ou a eu un cancer du sein dans le passé (voir rubrique « Quand faut-il faire attention avec Zoely »). Dans de rares cas, des tumeurs hépatiques bénignes (non cancéreuses) et, encore plus rarement, des tumeurs hépatiques malignes (cancéreuses) ont été signalées chez des utilisatrices de pilules contraceptives. Contactez votre médecin si vous ressentez une douleur inhabituelle particulièrement sévère dans l'abdomen.
Le cancer du col de l'utérus est causé par une infection par le virus du papillome humain (VPH). Ce cancer est plus fréquent chez les femmes qui utilisent la pilule depuis longtemps. On ne sait pas s'il est dû à l'utilisation de contraceptifs hormonaux ou aux contraceptifs hormonaux. • d'autres facteurs, tels que les différences de comportement sexuel.
Tests diagnostiques
Si vous devez subir des analyses de sang ou d'urine, informez votre médecin que vous utilisez Zoely, car cela peut modifier les résultats de certains tests.
Enfants et adolescents
Aucune donnée d'efficacité et de sécurité n'est disponible chez les adolescents de moins de 18 ans.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Zoely
Informez votre médecin ou pharmacien si vous prenez, avez récemment pris ou pourriez prendre tout autre médicament, y compris les préparations en vente libre et les préparations à base de plantes. Informez également les autres médecins ou dentistes qui vous prescrivent d'autres médicaments (ou le pharmacien qui les fournit) de votre utilisation de Zoely.Ils vous diront si vous devez prendre des mesures contraceptives supplémentaires (méthodes barrières) et, si oui, pendant combien de temps.
Certains médicaments peuvent rendre Zoely moins efficace pour prévenir une grossesse ou provoquer une perte de sang inattendue.
Il s'agit notamment des médicaments utilisés pour traiter :
- épilepsie (par exemple primidone, phénytoïne, phénobarbital, carbamazépine, oxcarbazépine, topiramate, felbamate);
- tuberculose (par exemple rifampicine) ;
- Infections à VIH (par exemple ritonavir, névirapine, nelfinavir, éfavirenz) ;
- d'autres maladies infectieuses (par exemple la griséofulvine) ;
- pression élevée dans les vaisseaux sanguins des poumons (bosentan).
- Le produit à base de plantes médicinales au millepertuis peut également empêcher Zoely de fonctionner correctement. Si vous souhaitez utiliser des produits à base de plantes contenant du millepertuis alors que vous utilisez déjà Zoely, vous devez d'abord consulter votre médecin.
- Certains médicaments peuvent augmenter les taux des substances actives de Zoely dans le sang. L'efficacité de la pilule est préservée, mais informez votre médecin si vous utilisez des médicaments antifongiques contenant du kétoconazole.
- Zoely peut également interférer avec l'action d'autres médicaments, tels que l'antiépileptique lamotrigine.
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Zoely ne doit pas être utilisé par les femmes enceintes ou qui pensent l'être. Si vous tombez enceinte pendant que vous utilisez Zoely, vous devez arrêter d'utiliser Zoely et contacter votre médecin.
Si vous souhaitez arrêter de prendre Zoely parce que vous souhaitez devenir enceinte, voir rubrique « Si vous arrêtez de prendre Zoely ».
L'utilisation de Zoely est généralement déconseillée pendant l'allaitement. Si vous souhaitez utiliser la pilule pendant l'allaitement, demandez conseil à votre médecin.
Demandez conseil à votre médecin ou à votre pharmacien avant de prendre tout médicament.
Conduire et utiliser des machines
Il est peu probable que Zoely altère l'aptitude à conduire des véhicules ou à utiliser des machines.
Zoely contient du lactose Zoely contient du lactose.
Si votre médecin vous a dit que vous aviez « une intolérance à certains sucres, contactez votre médecin avant de prendre ce médicament.
Dose, mode et heure d'administration Comment utiliser Zoely : Posologie
Quand et comment prendre les comprimés
Le blister Zoely contient 28 comprimés : 24 comprimés blancs avec principes actifs (numéros 1-24) et 4 comprimés jaunes sans principes actifs (numéros 25-28).
Chaque fois que vous commencez une nouvelle bandelette de Zoely, prenez le comprimé actif blanc numéro 1 dans le coin supérieur gauche (voir « Démarrer »). Choisissez, dans la colonne grise, l'étiquette adhésive qui commence par votre jour de départ entre 7 heures du matin. étiquettes disponibles. Par exemple, si cela commence un mercredi, vous utilisez l'étiquette adhésive commençant par « WED ». ici". De cette façon, vous pouvez vérifier si vous avez pris le comprimé tous les jours.
Prendre un comprimé par jour, à peu près à la même heure, avec un peu d'eau si besoin.
Suivez le sens des flèches sur la plaquette, c'est-à-dire utilisez d'abord les comprimés actifs blancs, puis les comprimés placebo jaunes.
Vos règles commenceront au cours des 4 jours où vous utilisez les comprimés placebo jaunes (ce qu'on appelle des saignements de privation). Les saignements de privation commencent généralement 2 à 3 jours après la prise du dernier comprimé actif blanc et peuvent ne pas encore s'être terminés au début de la nouvelle plaquette.
Commencer la nouvelle bandelette immédiatement après le dernier comprimé jaune, même si vos règles ne sont pas encore terminées.Cela signifie que vous devez toujours commencer une nouvelle bandelette le même jour de la semaine, et que vos règles doivent avoir lieu tous les mois, approximativement le mêmes jours.
Chez certaines femmes, vos règles peuvent ne pas survenir tous les mois pendant que vous prenez les comprimés jaunes. Si vous avez pris Zoely tous les jours conformément aux instructions, il est peu probable que vous soyez enceinte (voir également la rubrique « Si vous n'avez pas eu de règles soit ou à plusieurs reprises").
Comment démarrer le premier pack de Zoely
Si vous n'avez pas utilisé de contraceptifs hormonaux au cours du mois précédent
Commencez à prendre Zoely le premier jour de vos règles (c'est-à-dire le premier jour de vos règles). Zoely agit immédiatement. Il n'a pas besoin d'une méthode contraceptive supplémentaire.
Si vous avez déjà utilisé un autre contraceptif hormonal combiné (pilule combinée, anneau vaginal ou dispositif transdermique)
Vous pouvez commencer à prendre Zoely le lendemain de la prise du dernier comprimé contraceptif que vous prenez actuellement (c'est-à-dire sans respecter d'intervalle).Si la plaquette contraceptive que vous prenez actuellement contient également des comprimés inactifs (placebo), vous pouvez commencer par Zoely le lendemain de la prise le dernier comprimé actif (si vous ne savez pas lequel, demandez à votre médecin ou pharmacien) Il peut également commencer plus tard, mais jamais après le lendemain de l'intervalle sans comprimé du contraceptif que vous prenez actuellement (ou le jour après le dernier comprimé inactif). Si vous utilisez un anneau vaginal ou un dispositif transdermique, vous devez de préférence commencer Zoely le jour où vous retirez l'anneau ou le dispositif. Vous pouvez également commencer au plus tard le jour où vous utiliserez le jour suivant. bague ou patch. Si vous suivez ces instructions, vous n'avez pas besoin d'une méthode contraceptive supplémentaire.
Si vous avez déjà utilisé une pilule progestative seule (minipilule)
Vous pouvez arrêter de prendre la minipilule n'importe quel jour et commencer à prendre Zoely le lendemain. Si vous avez des relations sexuelles, n'oubliez pas d'utiliser une méthode contraceptive de barrière pendant les 7 premiers jours de la prise de Zoely.
Si vous avez déjà utilisé un progestatif injectable, un implant ou un dispositif intra-utérin hormonal (SIU)
Commencez à utiliser Zoely le jour où votre prochaine injection est prévue ou le jour où l'implant ou le dispositif intra-utérin est retiré. Si vous avez des relations sexuelles, n'oubliez pas d'utiliser une méthode contraceptive de barrière pendant les 7 premiers jours de la prise de Zoely.
Après l'accouchement
Vous pouvez commencer Zoely entre 21 et 28 jours après avoir eu un bébé. Si vous commencez après le 28e jour, vous devez également utiliser une méthode contraceptive barrière au cours des 7 premiers jours d'utilisation de Zoely. Si, après avoir eu un bébé, vous avez eu des relations sexuelles avant de commencer Zoely, assurez-vous de ne pas être enceinte ou attendez vos prochaines règles. Si vous souhaitez commencer Zoely après l'accouchement et pendant l'allaitement, veuillez également lire la rubrique « Grossesse et allaitement ».Demandez conseil à votre médecin si vous ne savez pas quand commencer.
Après une « interruption de grossesse ou un avortement
Suivez les instructions de votre médecin
Si vous oubliez de prendre Zoely
Les recommandations suivantes se réfèrent exclusivement à l'oubli des comprimés actifs blancs.
- Si vous avez moins de 12 heures de retard dans la prise d'un comprimé, la fiabilité de la pilule est préservée. Prenez le comprimé dès que vous vous en rendez compte et prenez les comprimés suivants à l'heure habituelle.
- Si vous avez plus de 12 heures de retard dans la prise d'un comprimé, la fiabilité de la pilule peut être réduite. Plus vous avez oublié de comprimés consécutifs, plus le risque de diminution de l'efficacité contraceptive est grand.Le risque de grossesse est particulièrement élevé si vous oubliez les comprimés actifs blancs au début ou à la fin de la plaquette. Vous devez alors suivre les instructions ci-dessous.
Jour 1-7 de la prise des comprimés actifs blancs
Prenez le dernier comprimé blanc actif oublié dès que vous vous en souvenez (même si cela signifie prendre deux comprimés en même temps) et prenez le comprimé suivant à l'heure habituelle. Cependant, utilisez une méthode barrière comme précaution supplémentaire pendant les 7 prochains jours.
Si vous avez eu des rapports sexuels au cours de la semaine précédant l'oubli des comprimés, il est possible que vous deveniez enceinte. Contactez immédiatement votre médecin.
Jour 8-17 de la prise des comprimés actifs blancs
Prenez le dernier comprimé oublié dès que vous vous en souvenez (même si cela signifie prendre deux comprimés en même temps) et prenez les comprimés suivants à l'heure habituelle. La protection contre la grossesse n'est pas réduite et vous n'avez pas besoin de prendre des mesures de précaution supplémentaires. Cependant, si vous avez oublié plus d'un comprimé, vous devez prendre des mesures de précaution supplémentaires pendant 7 jours.
Jour 18-24 de la prise des comprimés actifs blancs
Le risque de tomber enceinte est particulièrement élevé si vous oubliez les comprimés actifs blancs à l'approche de la plage des comprimés placebo jaunes.En modifiant le programme de prise, vous pouvez prévenir ce risque.
Il a deux options :
Option 1) Prenez le dernier comprimé blanc actif oublié dès que vous vous en souvenez (même si cela signifie prendre deux comprimés en même temps) et prenez les comprimés suivants à l'heure habituelle. Commencez la plaquette suivante dès que les comprimés actifs blancs de la plaquette actuelle sont épuisés et ne prenez pas les comprimés placebo jaunes. Vos règles peuvent ne pas apparaître tant que vous n'avez pas pris les comprimés placebo jaunes à la fin de la deuxième bandelette, mais vous pouvez avoir un léger écoulement (gouttes ou taches de sang) ou des saignements pendant la prise des comprimés actifs blancs.
Option 2) Arrêtez immédiatement de prendre les comprimés actifs blancs et passez directement aux comprimés placebo jaunes dans l'intervalle. Lorsque les comprimés placebo jaunes dans l'intervalle sont terminés, vous commencez avec la bandelette suivante.
Si vous ne vous souvenez plus du nombre de comprimés actifs blancs que vous avez oubliés, suivez la première option, utilisez une méthode barrière par précaution dans les 7 jours suivants et contactez votre médecin.
Si vous avez oublié des comprimés actifs blancs d'une plaquette et que vous n'avez pas vos règles pendant que vous prenez les comprimés placebo jaunes de la même plaquette, vous pouvez être enceinte.Contactez votre médecin avant de commencer une nouvelle plaquette.
Comprimés placebo jaunes oubliés
Les 4 derniers comprimés jaunes de la quatrième rangée sont des comprimés placebo qui ne contiennent pas d'ingrédients actifs. Si vous avez oublié un de ces comprimés, la fiabilité de Zoely est préservée.Jetez le(s) comprimé(s) placebo jaune oublié(s) et prenez les comprimés suivants à l'heure habituelle.
En cas de vomissements ou de diarrhée sévère
Si vous vomissez dans les 3 à 4 heures suivant la prise d'un comprimé actif blanc ou si vous avez une diarrhée sévère, il est possible que les ingrédients actifs du comprimé Zoely n'aient pas été complètement absorbés par l'organisme. La situation est similaire à l'oubli d'un comprimé actif blanc. Après des vomissements ou une diarrhée, vous devez prendre un autre comprimé actif blanc d'une plaquette de réserve dès que possible, si possible dans les 12 heures suivant l'heure habituelle. Si cela n'est pas possible ou si 12 heures se sont déjà écoulées, suivez les instructions données dans la rubrique « Si vous oubliez de prendre Zoely ». Si vous avez une diarrhée sévère, veuillez en informer votre médecin.
Les derniers comprimés jaunes sont des comprimés placebo qui ne contiennent pas d'ingrédients actifs. Si des vomissements ou une diarrhée sévère surviennent dans les 3 à 4 heures suivant la prise d'un comprimé jaune, la fiabilité de Zoely est préservée.
Si vous souhaitez retarder vos règles
Vous pouvez retarder vos règles en ne prenant pas les comprimés placebo jaunes et en passant directement à une nouvelle plaquette de Zoely. Vous pouvez ressentir des saignements légers ou semblables à ceux des menstruations lors de l'utilisation de cette deuxième bandelette. Si vous souhaitez que vos règles commencent pendant que vous utilisez la deuxième bandelette, arrêtez de prendre les comprimés actifs blancs et commencez à prendre les comprimés placebo jaunes. Une fois que vous avez terminé les 4 comprimés placebo jaunes de la deuxième bande, commencez par la prochaine (troisième) bande.
Si vous souhaitez modifier le jour de début de vos règles
Si vous prenez les comprimés conformément aux instructions, vos règles commenceront les jours de la prise du placebo. Si vous souhaitez modifier le jour de début de votre cycle, réduisez le nombre de jours placebo, c'est-à-dire les jours où vous prenez les comprimés placebo jaunes (mais n'augmentez jamais le nombre, 4 jours est le maximum). Par exemple, si vous commencez à prendre les comprimés placebo le vendredi et que vous souhaitez commencer le mardi à la place (3 jours plus tôt), vous devez commencer une nouvelle plaquette 3 jours plus tôt que d'habitude. Vous ne pouvez pas avoir de saignement pendant cette phase raccourcie de prise de comprimés placebo jaunes. Lors de l'utilisation de la bandelette suivante, vous pouvez ressentir un léger écoulement (gouttes ou taches de sang) ou des saignements abondants lors de la prise des comprimés actifs blancs.
Si vous ne savez pas quoi faire, consultez votre médecin.
Si vous avez des saignements inattendus
Avec toutes les pilules combinées, des saignements vaginaux irréguliers (écoulements légers ou abondants) entre les menstruations peuvent survenir au cours des premiers mois. Vous devrez peut-être utiliser une serviette hygiénique, mais continuez à prendre vos comprimés normalement. Les saignements vaginaux irréguliers s'arrêtent généralement une fois que votre corps s'habitue à la pilule (généralement après 3 mois).Si les saignements continuent, augmentent en intensité ou recommencent, contactez votre médecin.
Si vous n'avez pas eu vos règles une ou plusieurs fois
Des études cliniques menées avec Zoely ont montré que les menstruations n'apparaissent parfois pas après le jour 24.
- Si vous avez pris tous les comprimés correctement, si vous n'avez pas eu de vomissements ou de diarrhée sévères et si vous n'avez pris aucun autre médicament, il est très peu probable que vous soyez enceinte. Continuez à prendre Zoely normalement. Voir également dans la rubrique « Si vous avez des vomissements ou des diarrhées sévères » ou dans la rubrique « Autres médicaments et Zoely ».
- Si vous n'avez pas pris tous les comprimés correctement ou si vos règles ne surviennent pas deux fois de suite, vous êtes peut-être tombée enceinte. Contactez immédiatement votre médecin. Ne commencez pas la prochaine bandelette Zoely tant que votre médecin n'a pas déterminé que vous n'êtes pas enceinte.
Si vous arrêtez de prendre Zoely
Vous pouvez arrêter de prendre Zoely à tout moment. Si vous ne voulez pas devenir enceinte, consultez votre médecin au sujet d'autres méthodes contraceptives.
Si vous arrêtez de prendre Zoely parce que vous souhaitez devenir enceinte, il est recommandé d'attendre l'apparition des règles naturelles avant d'essayer de concevoir. Cela facilitera la détermination de la date de livraison prévue.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin, pharmacien ou infirmier/ère.
Surdosage Que faire si vous avez pris trop de Zoely
Si vous avez pris plus de Zoely que vous n'auriez dû
Il n'y a eu aucun rapport d'effets nocifs graves de la prise d'un trop grand nombre de comprimés de Zoely à la fois. Si vous avez pris plusieurs comprimés en même temps, des nausées, des vomissements ou des saignements vaginaux peuvent survenir. Si vous remarquez qu'un enfant a pris Zoely, demandez conseil à votre médecin.
Effets secondaires Quels sont les effets secondaires de Zoely
Comme tous les médicaments, Zoely est susceptible d'avoir des effets indésirables, bien que tout le monde n'y soit pas sujet.
Si vous ressentez des effets secondaires, en particulier s'ils sont graves ou persistants, ou si vous remarquez un changement dans votre état de santé et pensez que cela peut être dû à Zoely, veuillez consulter votre médecin.
Il existe un risque accru de caillots sanguins dans les veines (thromboembolie veineuse (TEV)) ou les artères (thromboembolie artérielle (TEA)) chez toutes les femmes prenant des contraceptifs hormonaux combinés. Pour des informations plus détaillées sur les différents risques liés à la prise de contraceptifs hormonaux combinés, voir rubrique "Ce qu'il faut savoir avant d'utiliser Zoely".
Les effets secondaires suivants ont été associés à l'utilisation de Zoely :
Très fréquent (peut affecter plus de 1 personne sur 10) :
- acné
- anomalies menstruelles (par exemple menstruations absentes ou irrégulières)
Fréquent (peut affecter jusqu'à 1 personne sur 10) :
- intérêt réduit pour les activités sexuelles, dépression / humeur dépressive, sautes d'humeur
- mal de tête ou migraine
- la nausée
- menstruation abondante; douleur mammaire; douleur pelvienne (ventre)
- gain de poids
Peu fréquent (peut affecter jusqu'à 1 personne sur 100) :
- augmentation de l'appétit, rétention d'eau (œdème)
- les bouffées de chaleur
- ventre gonflé
- transpiration accrue; perte de cheveux; démanger; peau sèche; peau grasse
- lourdeur dans les membres
- menstruation régulière, mais peu abondante ; augmentation mammaire; masse mammaire; production de lait en l'absence de grossesse; syndrome prémenstruel; douleur pendant les rapports sexuels; sécheresse du vagin ou de la vulve; spasme de l'utérus
- irritabilité
- augmentation des enzymes hépatiques
Rare (peut affecter jusqu'à 1 personne sur 1 000) :
- caillots nocifs dans une veine ou une « artère, par exemple : o dans une jambe ou un pied (c. accident ischémique (AIT) ou caillots sanguins dans le foie, l'estomac/les intestins, les reins ou les yeux.
La probabilité de formation d'un caillot sanguin peut être plus élevée si vous avez d'autres conditions qui augmentent votre risque (voir la section pour plus d'informations sur les conditions qui augmentent le risque de caillots et les symptômes d'un caillot.)
- diminution de l'appétit
- intérêt accru pour les activités sexuelles
- troubles de l'attention
- yeux secs; intolérance aux lentilles de contact
- bouche sèche
- taches pigmentées jaune-brun, surtout sur le visage; croissance excessive des cheveux
- odeur vaginale; gêne dans le vagin ou la vulve
- faim
- maladie de la vésicule biliaire
Des réactions allergiques (hypersensibilité) ont été rapportées chez des utilisateurs de Zoely, mais leur fréquence ne peut être estimée à partir des données disponibles.
De plus amples informations sur les effets secondaires possibles liés à vos règles (par exemple règles absentes ou irrégulières) pendant l'utilisation de Zoely peuvent être trouvées dans les rubriques « Quand et comment prendre les comprimés », « Si vous avez des saignements inattendus » et « Si vous n'a pas eu ses règles une ou plusieurs fois".
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin, pharmacien ou infirmier/ère. Cela inclut tout effet indésirable éventuel non mentionné dans cette notice. Vous pouvez également déclarer les effets indésirables directement via le système national de déclaration (voir Annexe V). effets indésirables que vous pouvez aider fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants.
N'utilisez pas ce médicament après la date de péremption indiquée sur la plaquette et la boîte après EXP. La date de péremption fait référence au dernier jour de ce mois.
Ce médicament ne nécessite aucune condition particulière de conservation.
Les comprimés combinés (y compris les comprimés de Zoely) qui ne sont plus nécessaires ne doivent pas être jetés au tout-à-l'égout ou avec les ordures ménagères.Les excipients à activité hormonale contenus dans le comprimé peuvent avoir des effets nocifs s'ils atteignent le milieu aquatique. Rapportez les comprimés à la pharmacie ou jetez-les autrement en toute sécurité conformément à la réglementation locale. Cela contribuera à protéger l'environnement.
Ce que contient Zoely
- Les ingrédients actifs sont :
dans les comprimés pelliculés actifs blancs : chaque comprimé contient 2,5 mg d'acétate de nomégestrol et 1,5 mg d'estradiol (sous forme d'hémihydrate) ; dans les comprimés pelliculés jaunes placebo : le comprimé ne contient pas de substances actives.
- Les autres ingrédients sont :
Noyau du comprimé (comprimés pelliculés actifs blancs et comprimés pelliculés placebo jaunes) :
Lactose monohydraté (voir rubrique "Zoely contient du lactose"), cellulose microcristalline (E460), crospovidone (E1201), talc (E553b), stéarate de magnésium (E572) et silice colloïdale anhydre
Pelliculage du comprimé (comprimés pelliculés actifs blancs) :
Poly (alcool vinylique) (E1203), dioxyde de titane (E171), macrogol 3350 et talc (E553b)
Pelliculage du comprimé (comprimés pelliculés placebo jaunes) :
Poly (alcool vinylique) (E1203), dioxyde de titane (E171), macrogol 3350, talc (E553b), oxyde de fer jaune (E172) et oxyde de fer noir (E172)
Description de l'apparence de Zoely et contenu du pack
Les comprimés pelliculés actifs (comprimés) sont blancs et ronds. Des deux côtés, il y a l'inscription "ne".
Les comprimés pelliculés placebo sont jaunes et ronds. Des deux côtés, l'inscription "p" est imprimée.
Zoely est disponible en 1, 3, 6 ou 13 plaquettes thermoformées de 28 comprimés pelliculés (24 comprimés pelliculés blancs actifs et 4 comprimés pelliculés placebo jaunes) conditionnés dans une boîte.
Toutes les présentations peuvent ne pas être commercialisées
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
ZOELY 2,5 MG / 1,5 MG COMPRIMÉS ENROBÉS DE FILM
Médicament soumis à une surveillance supplémentaire. Cela permettra l'identification rapide de nouvelles informations de sécurité. Les professionnels de santé sont priés de signaler tout effet indésirable suspecté. Voir rubrique 4.8 pour plus d'informations sur la manière de déclarer les effets indésirables.
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Comprimés pelliculés actifs blancs : Chaque comprimé pelliculé contient 2,5 mg d'acétate de nomégestrol et 1,5 mg d'estradiol (sous forme d'hémihydrate).
Comprimés pelliculés placebo jaunes : le comprimé ne contient pas de principes actifs.
Excipients à effet notoire:
Chaque comprimé pelliculé actif blanc contient 57,71 mg de lactose monohydraté.
Chaque comprimé placebo pelliculé jaune contient 61,76 mg de lactose monohydraté.
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Comprimé pelliculé (comprimé).
Comprimés pelliculés actifs : blancs, ronds, avec « ne » gravé sur les deux faces.
Comprimés pelliculés placebo : jaunes, ronds, avec « p » gravé sur les deux faces.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Contraception orale.
La décision de prescrire Zoely doit prendre en compte les facteurs de risque individuels actuels de la femme, en particulier ceux liés à la thromboembolie veineuse (TEV), et le risque de TEV associé à Zoely par rapport aux autres CHC (voir rubriques 4.3 et 4.4).
04.2 Posologie et mode d'administration
Dosage
Un comprimé doit être pris chaque jour pendant 28 jours consécutifs. Chaque boîte commence par 24 comprimés actifs blancs, suivis de 4 comprimés placebo jaunes. Une fois une plaquette terminée, la plaquette suivante est démarrée immédiatement, sans interrompre la prise quotidienne des comprimés et indépendamment de la présence ou de l'absence d'hémorragie de privation.Les saignements de privation commencent généralement 2 à 3 jours après la prise du dernier comprimé blanc et peuvent ne pas être terminé encore au début du nouveau pack. Voir « Contrôle du cycle » dans la section 4.4.
Populations particulières
Insuffisance rénale
Bien qu'aucune donnée ne soit disponible chez les patients atteints d'insuffisance rénale, il est peu probable que l'insuffisance rénale affecte l'élimination de l'acétate de nomégestrol et de l'estradiol.
Insuffisance hépatique
Aucune étude clinique n'a été menée chez des patients présentant une insuffisance hépatique. Étant donné que le métabolisme des hormones stéroïdes peut être altéré chez les patients atteints d'une maladie hépatique sévère, Zoely n'est pas indiqué chez ces femmes tant que les valeurs de la fonction hépatique ne sont pas revenues à la normale (voir rubrique 4.3).
Mode d'administration
Utilisation orale.
Comment prendre Zoély
Les comprimés doivent être pris approximativement à la même heure chaque jour, indépendamment des repas. Les comprimés doivent être pris avec un peu de liquide, au besoin, dans l'ordre indiqué sur la plaquette. Des étiquettes adhésives sont apposées sur l'emballage indiquant les 7 jours de la semaine.La femme doit choisir l'étiquette qui commence par le jour où elle commence à prendre les comprimés et l'appliquer sur le blister.
Comment commencer à prendre Zoely
En l'absence de contraception hormonale antérieure (au cours du dernier mois)
Le premier comprimé doit être pris le premier jour du cycle naturel (c'est-à-dire le premier jour des règles). Dans ce cas, il n'est pas nécessaire de prendre des mesures contraceptives supplémentaires.
Passer d'un contraceptif hormonal combiné (contraceptifs oraux combinés, anneau vaginal ou dispositif transdermique)
La femme doit de préférence prendre Zoely le jour suivant le dernier comprimé actif (le dernier comprimé contenant les substances actives) de son précédent contraceptif oral combiné, au plus tard le jour suivant l'intervalle sans comprimé ou la prise des comprimés placebo de son précédent contraceptif oral. En cas d'anneau vaginal ou de dispositif transdermique, la femme doit commencer à prendre Zoely de préférence le jour du retrait du dispositif, au plus tard le jour prévu pour la prochaine application.
Passer d'une méthode progestative seule (minipilule, implant, injectable) ou d'un dispositif intra-utérin hormonal (système intra-utérin, SIU)
Le passage de la minipilule peut se produire n'importe quel jour et Zoely doit être pris le lendemain. Un implant ou un SIU peut être retiré n'importe quel jour et Zoely doit être pris le jour du retrait. Dans le cas d'un contraceptif injectable, Zoely doit être pris le jour où la prochaine injection aurait été programmée.Dans tous ces cas, il faut conseiller à la femme d'utiliser une méthode barrière supplémentaire jusqu'à ce que 7 jours de prise ininterrompue se soient écoulés. comprimés actifs.
Après un avortement au premier trimestre de la grossesse
La prise peut être commencée immédiatement.Dans ce cas, il n'est pas nécessaire d'utiliser des mesures contraceptives supplémentaires.
Après un accouchement ou un avortement au deuxième trimestre de la grossesse
Il doit être conseillé à la femme de prendre entre les jours 21 et 28 après l'accouchement ou après un avortement au cours du deuxième trimestre de la grossesse. En cas d'apparition plus tardive, il doit être conseillé à la femme d'utiliser une méthode barrière supplémentaire jusqu'à 7 jours de prise ininterrompue des comprimés actifs blancs ne se sont pas écoulés. Cependant, si des rapports sexuels ont déjà eu lieu, une grossesse doit être exclue avant de commencer l'utilisation des COC, ou la femme doit attendre le début de son premier cycle menstruel.
Pour une utilisation pendant l'allaitement, voir rubrique 4.6.
Que faire si vous oubliez un ou plusieurs comprimés
Les recommandations suivantes se réfèrent exclusivement à l'oubli des comprimés actifs blancs :
En cas de retard moins de 24 heures lors de la prise d'un comprimé actif, la protection contraceptive n'est pas réduite.La femme doit prendre le comprimé oublié dès qu'elle s'en souvient, puis prendre les comprimés suivants à l'heure habituelle.
En cas de retard plus de 24 heures lors de la prise de tout comprimé actif, la protection contraceptive peut être réduite.Le comportement à suivre en cas d'oubli repose sur deux règles fondamentales :
• 7 jours de prise ininterrompue de comprimés actifs blancs sont nécessaires pour une suppression adéquate de l'axe hypothalamo-hypophyso-ovarien.
• plus vous avez oublié de comprimés actifs blancs et plus ils sont proches des 4 comprimés placebo jaunes, plus le risque de grossesse est élevé.
Jour 1-7
La femme doit prendre le dernier comprimé oublié dès qu'elle s'en souvient, même si cela signifie prendre deux comprimés en même temps, puis elle continue à prendre les comprimés à l'heure habituelle. De plus, une méthode barrière, comme un préservatif, doit être utilisée pendant les 7 prochains jours. En cas de rapport sexuel au cours des 7 jours précédents, la possibilité d'une grossesse doit être envisagée.
Jour 8-17
La femme doit prendre le dernier comprimé oublié dès qu'elle s'en souvient, même si cela signifie prendre deux comprimés en même temps, puis elle continue à prendre les comprimés à l'heure habituelle. À condition que la femme ait pris les comprimés correctement dans les 7 jours précédant le premier comprimé oublié, il n'est pas nécessaire d'utiliser des mesures contraceptives supplémentaires. Cependant, si plus d'un comprimé a été oublié, la femme doit être informée de prendre des mesures de précaution supplémentaires pendant 7 jours.
Jour 18-24
Le risque de fiabilité réduite est imminent en raison de la proximité de la phase comprimé placebo. Cependant, en modifiant le calendrier de prise des comprimés, une réduction de la protection contraceptive peut toujours être évitée. En adhérant à l'une des deux options suivantes, il n'est donc pas nécessaire de prendre des mesures contraceptives supplémentaires, à condition que la femme ait pris tous les comprimés correctement dans les 7 jours précédant le premier comprimé oublié. Sinon, vous devez vous en tenir à la première des deux options et prendre des mesures de précaution supplémentaires au cours des 7 prochains jours.
1. La femme doit prendre le dernier comprimé oublié dès qu'elle s'en souvient, même si cela signifie prendre deux comprimés en même temps. Elle continue ensuite à prendre les comprimés à l'heure habituelle, jusqu'à ce que les comprimés actifs soient terminés. Les 4 comprimés placebo de la dernière rangée doivent être jetés. La plaquette suivante doit être commencée immédiatement. Les saignements de privation sont peu probables avant la fin des comprimés actifs de la deuxième plaquette, mais des spottings peuvent survenir lors de la prise des comprimés ou des métrorragies.
2. On peut également demander à la femme d'arrêter de prendre les comprimés actifs de la plaquette actuelle. Dans ce cas, elle doit prendre les comprimés placebo de la dernière rangée pendant un maximum de 4 jours, y compris les jours où elle a oublié les comprimés. , puis continuez avec le prochain blister.
Si la femme a oublié de prendre quelques comprimés et par conséquent pas d'hémorragie de privation pendant la phase placebo, la possibilité d'une grossesse doit être envisagée.
Comprimés placebo jaunes oubliés
La protection contraceptive n'est pas réduite. Les comprimés jaunes de la dernière (4e) rangée de la plaquette peuvent être ignorés.Cependant, les comprimés oubliés doivent être jetés pour éviter de prolonger par inadvertance la phase placebo.
Mises en garde en cas de troubles gastro-intestinaux
En cas de troubles gastro-intestinaux sévères (par exemple vomissements ou diarrhée), l'absorption des substances actives peut être incomplète et des mesures contraceptives supplémentaires doivent être utilisées.
Si des vomissements surviennent dans les 3 à 4 heures suivant la prise d'un comprimé blanc, le comprimé doit être considéré comme perdu et un nouveau comprimé doit être pris dès que possible. Si possible, le nouveau comprimé doit être pris dans les 24 heures suivant la prise. habituel. Le prochain comprimé doit alors être pris à l'heure habituelle.Si plus de 24 heures se sont écoulées depuis la dernière prise d'un comprimé, les instructions sur l'oubli des comprimés de la rubrique 4.2 « Que faire en cas d'oubli d'un ou plusieurs comprimés » s'appliquent. prenez généralement le comprimé ou des comprimés blancs supplémentaires d'une autre boîte.
Comment décaler ou retarder les menstruations
Pour retarder les règles, la femme doit continuer avec une autre plaquette thermoformée de Zoely sans prendre les comprimés placebo jaunes de la plaquette actuelle. Le cycle peut être prolongé à volonté, jusqu'à ce que les comprimés actifs blancs de la deuxième plaquette soient terminés. Zoely régulier reprendra après les comprimés placebo jaunes de la deuxième boîte ont été pris. La femme en phase de prolongation du cycle peut présenter des saignements intermenstruels ou des spottings.
Afin de décaler ses règles à un autre jour de la semaine que dans le schéma posologique actuel, la femme peut raccourcir la phase des comprimés placebo jaunes jusqu'à 4 jours. Plus l'intervalle est court, plus le risque de saignement de privation ne se produira pas et que des saignements et des spottings se produiront lors de la prise de la plaquette suivante (comme lorsque vous retardez vos règles).
04.3 Contre-indications
Les contraceptifs hormonaux combinés (COC) ne doivent pas être utilisés dans les conditions suivantes. Les données épidémiologiques concernant les COC contenant du 17β-estradiol n'étant pas encore disponibles, les contre-indications des COC contenant de l'éthinylestradiol sont également considérées comme valables pour Zoely. Si l'une des conditions suivantes survient pour la première fois lors de l'utilisation de Zoely, la prise du médicament doit être immédiatement arrêtée.
• présence ou risque de thromboembolie veineuse (TEV)
- Thromboembolie veineuse "." TEV en cours (sous traitement anticoagulant) ou antécédents (p. ex. thrombose veineuse profonde [TVP] ou embolie pulmonaire [EP]).
- Prédisposition héréditaire ou acquise connue à la thromboembolie veineuse, telle que résistance aux APC (dont facteur V Leiden), déficit en antithrombine III, déficit en protéine C, déficit en protéine S.
- Chirurgie majeure avec immobilisation prolongée (voir rubrique 4.4).
- Risque élevé de thromboembolie veineuse en raison de la présence de multiples facteurs de risque (voir rubrique 4.4).
• présence ou risque de thromboembolie artérielle (TEA)
- Thromboembolie artérielle "." Thromboembolie artérielle actuelle, antécédents de thromboembolie artérielle (par exemple, infarctus du myocarde) ou état prodromique (par exemple, angine de poitrine).
- Maladie cérébrovasculaire "." accident vasculaire cérébral actuel, antécédents d'accident vasculaire cérébral ou état prodromique (par exemple, accident ischémique transitoire [accident ischémique transitoire, TIA]).
- Prédisposition héréditaire ou acquise connue à la thromboembolie artérielle, telle que l'hyperhomocystéinémie et les anticorps antiphospholipides (anticorps anticardiolipine, anticoagulant lupique).
- Antécédents de migraine avec symptômes neurologiques focaux.
- Risque élevé de thromboembolie artérielle dû à de multiples facteurs de risque (voir rubrique 4.4) ou à la présence d'un seul facteur de risque grave tel que :
• diabète sucré avec symptômes vasculaires
• hypertension sévère
• dyslipoprotéinémie sévère.
• pancréatite ou antécédents de pancréatite en cas d'hypertriglycéridémie sévère.
• présence ou antécédents de maladie hépatique sévère jusqu'à ce que les valeurs de la fonction hépatique reviennent à la normale.
• présence ou antécédents de tumeurs hépatiques (bénignes ou malignes).
• Des tumeurs malignes sexuellement dépendantes des stéroïdes connues ou suspectées (p. ex. cancers des organes génitaux ou du sein).
• saignements vaginaux non diagnostiqués.
• Hypersensibilité aux substances actives ou à l'un des excipients mentionnés à la rubrique 6.1.
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Mises en garde
En présence de l'une des conditions ou des facteurs de risque mentionnés ci-dessous, l'adéquation de Zoely doit être discutée avec la femme.
En cas d'aggravation ou d'apparition de l'une des affections ou facteurs de risque mentionnés, il doit être conseillé à la femme de contacter son médecin afin de déterminer si l'utilisation de Zoely doit être interrompue.Toutes les données ci-dessous sont basées sur les données épidémiologiques obtenues avec les COC contenant de l'éthinylestradiol Zoely contient du 17β-estradiol Comme les données épidémiologiques ne sont pas encore disponibles concernant les COC contenant de l'estradiol, les avertissements sont également considérés comme valables pour Zoely.
Risque de thromboembolie veineuse (TEV)
• L'utilisation de tout contraceptif hormonal combiné (COC) augmente le risque de thromboembolie veineuse (TEV) par rapport à l'absence d'utilisation. Les préparations contenant du lévonorgestrel, du norgestimate ou de la noréthistérone sont associées au plus faible risque de TEV. Il n'est pas encore connu. ampleur du risque associé à Zoely par rapport à ces préparations à faible risque. La décision d'utiliser une préparation autre que celles connues pour être associées au plus faible risque de TEV ne doit être prise qu'après un entretien avec la femme, afin de s'assurer qu'elle comprend le risque de TEV associé aux CHC, ainsi que ses facteurs de risque actuels .influencer ce risque et que le risque de TEV est le plus élevé au cours de la première année de première utilisation. De plus, certaines preuves suggèrent que le risque est plus grand si l'utilisation d'un COC est reprise après une pause de 4 semaines ou plus.
• Parmi les femmes qui n'utilisent pas de CHC et qui ne sont pas enceintes, environ 2 sur 10 000 développent une MTEV dans l'année.Cependant, chaque femme peut courir un risque beaucoup plus élevé en fonction de ses facteurs de risque préexistants (voir ci-dessous) .
• Études épidémiologiques chez les femmes utilisant des contraceptifs hormonaux combinés à faible dose (
• Sur 10 000 femmes qui utilisent un CHC contenant du lévonorgestrel, on pense qu'environ 6 développeront une TEV dans l'année.
• L'étendue du risque de TEV associé aux COC contenant de l'acétate de nomégestrol en association avec l'estradiol par rapport au risque associé aux COC contenant de faibles doses de lévonorgestrel n'est pas encore connue.
• Le nombre de TEV par an avec les CHC à faible dose est inférieur au nombre attendu chez les femmes enceintes ou en post-partum.
• La TEV peut être mortelle dans 1 à 2 % des cas.
• Dans des cas extrêmement rares, thrombose affectant d'autres vaisseaux sanguins, par ex. veines ou artères hépatiques, mésentériques, rénales ou rétiniennes.
Facteurs de risque de TEV
Le risque de complications thromboemboliques veineuses chez les utilisatrices de COC peut augmenter considérablement chez une femme présentant des facteurs de risque supplémentaires, en particulier si les facteurs de risque sont multiples (voir tableau).
Zoely est contre-indiqué si la femme présente de multiples facteurs de risque conduisant à un risque élevé de thrombose veineuse (voir rubrique 4.3). Si une femme présente plus d'un facteur de risque, il est possible que le risque accru soit supérieur à la somme des facteurs individuels, auquel cas son risque global de TEV doit être pris en compte. Une CHC doit être prescrite (voir rubrique 4.3).
Tableau : Facteurs de risque de TEV
• Il n'y a pas de consensus sur le rôle possible des varices et de la thrombophlébite superficielle dans l'apparition ou la progression de la thrombose veineuse.
• Le risque accru de thromboembolie pendant la grossesse et surtout au cours des 6 semaines de puerpéralité doit être pris en compte (pour des informations sur « Grossesse et allaitement », voir rubrique 4.6).
Symptômes de TEV (thrombose veineuse profonde et embolie pulmonaire)
Si des symptômes apparaissent, les femmes doivent être informées de la nécessité de consulter un médecin d'urgence et d'informer le professionnel de la santé de l'utilisation d'un COC.
Les symptômes de la thrombose veineuse profonde (TVP) peuvent inclure :
• gonflement unilatéral de la jambe et/ou du pied ou le long d'une veine de la jambe ;
• douleur ou sensibilité excessive dans la jambe, qui ne peut survenir qu'en position debout ou en marchant ;
• chaleur excessive dans la jambe affectée; rougeur ou décoloration de la peau de la jambe.
Les symptômes de l'embolie pulmonaire (EP) peuvent inclure :
• respiration sifflante inexpliquée ou respiration rapide d'apparition soudaine;
• toux soudaine, qui peut être associée à une hémoptysie ;
• douleur thoracique piquante;
• sensation intense de vertiges ou de vertiges ;
• rythme cardiaque rapide ou irrégulier.
Certains symptômes (par exemple « respiration sifflante », « toux ») ne sont pas spécifiques et peuvent être interprétés à tort comme des événements plus fréquents ou moins graves (par exemple, les infections des voies respiratoires).
D'autres signes d'occlusion vasculaire peuvent inclure : une douleur soudaine, un gonflement et une légère décoloration bleuâtre d'une "extrémité".
Si l'occlusion affecte l'œil, les symptômes peuvent inclure une vision floue indolore, qui peut évoluer vers une perte de vision. Parfois, la perte de vision peut survenir presque immédiatement.
Risque de thromboembolie artérielle (TEA)
Des études épidémiologiques ont associé l'utilisation des CHC à un risque accru de thromboembolie artérielle (infarctus du myocarde) ou d'accident vasculaire cérébral (par exemple, accident ischémique transitoire, accident vasculaire cérébral).Les événements thromboemboliques artériels peuvent être mortels.
Facteurs de risque de l'ETA
Le risque de complications thromboemboliques artérielles ou d'accident vasculaire cérébral chez les utilisatrices de COC augmente chez les femmes présentant des facteurs de risque (voir tableau). Zoely est contre-indiqué si une femme présente un seul facteur de risque grave ou plusieurs facteurs de risque d'EI qui comportent un risque élevé de thrombose artérielle (voir rubrique 4.3). Si une femme présente plus d'un facteur de risque, il est possible que l'augmentation du risque soit supérieure à la somme des facteurs individuels, auquel cas son risque global doit être pris en compte. doit être prescrit (voir rubrique 4.3).
Tableau : Facteurs de risque de l'ETA
Symptômes de l'ETA
Si des symptômes apparaissent, les femmes doivent être informées de la nécessité de consulter un médecin d'urgence et d'informer le professionnel de la santé de l'utilisation d'un COC.
Les symptômes d'un accident vasculaire cérébral peuvent inclure:
• engourdissement ou faiblesse soudaine du visage, du bras ou de la jambe, surtout d'un côté du corps ;
• difficulté soudaine à marcher, vertiges, perte d'équilibre ou de coordination;
• confusion soudaine ou difficulté à parler ou à comprendre;
• difficulté visuelle soudaine affectant un ou les deux yeux ;
• maux de tête soudains, sévères ou prolongés sans cause connue;
• perte de conscience ou évanouissement avec ou sans crise.
Des symptômes temporaires suggèrent que l'événement est un accident ischémique transitoire (accident ischémique transitoire, TIA).
Les symptômes de l'infarctus du myocarde (IM) peuvent inclure :
• douleur, inconfort, pression, lourdeur, sensation de constriction ou d'oppression dans la poitrine, le bras ou sous le sternum ;
• gêne irradiant vers le dos, la mâchoire, la gorge, le bras, le ventre ;
• sensation de satiété, d'indigestion ou d'étouffement ;
- sueurs, nausées, vomissements ou vertiges ;
• faiblesse extrême, anxiété ou essoufflement;
• rythme cardiaque rapide ou irrégulier.
Tumeurs
• Un risque accru de cancer du col de l'utérus chez les femmes utilisant des COC pendant des périodes prolongées (> 5 ans) a été rapporté dans certaines études épidémiologiques, mais il est encore controversé dans quelle mesure ces données sont attribuables à d'autres facteurs, tels que. comportement sexuel et le virus du papillome humain (VPH). Il n'existe pas de données épidémiologiques sur le risque de cancer du col de l'utérus chez les utilisatrices de Zoely.
• Avec l'utilisation de COC à dose plus élevée (50 mcg d'éthinylestradiol), le risque de cancer de l'endomètre et de l'ovaire est réduit. Il faut confirmer si cela s'applique également aux COC contenant du 17β-estradiol.
• Une méta-analyse de 54 études épidémiologiques a montré qu'il existe un risque relatif légèrement accru (RR = 1,24) de diagnostic de cancer du sein chez les femmes utilisant actuellement des COC. Le risque supplémentaire disparaît progressivement sur 10 ans après l'arrêt de l'utilisation des COC. Le cancer du sein étant rare chez les femmes de moins de 40 ans, le nombre plus élevé de diagnostics de cancer du sein chez les utilisatrices actuelles et récentes de COC est faible par rapport au risque global de cancer du sein. à un stade cliniquement moins avancé du cancer diagnostiqué chez les femmes n'ayant jamais utilisé de COC. L'augmentation observée du risque peut être due à un diagnostic plus précoce du cancer. chez les utilisatrices de COC, aux effets biologiques des COC ou à une combinaison de ces deux facteurs .
• Dans de rares cas, des tumeurs hépatiques bénignes et, encore plus rarement, des tumeurs hépatiques malignes ont été signalées chez les utilisatrices de COC. Dans des cas isolés, ces tumeurs ont provoqué des hémorragies intra-abdominales menaçant le pronostic vital. Le cancer hépatique doit donc être envisagé dans le diagnostic différentiel en présence de douleurs abdominales hautes sévères, d'hypertrophie hépatique ou de signes d'hémorragie intra-abdominale chez les utilisatrices de COC.
D'autres conditions
• Les femmes présentant une hypertriglycéridémie ou des antécédents familiaux d'hypertriglycéridémie peuvent présenter un risque accru de pancréatite lorsqu'elles utilisent des COC.
• Bien que de légères augmentations de la pression artérielle aient été observées chez de nombreux utilisateurs de COC, les augmentations cliniquement pertinentes sont rares. Une corrélation entre l'utilisation de COC et l'hypertension clinique n'a pas été établie. Cependant, si vous développez une hypertension cliniquement significative et durable lors de l'utilisation d'un COC, il est prudent que votre médecin arrête de prendre les comprimés et traite l'hypertension. Le cas échéant, l'utilisation des COC peut être reprise si des valeurs de pression artérielle normales peuvent être atteintes avec un traitement antihypertenseur.
• L'apparition ou l'aggravation des affections suivantes a été signalée à la fois pendant la grossesse et pendant l'utilisation de COC, mais la preuve d'une corrélation avec l'utilisation de COC n'est pas concluante : ictère et/ou démangeaisons liées à la cholestase ; formation de calculs biliaires ; porphyrie ; systémique lupus érythémateux ; syndrome urémique-hémolytique ; chorée de Sydenham ; herpès gestationnel ; surdité liée à l'otosclérose.
• Chez les femmes atteintes d'œdème de Quincke héréditaire, les œstrogènes exogènes peuvent induire ou aggraver les symptômes de l'œdème de Quincke.
• La présence d'un dysfonctionnement hépatique aigu ou chronique peut nécessiter l'arrêt de l'utilisation des COC jusqu'à ce que les marqueurs de la fonction hépatique reviennent à la normale. Une récidive d'ictère cholestatique survenue pour la première fois pendant la grossesse ou l'utilisation antérieure de stéroïdes sexuels rend nécessaire l'arrêt de l'utilisation des contraceptifs oraux combinés.
• Bien que les COC puissent avoir un effet sur la résistance périphérique à l'insuline et la tolérance au glucose, rien n'indique qu'il soit nécessaire de modifier le schéma thérapeutique chez les femmes diabétiques utilisant des contraceptifs oraux combinés à faible dose (contenant
• L'aggravation de la dépression, la maladie de Crohn et la colite ulcéreuse ont été associées à l'utilisation des COC.
• Un chloasma peut occasionnellement survenir, en particulier chez les femmes ayant des antécédents de chloasma gravidarum. Les femmes ayant une tendance au chloasma doivent éviter l'exposition au soleil ou aux rayons ultraviolets lorsqu'elles utilisent des COC.
• Les patients présentant des problèmes héréditaires rares d'intolérance au galactose, de déficit en lactase de Lapp ou de malabsorption du glucose et du galactose ne doivent pas prendre ce médicament.
Examen médical / consultation
Avant de commencer ou de reprendre l'utilisation de Zoely, des antécédents médicaux complets (y compris les antécédents familiaux) doivent être recueillis et une grossesse exclue. La pression artérielle doit être mesurée et un examen physique doit être effectué en tenant compte des contre-indications (voir rubrique 4.3) et des mises en garde (voir rubrique 4.4) Il est important d'attirer l'attention d'une femme sur les informations concernant la thrombose veineuse et artérielle, notamment le risque associé à Zoely par rapport au risque associé aux autres CHC, les symptômes de la MTEV et de l'ETA, les facteurs de risque connus et les mesures à prendre en cas de suspicion de thrombose.
La femme doit également être invitée à lire attentivement la notice et à suivre les instructions. La fréquence et la nature des examens doivent être basées sur des directives standard et adaptées aux besoins individuels de chaque femme.
Les femmes doivent être informées que les contraceptifs hormonaux ne protègent pas contre l'infection par le VIH (SIDA) et d'autres maladies sexuellement transmissibles.
Efficacité réduite
L'efficacité des COC peut être réduite, par exemple, en cas d'oubli de comprimés (voir rubrique 4.2), de troubles gastro-intestinaux lors de la prise de principes actifs (voir rubrique 4.2) ou de traitements concomitants avec d'autres médicaments (voir rubrique 4.5).
Contrôle du cycle
Avec tous les COC, des saignements irréguliers (spotting ou saignements intermenstruels) peuvent survenir, en particulier au cours des premiers mois d'utilisation. Par conséquent, l'évaluation de toute perte sanguine irrégulière n'a de sens qu'après un intervalle d'adaptation d'environ 3 cycles. Le pourcentage d'utilisatrices de Zoely ayant subi des saignements menstruels après cette période d'adaptation était de 15 à 20 %.
Si des irrégularités persistent ou surviennent après des cycles précédemment réguliers, des causes non hormonales doivent être envisagées et des procédures de diagnostic adéquates sont indiquées pour exclure la présence d'une tumeur maligne ou d'une grossesse. Un curetage peut être nécessaire.
Chez les utilisateurs de Zoely, la durée des saignements de retrait est en moyenne de 3 à 4 jours. Chez les utilisatrices de Zoely, il est également possible que l'hémorragie de privation ne se produise pas, même si elles ne sont pas enceintes. Dans les études cliniques, l'hémorragie de privation était absente dans les cycles 1 à 12 dans 18 à 32 % des cas. Dans de tels cas, l'absence d'hémorragie de privation n'a pas été associée à une fréquence accrue d'hémorragies de privation / spotting dans les cycles suivants.4,6 % des femmes n'ont pas eu d'hémorragie de privation au cours des trois premiers cycles d'utilisation, et dans ce sous-groupe, l'absence d'hémorragie de privation au cours des cycles suivants était fréquent, allant de 76 % à 87 % des femmes. 28 % des femmes n'ont pas présenté d'hémorragie de privation dans au moins un des cycles 2, 3 et 4, associée à une « absence d'hémorragie de privation plus fréquente dans les cycles suivants , allant de 51% à 62%."
Si l'hémorragie de privation est absente et que Zoely a été pris conformément aux instructions décrites à la rubrique 4.2, il est peu probable que la femme soit enceinte. Cependant, une grossesse doit être exclue avant de poursuivre l'utilisation de Zoely si Zoely n'a pas été pris comme prescrit ou s'il n'y a pas deux saignements de privation consécutifs.
Population pédiatrique
On ne sait pas si la quantité d'estradiol contenue dans Zoely est suffisante pour maintenir des taux d'estradiol adéquats chez les adolescents, en particulier en ce qui concerne l'acquisition de la masse osseuse (voir rubrique 5.2).
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Interactions
Interactions d'autres médicaments avec Zoely
Les interactions entre les contraceptifs oraux et les médicaments inducteurs enzymatiques peuvent entraîner des saignements et même un échec de la contraception.
Des exemples de substances actives qui induisent des enzymes hépatiques et augmentent ainsi la clairance des hormones sexuelles comprennent : la phénytoïne, le phénobarbital, la primidone, le bosentan, la carbamazépine, la rifampicine et les préparations médicinales ou à base de plantes contenant du millepertuis et, dans une moindre mesure, de l'oxcarbazépine, du topiramate, felbamato et griséofulvine. Les inhibiteurs de la protéase du VIH qui ont un potentiel d'induction (par exemple, le ritonavir et le nelfinavir) et les inhibiteurs non nucléosidiques de la transcriptase inverse (par exemple, la névirapine et l'éfavirenz) peuvent également affecter le métabolisme hépatique.
Avec les substances inductrices d'enzymes hépatiques, une méthode barrière doit être utilisée pendant la co-administration de médicaments et pendant 28 jours après leur arrêt. En cas de traitement prolongé par des substances inductrices d'enzymes hépatiques, une autre méthode de contraception doit être envisagée.
Aucune étude d'interaction n'a été réalisée avec Zoely, mais deux études avec la rifampicine et le kétoconazole, respectivement, ont été réalisées avec une association à forte dose d'acétate de nomégestrol et d'estradiol (acétate de nomégestrol 3,75 mg + 1,5 mg d'estradiol) chez des femmes en-ménopause. L'utilisation concomitante de rifampicine réduit l'ASC0-∞ de l'acétate de nomégestrol de 95 % et augmente l'ASC0-tlast de l'estradiol de 25 %. la pertinence de la concentration maximale (85 %) et de l'ASC0-∞ (115 %) de l'acétate de nomégestrol a été observée. Des conclusions similaires sont attendues chez les femmes en âge de procréer.
Effets de Zoely sur d'autres médicaments
Les contraceptifs oraux peuvent modifier le métabolisme d'autres médicaments. Une attention particulière doit être portée à l'interaction avec la lamotrigine.
Tests diagnostiques
L'utilisation de stéroïdes contraceptifs peut affecter les résultats de certains tests de diagnostic, y compris les paramètres biochimiques de la fonction hépatique, thyroïdienne, surrénale et rénale, les taux plasmatiques de protéines (porteuses), par exemple de globuline qui se lie aux corticostéroïdes et les fractions de paramètres lipidiques/lipoprotéiques, glucides paramètres du métabolisme et paramètres de coagulation et de fibrinolyse Les variations se situent généralement dans les plages normales de laboratoire.
04.6 Grossesse et allaitement
Grossesse
Zoely n'est pas indiqué pendant la grossesse.
Si une grossesse survient pendant la prise de Zoely, toute prise supplémentaire doit être arrêtée. La plupart des études épidémiologiques n'ont révélé ni un risque accru de malformations congénitales chez les enfants de femmes ayant utilisé des contraceptifs oraux combinés contenant de l'éthinylœstradiol avant la grossesse, ni un effet tératogène en cas d'utilisation accidentelle de contraceptifs oraux combinés contenant de l'éthinylœstradiol en début de grossesse.
Les données cliniques sur un nombre limité de grossesses exposées n'indiquent aucun effet indésirable de Zoely sur le fœtus ou le nouveau-né.
Dans les études animales, une toxicité pour la reproduction a été observée avec l'association nomégestrol acétate / estradiol (voir les données de sécurité préclinique à la rubrique 5.3).
Le risque accru de TEV pendant la période post-partum doit être pris en compte lors de la reprise de l'utilisation de Zoely (voir rubriques 4.2 et 4.4).
L'heure du repas
De petites quantités de stéroïdes contraceptifs et/ou de leurs métabolites peuvent être excrétées dans le lait maternel, mais il n'y a aucune preuve d'effets indésirables sur la santé du bébé.
L'allaitement peut être influencé par les COC, car ils peuvent réduire la quantité et modifier la composition du lait maternel. Par conséquent, l'utilisation des COC ne doit pas être recommandée jusqu'au sevrage complet et pour les femmes souhaitant allaiter, il convient de proposer une méthode alternative de contraception.
La fertilité
Zoely est indiqué pour la prévention de la grossesse. Pour plus d'informations sur la restauration de la fertilité, voir rubrique 5.1.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Aucune étude sur l'aptitude à conduire des véhicules ou à utiliser des machines n'a été réalisée avec Zoely. Cependant, aucun effet sur l'aptitude à conduire des véhicules et à utiliser des machines n'a été observé chez les utilisateurs de COC.
04.8 Effets indésirables
Résumé du profil de sécurité
Six essais cliniques multicentriques d'une durée maximale d'un an ont été menés pour évaluer l'innocuité de Zoely. Au total, 3 434 femmes âgées de 18 à 50 ans ont été inscrites et ont terminé 33 828 cycles.
Description des effets indésirables sélectionnés
Un risque accru d'événements thrombotiques et thromboemboliques artériels et veineux, notamment d'infarctus du myocarde, d'accident vasculaire cérébral, d'accidents ischémiques transitoires, de thrombose veineuse et d'embolie pulmonaire, a été observé chez les utilisateurs de COC ; de tels événements sont discutés plus en détail dans la section 4.4.
Tableau récapitulatif des effets indésirables
Effets indésirables éventuellement liés qui ont été observés avec Zoely dans les études cliniques ou pendant l'utilisation post-commercialisation sont répertoriés dans le tableau suivant.
Tous les effets indésirables sont répertoriés par classe de système d'organes et fréquence ; très fréquent (≥ 1/10), fréquent (≥ 1/100,
1 Le terme MedDRA qui décrit le mieux un effet indésirable donné est répertorié dans la « liste. Les synonymes ou les affections apparentées n'apparaissent pas dans la » liste, mais doivent être pris en compte.
En plus des effets indésirables listés ci-dessus, des réactions d'hypersensibilité ont été rapportées chez les utilisateurs de Zoely (fréquence indéterminée).
Déclaration des effets indésirables suspectés
La déclaration des effets indésirables suspectés survenant après l'autorisation du médicament est importante, car elle permet une surveillance continue du rapport bénéfice/risque du médicament.Les professionnels de santé sont invités à déclarer tout effet indésirable via l'Agence italienne du médicament.
Site Internet:
http://www.agenziafarmaco.gov.it/it/responsabili
04.9 Surdosage
Des doses maximales multiples de cinq fois la dose quotidienne de Zoely et des doses uniques maximales de 40 fois la dose quotidienne d'acétate de nomégestrol seule ont été utilisées chez des femmes sans aucun problème de sécurité. Sur la base de l'expérience globale avec les COC, les symptômes qui peuvent survenir sont des nausées, des vomissements et, chez les filles, une légère perte de sang vaginal. Il n'y a pas d'antidote et le traitement doit être symptomatique.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : hormones sexuelles et modulateurs de l'appareil génital, progestatifs et œstrogènes, associations fixes, code ATC : G03AA14.
L'acétate de nomégestrol est un progestatif hautement sélectif, dérivé de la progestérone, une hormone stéroïde naturelle. L'acétate de nomégestrol présente « une affinité élevée pour le récepteur de la progestérone humaine et possède » une activité anti-gonadotrope, « une activité anti-œstrogénique médiée par les récepteurs de la progestérone, une activité anti-androgène modérée et est dépourvue de toute activité œstrogénique », androgène, glucocorticoïde ou minéralocorticoïde.
L'œstrogène contenu dans Zoely est le 17β-estradiol, un œstrogène naturel identique au 17β-estradiol humain endogène.
L'effet contraceptif de Zoely repose sur l'interaction de divers facteurs, dont les plus importants sont considérés comme l'inhibition de l'ovulation et les modifications de la sécrétion cervicale.
Dans deux études randomisées, ouvertes et comparatives d'efficacité et d'innocuité, plus de 3 200 femmes ont été traitées pendant jusqu'à 13 cycles consécutifs avec Zoely et plus de 1 000 femmes avec la drospirénone 3 mg "." éthinylestradiol 30 mcg (schéma 21/7).
De l'acné dans le groupe Zoely a été rapportée chez 15,4% des femmes (vs 7,9% dans le groupe de comparaison), une prise de poids chez 8,6% des femmes (vs.5,7 % dans le groupe de comparaison) et des saignements de privation anormaux (principalement absence de saignement de privation ) chez 10,5 % des femmes (contre 0,5 % dans le groupe comparateur).
Dans l'étude clinique menée avec Zoely dans l'Union européenne, les indices de Pearl suivants ont été déterminés pour la tranche d'âge entre 18 et 35 ans :
Échec de la méthode : 0,40 (limite supérieure de l'intervalle de confiance à 95 % 1,03)
Échec de la méthode et erreur de l'utilisateur : 0,38 (limite supérieure intervalle de confiance à 95 % 0,97)
Dans l'étude clinique menée avec Zoely aux États-Unis, les indices de Pearl suivants ont été déterminés pour la tranche d'âge entre 18 et 35 ans :
Échec de la méthode : 1,22 (limite supérieure de l'intervalle de confiance à 95 % 2,18)
Échec de la méthode et erreur de l'utilisateur : 1,16 (limite supérieure de l'intervalle de confiance à 95 % 2,08)
Dans une étude randomisée en ouvert, 32 femmes ont été traitées pendant 6 cycles avec Zoely.
Après l'arrêt de Zoely, une reprise de l'ovulation a été observée chez 79 % des femmes dans les 28 jours suivant la prise du dernier comprimé.
Dans une étude clinique, l'histologie de l'endomètre a été évaluée dans un sous-groupe de femmes (n = 32) après 13 cycles de traitement. Aucun résultat anormal n'a été trouvé.
Population pédiatrique
Aucune donnée d'efficacité et de sécurité n'est disponible chez les adolescents de moins de 18 ans Les données pharmacocinétiques disponibles sont décrites dans la rubrique 5.2.
05.2 Propriétés pharmacocinétiques
Acétate de nomégestrol
Absorption
L'acétate de nomégestrol administré par voie orale est rapidement absorbé.
Les concentrations plasmatiques maximales d'acétate de nomégestrol, d'environ 7 ng/ml, sont atteintes 2 heures après une administration unique. La biodisponibilité absolue de l'acétate de nomégestrol après une dose unique est de 63 %. Aucun effet cliniquement pertinent n'a été observé avec la prise alimentaire sur la biodisponibilité de l'acétate de nomégestrol.
Distribution
L'acétate de nomégestrol est fortement lié à l'albumine (97-98 %), mais ne se lie pas à la globuline liant les hormones sexuelles (Globuline de liaison aux hormones sexuelles, SHBG) ou la globuline liant les corticoïdes (Globuline de liaison aux corticoïdes, CBG). Le volume apparent de distribution de l'acétate de nomégestrol à l'état d'équilibre est de 1 645 ± 576 l.
Biotransformation
L'acétate de nomégestrol est métabolisé en de nombreux métabolites hydroxylés inactifs par les enzymes hépatiques du cytochrome P450, principalement le CYP3A4 et le CYP3A5, avec des contributions possibles du CYP2C19 et du CYP2C8. L'acétate de nomégestrol et ses métabolites hydroxylés subissent un important métabolisme de phase 2, avec formation de conjugués glucuronide et sulfate. La clairance apparente à l'état d'équilibre est de 26 l/h.
Élimination
La demi-vie d'élimination (t & 1frac12;) est de 46 h (intervalle 28-83 h) à l'état d'équilibre. La demi-vie d'élimination des métabolites n'a pas été déterminée.
L'acétate de nomégestrol est excrété dans les urines et les fèces. Environ 80 % de la dose est excrétée dans les urines et les fèces dans les 4 jours.L'excrétion de l'acétate de nomégestrol était presque complète après 10 jours et les quantités excrétées étaient plus élevées dans les fèces que dans les urines.
Linéarité
Une linéarité de la dose a été observée dans la plage de 0,625 à 5 mg (évaluée chez les femmes fertiles et ménopausées).
Conditions d'état stable
La pharmacocinétique de l'acétate de nomégestrol n'est pas affectée par la SHBG.
L'état d'équilibre est atteint au bout de 5 jours. Les concentrations plasmatiques maximales d'acétate de nomégestrol, environ 12 ng/ml, sont atteintes 1,5 h après l'administration. Les concentrations plasmatiques moyennes à l'état d'équilibre sont de 4 ng/mL.
Interactions médicamenteuses
In vitro, l'acétate de nomégestrol ne provoque pas d'induction ou d'inhibition significative des enzymes du cytochrome P450 et n'a aucune interaction cliniquement pertinente avec la protéine de transport P-gp.
Estradiol
Absorption
L'estradiol est sujet à un effet de premier passage marqué après administration orale.La biodisponibilité absolue est d'environ 1%. Aucun effet cliniquement pertinent n'a été observé avec la prise alimentaire sur la biodisponibilité de l'estradiol.
Distribution
La distribution de l'œstradiol exogène et endogène est similaire.Les œstrogènes sont largement distribués dans le corps et généralement présents à des concentrations plus élevées dans les organes cibles des hormones sexuelles. L'estradiol circule dans le sang lié à la SHBG (37 %) et à l'albumine (61 %), alors que seulement 1 à 2 % environ est libre.
Biotransformation
L'estradiol exogène administré par voie orale est largement métabolisé.Le métabolisme de l'estradiol exogène et endogène est similaire. L'estradiol est rapidement transformé dans l'intestin et le foie en de nombreux métabolites, principalement l'estrone, qui sont ensuite conjugués et pénètrent dans la circulation entérohépatique. Il existe un équilibre dynamique entre l'œstradiol, l'œstrone et l'œstrone-sulfate en raison de différentes activités enzymatiques, notamment l'œstradiol-déshydrogénase, la sulfotransférase et l'aryl sulfatase. L'oxydation de l'estrone et de l'estradiol est due aux enzymes du cytochrome P450, principalement CYP1A2, CYP1A2 (extrahépatique), CYP3A4, CYP3A5, CYP1B1 et CYP2C9.
Élimination
L'œstradiol est rapidement éliminé de la circulation. En raison du métabolisme et de la circulation entérohépatique, il existe un important pool circulant de sulfates d'œstrogènes et de glucuronides. Cela se traduit par une demi-vie d'élimination très variable de l'œstradiol corrigée au départ, qui est égale à 3, 6 ± 1,5 h après l'administration intraveineuse.
Conditions d'état stable
Les concentrations sériques maximales d'estradiol s'élèvent à environ 90 pg/ml et sont atteintes 6 heures après l'administration.Les concentrations sériques moyennes sont de 50 pg/ml et ces taux d'estradiol correspondent à la phase précoce et tardive du cycle menstruel.
Populations particulières
Population pédiatrique
La pharmacocinétique de l'acétate de nomégestrol (critère d'évaluation principal) après une dose orale unique de Zoely était similaire chez les adolescentes en bonne santé post-ménarche et les sujets adultes. Cependant, après une dose orale unique, l'exposition au composant estradiol (objectif secondaire) était inférieure de 36 % chez les adolescents par rapport aux adultes.La pertinence clinique de ce résultat est inconnue.
Effets de l'insuffisance rénale
Aucune étude n'a été réalisée pour déterminer les effets d'une maladie rénale sur la pharmacocinétique de Zoely.
Effets de l'insuffisance hépatique
Aucune étude n'a été réalisée pour déterminer les effets d'une maladie du foie sur la pharmacocinétique de Zoely. Cependant, les hormones stéroïdes peuvent être mal métabolisées chez les femmes présentant une insuffisance hépatique.
Groupes ethniques
Aucune étude formelle n'a été réalisée pour déterminer la pharmacocinétique dans les groupes ethniques.
05.3 Données de sécurité précliniques
Des études de toxicité à doses répétées avec l'œstradiol, l'acétate de nomégestrol ou l'association ont montré les effets attendus des œstrogènes et des progestatifs.
Les études de toxicité pour la reproduction réalisées avec l'association ont montré une toxicité fœtale compatible avec une exposition à l'estradiol.
Aucune étude de génotoxicité et de cancérogénicité n'a été menée avec l'association. L'acétate de nomégestrol n'est pas génotoxique.
Cependant, il faut se rappeler que les stéroïdes sexuels peuvent favoriser la prolifération de certains tissus et tumeurs hormono-dépendants.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Noyau du comprimé (comprimés pelliculés actifs blancs et comprimés pelliculés placebo jaunes)
Lactose monohydraté
Cellulose microcristalline (E460)
Crospovidone (E1201)
Talc (E553b)
Stéarate de magnésium (E572)
Silice colloïdale anhydre
Pelliculage du comprimé (comprimés pelliculés actifs blancs)
Poly (alcool vinylique) (E1203)
Dioxyde de titane (E171)
Macrogol 3350
Talc (E553b)
Pelliculage du comprimé (comprimés pelliculés placebo jaunes)
Poly (alcool vinylique) (E1203)
Dioxyde de titane (E171)
Macrogol 3350
Talc (E553b)
Oxyde de fer jaune (E172)
Oxyde de fer noir (E172)
06.2 Incompatibilité
Non pertinent.
06.3 Durée de validité
3 années
06.4 Précautions particulières de conservation
Ce médicament ne nécessite aucune condition particulière de conservation.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Plaquette thermoformée PVC/aluminium contenant 28 comprimés pelliculés (24 comprimés pelliculés blancs et 4 comprimés pelliculés jaunes).
Présentations : 28, 84, 168 et 364 comprimés pelliculés.
Toutes les présentations peuvent ne pas être commercialisées.
06.6 Instructions d'utilisation et de manipulation
Les comprimés de COC (y compris les comprimés de Zoely) qui ne sont plus nécessaires ne doivent pas être jetés au tout-à-l'égout ou avec les ordures ménagères.Les substances hormonalement actives contenues dans le comprimé peuvent avoir des effets nocifs si elles atteignent le milieu aquatique. Les comprimés doivent être retournés à la pharmacie ou éliminés en toute sécurité conformément aux réglementations locales. Cela contribuera à protéger l'environnement.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Teva B.V.
Swensweg 5
2031 AG Haarlem
Pays-Bas
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
UE / 1/11/690/001
UE / 1/11/690/002
UE / 1/11/690/003
UE / 1/11/690/004
041400019
041400021
041400033
041400045
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
Date de première autorisation : 27 juillet 2011
10.0 DATE DE RÉVISION DU TEXTE
Mai 2015







-cloruro.jpg)