Ingrédients actifs : Epoétine alfa
Binocrit 1000 UI / 0,5 mL solution injectable en seringue préremplie
Binocrit 2 000 UI/1 mL solution injectable en seringue préremplie
Binocrit 3000 UI/0,3 mL solution injectable en seringue préremplie
Binocrit 4000 UI/0,4 mL solution injectable en seringue préremplie
Binocrit 5 000 UI / 0,5 mL solution injectable en seringue préremplie
Binocrit 6 000 UI / 0,6 mL solution injectable en seringue préremplie
Binocrit 7 000 UI/0,7 mL solution injectable en seringue préremplie
Binocrit 8 000 UI / 0,8 mL solution injectable en seringue préremplie
Binocrit 9 000 UI/0,9 mL solution injectable en seringue préremplie
Binocrit 10 000 UI/1 mL solution injectable en seringue préremplie
Binocrit 20 000 UI / 0,5 mL solution injectable en seringue préremplie
Binocrit 30 000 UI/0,75 mL solution injectable en seringue préremplie
Binocrit 40 000 UI/1 mL solution injectable en seringue préremplie
Indications Pourquoi utiliser Binocrit ? Pourquoi est-ce?
Binocrit contient le principe actif époétine alfa, une protéine qui stimule la moelle osseuse à produire davantage de globules rouges qui transportent l'hémoglobine (une substance qui transporte l'oxygène).
L'époétine alfa est une copie de la protéine humaine érythropoïétine et agit de la même manière.
Binocrit est utilisé pour traiter l'anémie symptomatique causée par une maladie rénale :
- chez les enfants hémodialysés
- chez l'adulte en hémodialyse ou en dialyse péritonéale,
- chez les adultes sévèrement anémiques non encore dialysés.
Si vous souffrez d'une maladie rénale, vous pouvez avoir une pénurie de globules rouges si le rein ne produit pas suffisamment d'érythropoïétine (ce qui est nécessaire à la production de globules rouges). Binocrit est prescrit pour stimuler la moelle osseuse à produire plus de globules rouges.
Binocrit est utilisé pour traiter l'anémie lorsque vous recevez une chimiothérapie pour des tumeurs solides, un lymphome malin ou un myélome multiple (cancer de la moelle osseuse) et si votre médecin décide que vous pourriez avoir besoin d'une transfusion sanguine. Binocrit peut réduire le besoin de transfusions sanguines.
Binocrit est utilisé chez les personnes modérément anémiques qui donnent une partie de leur sang avant la chirurgie afin que le sang collecté puisse leur être donné pendant ou après la chirurgie. Parce que Binocrit stimule la production de globules rouges, les médecins peuvent prélever plus de sang chez ces personnes.
Binocrit est utilisé chez les adultes modérément anémiques qui sont sur le point de subir une chirurgie orthopédique majeure (telle qu'une arthroplastie de la hanche ou du genou) afin de réduire le besoin potentiel de transfusions sanguines.
Contre-indications Quand Binocrit ne doit pas être utilisé
Ne pas utiliser Binocrit
- si vous êtes allergique à l'époétine alfa ou à l'un des autres composants contenus dans ce médicament (mentionnés dans la rubrique 6).
- si vous avez reçu un diagnostic d'aplasie pure des globules rouges (la moelle osseuse ne peut pas fabriquer suffisamment de globules rouges) après un traitement par un médicament stimulant la production de globules rouges (y compris Binocrit). Voir rubrique 4.
- si vous souffrez d'hypertension artérielle insuffisamment contrôlée par des médicaments.
- pour stimuler la production de globules rouges (afin que les médecins puissent vous prélever plus de sang) si vous ne pouvez pas recevoir de transfusions avec votre propre sang pendant ou après la chirurgie.
- si vous êtes sur le point de subir une chirurgie orthopédique élective majeure (comme une chirurgie de la hanche ou du genou) et :
- avez une maladie cardiaque grave
- avez une maladie veineuse ou artérielle grave
- avez récemment eu une crise cardiaque ou un accident vasculaire cérébral
- ne pouvez pas prendre de médicaments pour fluidifier le sang Binocrit peut ne pas vous convenir. Discutez-en avec votre médecin.
Certaines personnes ont besoin de médicaments pour réduire le risque de caillots sanguins pendant leur traitement par Binocrit. Si vous ne pouvez pas prendre de médicaments qui empêchent la formation de caillots sanguins, vous ne devez pas prendre Binocrit.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Binocrit
Adressez-vous à votre médecin, pharmacien ou infirmier/ère avant d'utiliser Binocrit.
Binocrit et d'autres produits qui stimulent la production de globules rouges peuvent augmenter le risque de caillots sanguins chez tous les patients. Ce risque peut être plus élevé si vous avez d'autres facteurs de risque de développer des caillots sanguins (par exemple, si vous avez déjà eu un caillot sanguin ou si vous êtes en surpoids, souffrez de diabète, souffrez d'une maladie cardiaque ou devez vous allonger pendant un certain temps). à long terme en raison d'une intervention chirurgicale ou d'une maladie). Informez votre médecin d'une telle situation. Votre médecin vous aidera à décider si Binocrit vous convient.
Il est important que vous informiez votre médecin si l'un des cas suivants s'applique à vous.
Vous pourrez peut-être utiliser Binocrit de toute façon, mais vous devez d'abord en parler à votre médecin.
Si vous savez que vous souffrez ou avez souffert dans le passé de :
- hypertension artérielle;
- convulsions ou convulsions;
- maladie du foie;
- anémie d'autres causes;
- porphyrie (une maladie rare du sang).
Si vous êtes un patient cancéreux, sachez que les médicaments qui stimulent la production de globules rouges (comme Binocrit) peuvent agir comme des facteurs de croissance et pourraient donc théoriquement affecter la progression de la tumeur.
Selon votre situation personnelle, une transfusion sanguine peut être préférable. Discutez-en avec votre médecin.
Si vous avez une hépatite C et que vous recevez de l'interféron et de la ribavirine, vous devez en discuter avec votre médecin car l'association de l'époétine alfa avec l'interféron et la ribavirine a, dans de rares cas, entraîné une réduction de l'effet du traitement et une affection appelée aplasie pure des globules rouges ( PRCA), une forme sévère d'anémie. Binocrit n'est pas approuvé pour le traitement de l'anémie associée à l'hépatite C.
Si vous êtes un patient atteint d'insuffisance rénale chronique, et en particulier si vous ne répondez pas adéquatement à Binocrit, votre médecin vérifiera la dose de Binocrit que vous recevez car l'augmentation répétée de la dose de Binocrit s'il ne répond pas au traitement peut augmenter le risque. au cœur ou aux vaisseaux sanguins et le risque d'infarctus du myocarde, d'accident vasculaire cérébral et de décès.
Faites attention avec les autres produits stimulant la production de globules rouges : Binocrit appartient à un groupe de produits qui, comme la protéine humaine érythropoïétine, stimulent la production de globules rouges. Votre professionnel de la santé prendra toujours note du produit spécifique que vous utilisez.Si vous recevez un médicament de ce groupe, autre que Binocrit, pendant votre traitement, parlez-en à votre médecin ou votre pharmacien avant de l'utiliser.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Binocrit
Binocrit ne réagit généralement pas avec d'autres médicaments, mais informez votre médecin si vous utilisez, avez récemment utilisé ou pourriez utiliser tout autre médicament, y compris ceux obtenus sans ordonnance.
Si vous prenez un médicament appelé ciclosporine (utilisé, par exemple, après une greffe de rein), votre médecin pourra vous faire effectuer des analyses de sang pour mesurer votre taux de ciclosporine pendant que vous prenez Binocrit.
Les suppléments de fer et autres stimulants sanguins peuvent augmenter l'efficacité de Binocrit.Votre médecin décidera s'il est bon pour vous de les prendre.
Si vous vous rendez dans un hôpital, une clinique ou un médecin de famille, dites-leur que vous êtes traité par Binocrit. Cela pourrait affecter d'autres traitements ou résultats de tests.
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Il est important d'informer votre médecin si l'un des cas suivants s'applique à vous.
Vous pourrez peut-être utiliser Binocrit de toute façon, mais vous devriez d'abord en discuter avec votre médecin :
- si vous êtes enceinte ou si vous soupçonnez une grossesse.
- si vous allaitez.
Binocrit contient du sodium
Binocrit contient moins de 1 mmol (23 mg) de sodium par dose, c'est-à-dire qu'il est essentiellement « sans sodium ».
Dose, mode et heure d'administration Comment utiliser Binocrit : Posologie
Utilisez toujours ce médicament en suivant exactement les indications de votre médecin. En cas de doute, consultez votre médecin.
Le médecin a fait un test sanguin et a décidé que vous aviez besoin de Binocrit.
Binocrit peut être administré par injection :
- dans une veine ou un tube qui pénètre dans une veine (par voie intraveineuse)
- ou sous la peau (sous-cutanée).
Votre médecin décidera comment Binocrit sera injecté. Habituellement, les injections sont administrées par un médecin, une infirmière ou un autre professionnel de la santé. Certaines personnes, selon la raison pour laquelle elles ont besoin d'un traitement par Binocrit, peuvent apprendre plus tard comment l'injecter sous la peau : voir Instructions pour l'auto-injection de Binocrit à la fin de cette notice.
Binocrit ne doit pas être utilisé :
- après la date de péremption indiquée sur l'étiquette et l'emballage extérieur
- si vous savez ou pensez qu'il a été accidentellement congelé ou
- si une panne du réfrigérateur s'est produite.
La dose de Binocrit qui vous sera administrée dépend de votre poids corporel en kilogrammes. La cause de l'anémie est également importante pour que le médecin choisisse la bonne dose.
Votre médecin contrôlera régulièrement votre tension artérielle pendant le traitement par Binocrit.
Les personnes atteintes d'une maladie rénale
- Votre médecin maintiendra votre taux d'hémoglobine entre 10 et 12 g/dL, car un taux d'hémoglobine élevé pourrait augmenter le risque de caillots sanguins et de décès.
- La dose initiale habituelle de Binocrit chez les adultes et les enfants est de 50 unités internationales (UI) par kilogramme (/kg) de poids corporel, administrée trois fois par semaine. Chez les patients sous dialyse péritonéale, Binocrit peut être administré deux fois par semaine.
- Chez l'adulte et l'enfant, Binocrit est administré par injection dans une veine (intraveineuse) ou dans un tube qui pénètre dans une veine. Lorsque cet accès (par une veine ou un tube) n'est pas facilement accessible, le médecin peut décider d'injecter Binocrit sous la peau (par voie sous-cutanée). Cela affecte les patients dialysés et les patients non encore dialysés.
- Votre médecin vous prescrira des analyses de sang sur une base régulière pour voir comment l'anémie répond au traitement et peut ajuster la dose, généralement toutes les quatre semaines au plus tard.
- Une fois l'anémie corrigée, votre médecin continuera à contrôler votre sang régulièrement. La posologie et la fréquence d'administration de Binocrit peuvent être ajustées davantage pour maintenir votre réponse au traitement. Votre médecin utilisera la dose efficace la plus faible pour contrôler vos symptômes. .
- Si vous ne répondez pas de manière adéquate à Binocrit, votre médecin vérifiera la dose que vous recevez et vous informera si vos doses de Binocrit doivent être modifiées.
- Si vous utilisez un intervalle plus long (supérieur à une fois par semaine) entre les doses de Binocrit, vous ne pourrez peut-être pas maintenir des taux d'hémoglobine adéquats et devrez peut-être augmenter la dose ou la fréquence d'administration de Binocrit.
- Vous pouvez également recevoir une supplémentation en fer avant et pendant le traitement par Binocrit pour augmenter l'efficacité du traitement.
- Si vous êtes sous dialyse au début du traitement par Binocrit, votre programme de dialyse devra peut-être être modifié. Le médecin décidera de cela.
Adultes sous chimiothérapie
- Votre médecin peut commencer le traitement par Binocrit si votre taux d'hémoglobine est de 10 g/dL ou moins.
- Votre médecin maintiendra votre taux d'hémoglobine entre 10 et 12 g/dL, car un taux d'hémoglobine élevé peut augmenter le risque de caillots sanguins et de décès.
- La dose initiale habituelle est de 150 UI par kilogramme de poids corporel trois fois par semaine ou de 450 UI par kilogramme de poids corporel une fois par semaine.
- Binocrit est administré par injection sous la peau.
- Votre médecin vous prescrira des analyses de sang et pourra ajuster la dose en fonction de la réponse de votre anémie au traitement.
- Vous pouvez également recevoir une supplémentation en fer avant et pendant le traitement par Binocrit pour augmenter l'efficacité du traitement.
- Le traitement par Binocrit se poursuivra généralement pendant un mois après la fin de la chimiothérapie.
Adultes qui donnent leur propre sang
- La dose habituelle est de 600 UI par kilogramme de poids corporel, deux fois par semaine.
- Binocrit est administré par injection dans une veine, immédiatement après le don de sang, pendant 3 semaines avant la chirurgie.
- Vous pouvez également recevoir une supplémentation en fer avant et pendant le traitement par Binocrit pour augmenter l'efficacité du traitement. Adultes devant subir une chirurgie orthopédique majeure
- La dose recommandée est de 600 UI par kilogramme de poids corporel une fois par semaine.
- Binocrit est administré par injection sous la peau chaque semaine pendant trois semaines avant l'intervention et le jour de l'intervention.
- S'il est nécessaire de raccourcir le délai avant l'intervention, une dose quotidienne de 300 UI/kg est administrée pendant dix jours maximum avant l'intervention, le jour de l'intervention et dans les quatre jours suivants.
- Si les tests sanguins montrent des valeurs d'hémoglobine trop élevées avant la chirurgie, le traitement sera arrêté.
- Vous pouvez recevoir une supplémentation en fer avant et pendant le traitement par Binocrit pour augmenter l'efficacité du traitement.
Instructions pour l'injection de Binocrit sous la peau
Au début du traitement, Binocrit est généralement injecté par le personnel médical ou paramédical.Par la suite, votre médecin peut vous suggérer, ou un soignant, d'apprendre à injecter Binocrit sous la peau (par voie sous-cutanée) par vous-même.
- N'essayez pas de vous injecter ce médicament à moins que votre médecin ou votre infirmier/ère ne vous ait montré comment procéder.
- Utilisez toujours Binocrit exactement comme indiqué par votre médecin ou votre infirmière.
- Assurez-vous de n'injecter que la quantité de liquide indiquée par votre médecin ou votre infirmière.
- N'utilisez Binocrit que s'il a été correctement conservé - voir la rubrique 5, Comment conserver Binocrit.
- Avant utilisation, laissez reposer la seringue Binocrit jusqu'à ce qu'elle atteigne la température ambiante. Cela prend généralement 15 à 30 minutes. Utilisez la seringue dans les 3 jours suivant sa sortie du réfrigérateur.
Prenez une seule dose de Binocrit par seringue.
Si Binocrit est injecté sous la peau (par voie sous-cutanée), le volume injecté ne dépasse généralement pas un millilitre (1 ml) par injection unique.
Binocrit est administré seul et non mélangé avec d'autres liquides injectables.
Ne pas agiter les seringues Binocrit. Une agitation vigoureuse prolongée peut endommager le produit. Si le produit a été secoué vigoureusement, ne l'utilisez pas.
Les instructions d'auto-injection de Binocrit sont fournies à la fin de cette notice.
Si vous oubliez d'utiliser Binocrit
Administrez la prochaine injection dès que vous vous en souvenez. S'il reste moins d'un jour avant la prochaine injection, sautez l'injection manquée et poursuivez votre programme normal. N'injectez pas une double dose.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin, votre infirmière ou votre pharmacien.
Surdosage Que faire si vous avez pris trop de Binocrit
Informez immédiatement votre médecin ou votre infirmier/ère si vous pensez qu'une trop grande quantité de Binocrit a été injectée. Des effets secondaires sont peu probables en cas de surdosage de Binocrit.
Effets secondaires Quels sont les effets secondaires de Binocrit
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Informez immédiatement votre médecin ou votre infirmier/ère si vous remarquez l'un des effets indésirables mentionnés.
Effets secondaires très fréquents
Ils peuvent affecter plus de 1 personne sur 10 utilisant Binocrit.
- La diarrhée
- La nausée
- il vomit
- Fièvre
- Une congestion des voies respiratoires, telle qu'un nez bouché et un mal de gorge, a été signalée chez des patients atteints d'insuffisance rénale et non encore dialysés.
Effets secondaires courants
Ceux-ci peuvent affecter jusqu'à 1 personne sur 10 utilisant Binocrit.
- Augmentation de la pression artérielle. Les signes suivants peuvent indiquer une augmentation soudaine de la pression artérielle : maux de tête, en particulier en cas d'apparition soudaine et de type lancinant semblable à la migraine, confusion ou convulsions. Ces signes nécessitent un traitement urgent. L'augmentation de la pression artérielle peut nécessiter un traitement avec d'autres médicaments (ou un ajustement de la posologie des médicaments que vous prenez déjà pour l'hypertension).
- Caillots sanguins (y compris thrombose veineuse profonde et embolie) pouvant nécessiter une intervention urgente. Les symptômes peuvent inclure des douleurs thoraciques, un essoufflement et un gonflement avec douleur et rougeur, généralement dans les jambes.
- La toux.
- Éruption cutanée, qui peut être due à une réaction allergique.
- Douleur dans les os ou les muscles.
- Symptômes pseudo-grippaux tels que maux de tête, douleurs articulaires, sensation de faiblesse, frissons, fatigue et vertiges. Ces symptômes peuvent être plus fréquents au début du traitement. Si vous ressentez ces symptômes lors d'une injection dans la veine, une injection plus lente peut aider à les éviter à l'avenir.
- Rougeur, sensation de brûlure et douleur au site d'injection.
- Gonflement des chevilles, des pieds ou des doigts.
Effets secondaires peu fréquents
Ceux-ci peuvent affecter jusqu'à 1 personne sur 100 utilisant Binocrit.
- Des niveaux élevés de potassium dans le sang, ce qui peut entraîner des rythmes cardiaques anormaux (il s'agit d'un effet secondaire très fréquent chez les patients dialysés).
- Convulsions.
- Congestion du nez ou des voies respiratoires.
Effets secondaires très rares
Ceux-ci peuvent affecter jusqu'à 1 personne sur 10 000 qui utilisent Binocrit.
- Symptômes de l'aplasie pure des globules rouges (PRCA) L'aplasie pure des globules rouges (PRCA) signifie que la moelle osseuse ne fabrique pas suffisamment de globules rouges. PRCA provoque « une anémie soudaine et sévère. Les symptômes sont :
- fatigue inhabituelle,
- avoir le vertige,
- essoufflement.
Un PRCA a été très rarement rapporté, en particulier chez des patients atteints d'insuffisance rénale, après des mois ou des années de traitement par l'époétine alfa et d'autres médicaments stimulant la production de globules rouges.
- En particulier au début du traitement, il peut y avoir une augmentation du nombre de certaines petites cellules sanguines (appelées plaquettes), qui sont normalement impliquées dans la formation de caillots.Votre médecin effectuera les contrôles appropriés.
Si vous êtes sous hémodialyse :
- Des caillots (thrombose) peuvent se former dans la fistule de dialyse. Cela est plus susceptible de se produire si vous avez une pression artérielle basse ou s'il y a des complications avec la fistule.
- Des caillots peuvent également se former dans le système d'hémodialyse. Le médecin peut décider d'augmenter la dose d'héparine pendant la dialyse.
Informez immédiatement votre médecin ou votre infirmier/ère si vous remarquez l'un de ces effets ou si vous remarquez tout autre effet pendant que vous prenez Binocrit.
Si l'un des effets indésirables devient grave ou si vous remarquez un effet indésirable non mentionné dans cette notice, veuillez en informer votre médecin, votre infirmière ou votre pharmacien.
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin, pharmacien ou infirmier/ère. Cela inclut tout effet indésirable éventuel non mentionné dans cette notice. Vous pouvez également déclarer les effets indésirables directement via le système national de déclaration (voir Annexe V). effets indésirables que vous pouvez aider fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
- Gardez ce médicament hors de la vue et de la portée des enfants.
- N'utilisez pas ce médicament après la date de péremption indiquée sur l'étiquette après « EXP » et sur la boîte après « EXP ».
- A conserver et transporter réfrigéré (2°C - 8°C).
- Vous pouvez sortir Binocrit du réfrigérateur et le conserver à température ambiante (jusqu'à 25°C) jusqu'à 3 jours. Une fois que la seringue a été sortie du réfrigérateur et a atteint la température ambiante (jusqu'à 25°C), elle doit être utilisée dans les 3 jours ou jetée.
- Ne pas congeler ni secouer.
- Conserver dans l'emballage d'origine pour protéger le médicament de la lumière.
N'utilisez pas ce médicament si vous remarquez
- qui a été accidentellement congelé ou
- qu'une panne du réfrigérateur s'est produite
- que le liquide est coloré ou s'il voit des particules flotter dedans
- que le sceau est brisé.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Délai "> Autres informations
Ce que contient Binocrit
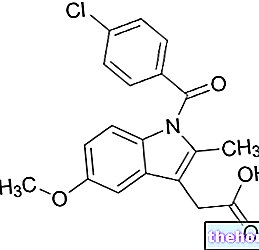
- L'ingrédient actif est l'époétine alfa (pour la quantité, voir le tableau ci-dessous).
- Les autres composants sont le phosphate monosodique dihydraté, le phosphate disodique dihydraté, le chlorure de sodium, la glycine, le polysorbate 80, l'acide chlorhydrique (pour ajuster le pH), l'hydroxyde de sodium (pour ajuster le pH), l'eau pour préparations injectables.
A quoi ressemble Binocrit et contenu de l'emballage extérieur
Binocrit se présente sous la forme d'une solution injectable limpide et incolore en seringue préremplie. Les seringues sont scellées dans des blisters.
* Boîtes de 1, 4 ou 6 seringues préremplies avec ou sans protège-aiguille.
Toutes les présentations peuvent ne pas être commercialisées.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT -
SOLUTION BINOCRIT POUR INJECTION EN SERINGUE PREREMPLIE
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE -
Binocrit 1000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque mL de solution contient 2 000 UI d'époétine alfa*, correspondant à 16,8 mcg par mL
Une seringue préremplie de 0,5 ml contient 1 000 unités internationales (UI), correspondant à 8,4 mcg d'époétine alfa. *
Binocrit 2 000 UI/1 mL solution injectable en seringue préremplie
Chaque mL de solution contient 2 000 UI d'époétine alfa*, correspondant à 16,8 mcg par mL
Une seringue préremplie de 1 mL contient 2 000 unités internationales (UI), correspondant à 16,8 mcg d'époétine alfa. *
Binocrit 3000 UI/0,3 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,3 ml contient 3 000 unités internationales (UI), correspondant à 25,2 mcg d'époétine alfa. *
Binocrit 4000 UI/0,4 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,4 mL contient 4 000 unités internationales (UI), correspondant à 33,6 mcg d'époétine alfa. *
Binocrit 5 000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,5 ml contient 5 000 unités internationales (UI), correspondant à 42,0 mcg d'époétine alfa. *
Binocrit 6 000 UI / 0,6 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,6 ml contient 6 000 unités internationales (UI), correspondant à 50,4 mcg d'époétine alfa. *
Binocrit 7 000 UI/0,7 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,7 ml contient 7 000 unités internationales (UI), correspondant à 58,8 mcg d'époétine alfa. *
Binocrit 8 000 UI / 0,8 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,8 ml contient 8 000 unités internationales (UI), correspondant à 67,2 mcg d'époétine alfa. *
Binocrit 9 000 UI/0,9 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 0,9 ml contient 9 000 unités internationales (UI), correspondant à 75,6 mcg d'époétine alfa. *
Binocrit 10 000 UI/1 mL solution injectable en seringue préremplie
Chaque mL de solution contient 10 000 UI d'époétine alfa*, correspondant à 84,0 mcg par mL
Une seringue préremplie de 1 mL contient 10 000 unités internationales (UI), correspondant à 84,0 mcg d'époétine alfa. *
Binocrit 20 000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque mL de solution contient 40 000 UI d'époétine alfa*, correspondant à 336,0 mcg par mL
Une seringue préremplie de 0,5 ml contient 20 000 unités internationales (UI), correspondant à 168,0 mcg d'époétine alfa. *
Binocrit 30 000 UI/0,75 mL solution injectable en seringue préremplie
Chaque mL de solution contient 40 000 UI d'époétine alfa*, correspondant à 336,0 mcg par mL
Une seringue préremplie de 0,75 ml contient 30 000 unités internationales (UI), correspondant à 252,0 mcg d'époétine alfa. *
Binocrit 40 000 UI/1 mL solution injectable en seringue préremplie
Chaque mL de solution contient 40 000 UI d'époétine alfa*, correspondant à 336,0 mcg par mL
Une seringue préremplie de 1 ml contient 40 000 unités internationales (UI), correspondant à 336,0 mcg d'époétine alfa. *
* Produit dans des cellules d'ovaire de hamster chinois (CHO) par la technologie de l'ADN recombinant
Pour la liste complète des excipients, voir rubrique 6.1.
Ce médicament contient moins de 1 mmol (23 mg) de sodium par dose, c'est-à-dire qu'il est essentiellement « sans sodium ».
03.0 FORME PHARMACEUTIQUE -
Solution injectable en seringue préremplie (injection)
Solution claire et incolore
04.0 INFORMATIONS CLINIQUES -
04.1 Indications thérapeutiques -
Binocrit est indiqué dans le traitement de l'anémie symptomatique associée à l'insuffisance rénale chronique (IRC) :
• chez les adultes et les enfants hémodialysés âgés de 1 à 18 ans et les patients adultes sous dialyse péritonéale (voir rubrique 4.4).
• chez les patients adultes insuffisants rénaux non encore dialysés pour le traitement de l'anémie sévère d'origine rénale accompagnée de symptômes cliniques (voir rubrique 4.4).
Binocrit est indiqué chez l'adulte recevant une chimiothérapie pour tumeurs solides, lymphome malin ou myélome multiple et à risque transfusionnel, en fonction de l'état général du patient (situation cardiovasculaire, anémie préexistante au début de la chimiothérapie) pour le traitement de l'anémie et la réduction besoins transfusionnels.
Binocrit est indiqué chez les adultes faisant partie d'un programme de prédon autologue pour augmenter la production de sang autologue.Le traitement ne doit être réalisé que chez les patients présentant une anémie modérée (plage de concentration en hémoglobine [Hb] comprise entre 10 et 13 g/dL [6,2 et 8,1 mmol/L], sans carence en fer), lorsque les techniques d'économie de sang ne sont pas disponibles ou insuffisantes et planifiées une chirurgie élective majeure nécessite une grande quantité de sang (4 unités de sang ou plus pour les femmes, 5 unités ou plus pour les hommes).
Binocrit est indiqué chez les adultes non carencés en fer, considérés comme à haut risque de complications transfusionnelles, avant une chirurgie orthopédique élective majeure, afin de réduire l'exposition aux transfusions sanguines allogéniques. Limiter l'utilisation aux patients présentant une anémie modérée (plage de concentration d'hémoglobine entre 10 et 13 g/dL ou entre 6,2 et 8,1 mmol/L) ne faisant pas partie d'un programme de prédon autologue et pour lequel une perte de sang modérée est attendue (900-1 800 mL).
04.2 Posologie et mode d'administration -
Le traitement par Binocrit doit être instauré sous la surveillance de médecins expérimentés dans le traitement des patients présentant les indications ci-dessus.
Dosage
Toutes les autres causes d'anémie (carence en fer, en acide folique ou en vitamine B12, intoxication à l'aluminium, infection ou inflammation, perte de sang, hémolyse et fibrose de la moelle osseuse de toute origine) doivent être évaluées et traitées avant de commencer le traitement par époétine alfa et au moment de décider d'un augmentation de la dose. Pour assurer une réponse optimale à l'époétine alfa, il est nécessaire de s'assurer que des réserves de fer suffisantes existent et, si nécessaire, d'administrer un supplément de fer (voir rubrique 4.4).
Traitement de l'anémie symptomatique chez les patients adultes atteints d'insuffisance rénale chronique
Les symptômes et les conséquences de l'anémie peuvent varier en fonction de l'âge, du sexe et des comorbidités médicales ; une évaluation individuelle de l'évolution clinique et de l'état de chaque patient est requise par le médecin.
La plage de concentration d'hémoglobine souhaitée recommandée est de 10 g/dL à 12 g/dL (6,2 à 7,5 mmol/L).Binocrit doit être administré afin que les valeurs d'hémoglobine n'augmentent pas au-delà de 12 g/dL (7,5 mmol/L). Une élévation du taux d'hémoglobine supérieure à 2 g/dL (1,25 mmol/L) sur une période de plusieurs semaines doit être évitée. Si cela se produit, un ajustement posologique doit être fait en conséquence.
En raison de la variabilité intra-patient, des valeurs individuelles d'hémoglobine supérieures et inférieures à la plage de concentration d'hémoglobine souhaitée peuvent parfois être observées.10 g/dL (6,2 mmol/L) et 12 g/dL (7,5 mmol/L).
Des taux d'hémoglobine prolongés supérieurs à 12 g/dL (7,5 mmol/L) doivent être évités. Si le taux d'hémoglobine augmente de plus de 2 g/dL (1,25 mmol/L) par mois, ou si vous avez maintenu des taux d'hémoglobine supérieurs à 12 g/dL (7,5 mmol/L), réduisez la dose de Binocrit de 25 %. 13 g/dL (8,1 mmol/L), interrompre le traitement jusqu'à ce que les valeurs descendent en dessous de 12 g/dL (7,5 mmol/L) puis reprendre le traitement par Binocrit à une dose 25 % inférieure à la dose précédente.
Les patients doivent être étroitement surveillés pour s'assurer que la dose efficace approuvée la plus faible de Binocrit est utilisée pour un contrôle adéquat de l'anémie et des symptômes de l'anémie, tout en maintenant une concentration d'hémoglobine inférieure ou égale à 12 g/dL (7,45 mmol/L).
Faire preuve de prudence en augmentant les doses de Binocrit chez les patients atteints d'insuffisance rénale chronique. Chez les patients présentant une faible réponse de l'hémoglobine à Binocrit, d'autres explications de la faible réponse doivent être envisagées (voir rubriques 4.4 et 5.1).
Le traitement par Binocrit comprend deux phases : la phase de correction et la phase d'entretien.
Patients adultes hémodialysés
Chez les patients hémodialysés où l'accès intraveineux est facilement accessible, l'administration intraveineuse est préférable.
Phase de correction
La dose initiale est de 50 UI/kg, trois fois par semaine.
Si nécessaire, augmentez ou diminuez la dose de 25 UI/kg (trois fois par semaine) jusqu'à atteindre la plage de concentration d'hémoglobine souhaitée, entre 10 g/dL et 12 g/dL (entre 6,2 et 7,5 mmol/L) (cela doit se produire progressivement à des intervalles d'au moins quatre semaines).
Phase d'entretien
La dose hebdomadaire totale recommandée est comprise entre 75 UI/kg et 300 UI/kg.
Un ajustement adéquat de la dose doit être effectué pour maintenir les valeurs d'hémoglobine dans la plage de concentration souhaitée de 10 g/dL à 12 g/dL (6,2 à 7,5 mmol/L).
Patients avec des valeurs d'hémoglobine initialement très faibles (8 g/dL ou > 5 mmol/L).
Patients adultes insuffisants rénaux non encore dialysés
Lorsque l'accès intraveineux n'est pas facilement accessible, Binocrit peut être administré par voie sous-cutanée.
Phase de correction
Dose initiale de 50 UI/kg, 3 fois par semaine, suivie, si nécessaire, par paliers de 25 UI/kg (3 fois par semaine) jusqu'à atteindre la valeur souhaitée (ceci doit être fait progressivement à des intervalles d'au moins quatre semaines ).
Phase d'entretien
Pendant la phase d'entretien, Binocrit peut être administré 3 fois par semaine ou, en cas d'administration sous-cutanée, une fois par semaine ou une fois toutes les 2 semaines.
Un ajustement approprié de la dose et des intervalles de dosage doit être fait pour maintenir les valeurs d'hémoglobine au niveau souhaité : hémoglobine entre 10 g/dL et 12 g/dL (6,2 à 7,5 mmol/L). L'allongement de l'intervalle entre les doses peut nécessiter une augmentation de la posologie.
La posologie maximale ne doit pas dépasser 150 UI/kg 3 fois par semaine, 240 UI/kg (jusqu'à un maximum de 20 000 UI) une fois par semaine, ou 480 UI/kg (jusqu'à un maximum de 40 000 UI) une fois tous les 2 semaines.
Patients adultes sous dialyse péritonéale
Lorsque l'accès intraveineux n'est pas facilement accessible, Binocrit peut être administré par voie sous-cutanée.
Phase de correction
La dose initiale est de 50 UI/kg, deux fois par semaine.
Phase d'entretien
La dose d'entretien recommandée est comprise entre 25 UI/kg et 50 UI/kg, deux fois par semaine, en 2 injections égales.
Un ajustement adéquat de la dose doit être effectué pour maintenir les valeurs d'hémoglobine au niveau souhaité, entre 10 g/dL et 12 g/dL (6,2 à 7,5 mmol/L).
Traitement des patients adultes atteints d'anémie induite par la chimiothérapie
Les symptômes et les conséquences de l'anémie peuvent varier en fonction de l'âge, du sexe et de la gravité globale de la maladie ; une évaluation individuelle de l'évolution clinique et de l'état de chaque patient est requise par le médecin.
Binocrit doit être administré aux patients anémiques (par exemple avec une concentration en hémoglobine ≤ 10 g/dL (6,2 mmol/L)).
La dose initiale est de 150 UI/kg par voie sous-cutanée, 3 fois par semaine.
Alternativement, Binocrit peut être administré à une dose initiale de 450 UI/kg par voie sous-cutanée une fois par semaine.
Un ajustement adéquat de la dose doit être effectué pour maintenir les valeurs d'hémoglobine dans la plage de concentration souhaitée de 10 g/dL à 12 g/dL (6,2 à 7,5 mmol/L).
En raison de la variabilité intra-patient, des concentrations uniques d'hémoglobine supérieures et inférieures à la plage de concentration d'hémoglobine souhaitée peuvent parfois être observées. Il est recommandé de traiter la variabilité de l'hémoglobine par une gestion optimale de la dose, en tenant compte de la plage de concentration d'hémoglobine. 10 g / dL (6,2 mmol/L) à 12 g/dL (7,5 mmol/L) souhaité Les concentrations d'hémoglobine prolongées supérieures à 12 g/dL (7,5 mmol/L) doivent être évitées ; directives pour un ajustement approprié de la dose pour les concentrations d'hémoglobine supérieures à 12 g/dL (7,5 mmol) / L) sont décrits ci-dessous.
• Si la concentration en hémoglobine a augmenté d'au moins 1 g/dL (0,62 mmol/L) ou si le nombre de réticulocytes a augmenté de ? 40 000 cellules/mcL au-dessus de la valeur initiale après quatre semaines de traitement, une dose de 150 UI/kg trois fois par semaine ou 450 UI/kg une fois par semaine doit être maintenue.
• Si l'augmentation de la concentration d'hémoglobine est
• Si l'augmentation de la concentration d'hémoglobine est
Ajustement de la dose pour maintenir une concentration en hémoglobine entre 10 g/dL et 12 g/dL (6,2 et 7,5 mmol/L)
Si la concentration en hémoglobine augmente de plus de 2 g/dL (1,25 mmol/L) par mois, ou si le taux de concentration en hémoglobine dépasse 12 g/dL (7,5 mmol/L), réduisez la dose de Binocrit d'environ 25-50%.
Si le taux d'hémoglobine dépasse 13 g/dL (8,1 mmol/L), interrompre le traitement jusqu'à ce que les valeurs descendent en dessous de 12 g/dL (7,5 mmol/L) puis reprendre le traitement par Binocrit à une dose 25 % inférieure à la dose précédente.
Le schéma posologique recommandé est indiqué dans le tableau suivant :
Les patients doivent être étroitement surveillés pour s'assurer que la plus faible dose approuvée de l'agent stimulant l'érythropoïèse (agent stimulant l'érythropoïèse, ESA) pour un contrôle adéquat des symptômes de l'anémie.
Le traitement par époétine alfa doit se poursuivre jusqu'à un mois après la fin de la chimiothérapie.
Traitement des patients chirurgicaux adultes participant à un programme de prédon autologue
Les patients présentant une anémie légère (hématocrite entre 33 et 39%), qui nécessitent un pré-dépôt de 4 unités de sang ou plus, doivent être traités avec 600 UI/kg de Binocrit par voie intraveineuse, deux fois par semaine, pendant trois semaines avant la chirurgie. Binocrit doit être administré une fois la procédure de don terminée.
Traitement des patients adultes devant subir une chirurgie orthopédique majeure élective
La dose recommandée est de 600 UI/kg de Binocrit, administrée par voie sous-cutanée une fois par semaine pendant trois semaines (jours -21, -14 et -7) avant l'intervention et le jour de l'intervention (jour 0).
Dans les cas où, pour des raisons médicales, il est nécessaire de réduire le délai opératoire à moins de trois semaines, 300 UI/kg de Binocrit doivent être administrés par voie sous-cutanée pendant 10 jours consécutifs avant l'intervention, le jour de l'intervention et dans les quatre jours qui suivent immédiatement.
Si le taux d'hémoglobine atteint ou dépasse 15 g/dL (9,38 mmol/L) au cours de la période préopératoire, Binocrit doit être arrêté et les doses suivantes ne doivent pas être administrées.
Population pédiatrique
Traitement de l'anémie symptomatique chez les patients atteints d'insuffisance rénale chronique sous hémodialyse
Les symptômes et les conséquences de l'anémie peuvent varier en fonction de l'âge, du sexe et des comorbidités médicales ; une évaluation individuelle de l'évolution clinique et de l'état de chaque patient est requise par le médecin.
Chez les patients pédiatriques, la plage de concentration d'hémoglobine recommandée est de 9,5 g/dL à 11 g/dL (5,9 à 6,8 mmol/L).Binocrit doit être administré de manière à ce que les valeurs d'hémoglobine ne dépassent pas 11 g/dL (6,8 mmol/L). Une augmentation du taux d'hémoglobine supérieure à 2 g/dL (1,25 mmol/L) sur une période de quatre semaines doit être évitée. Un ajustement posologique approprié doit être effectué.
Les patients doivent être étroitement surveillés pour s'assurer que la plus faible dose approuvée de Binocrit est utilisée pour un contrôle adéquat de l'anémie et des symptômes de l'anémie.
Le traitement par Binocrit comprend deux phases : la phase de correction et la phase d'entretien.
Chez les patients pédiatriques sous hémodialyse où l'accès intraveineux est facilement accessible, l'administration intraveineuse est préférable.
Phase de correction
La dose initiale est de 50 UI/kg par voie intraveineuse, 3 fois par semaine.
Si nécessaire, augmenter ou diminuer la dose de 25 UI/kg (trois fois par semaine) jusqu'à atteindre la plage de concentration d'hémoglobine souhaitée, entre 9,5 g/dL et 11 g/dL (entre 5,9 et 6,8 mmol/L) ( cela doit être fait progressivement à des intervalles d'au moins quatre semaines).
Phase d'entretien
Un ajustement approprié de la dose doit être effectué pour maintenir les taux d'hémoglobine dans la plage de concentration souhaitée de 9,5 g/dL à 11 g/dL (5,9 à 6,8 mmol/L).
En règle générale, les enfants pesant moins de 30 kg ont besoin de doses d'entretien plus élevées que les enfants pesant plus de 30 kg et les adultes.
Patients pédiatriques avec des valeurs d'hémoglobine de base très faibles (6,8 g/dL ou > 4,25 mmol/L).
Anémie chez les patients atteints d'insuffisance rénale chronique avant de commencer la dialyse ou de subir une dialyse péritonéale
La sécurité et l'efficacité de l'époétine alfa chez les patients atteints d'insuffisance rénale chronique présentant une anémie avant le début de la dialyse ou devant subir une dialyse péritonéale n'ont pas été établies. Les données actuellement disponibles pour l'utilisation sous-cutanée de l'époétine alfa dans ces populations sont décrites dans la rubrique 5.1, mais aucune recommandation sur la posologie ne peut être faite.
Traitement des patients pédiatriques atteints d'anémie induite par la chimiothérapie
La sécurité et l'efficacité de l'époétine alfa chez les patients pédiatriques sous chimiothérapie n'ont pas été établies (voir rubrique 5.1).
Traitement des patients chirurgicaux pédiatriques participant à un programme de prédon autologue
La sécurité et l'efficacité de l'époétine alfa chez les sujets pédiatriques n'ont pas été établies. Aucune donnée n'est disponible.
Traitement des patients pédiatriques en attente de chirurgie orthopédique majeure élective
La sécurité et l'efficacité de l'époétine alfa chez les sujets pédiatriques n'ont pas été établies. Aucune donnée n'est disponible.
Mode d'administration
Précautions à prendre avant la manipulation ou l'administration du médicament.
Avant utilisation, laissez reposer la seringue Binocrit jusqu'à ce qu'elle atteigne la température ambiante, ce qui prend généralement 15 à 30 minutes.
Comme pour tous les autres produits injectables, vérifiez que la solution ne contient pas de particules et ne présente aucune décoloration. Binocrit est un produit stérile mais sans conservateur et est à usage unique. Administrer le montant requis.
Traitement de l'anémie symptomatique chez les patients adultes atteints d'insuffisance rénale chronique
Chez les patients insuffisants rénaux chroniques pour lesquels un accès intraveineux est régulièrement disponible (patients hémodialysés), l'administration intraveineuse de Binocrit est préférable.
Lorsque l'accès intraveineux n'est pas facilement accessible (patients non encore dialysés et patients subissant une dialyse péritonéale), Binocrit peut être administré par injection sous-cutanée.
Traitement des patients adultes atteints d'anémie induite par la chimiothérapie
Binocrit doit être administré par injection sous-cutanée.
Traitement des patients chirurgicaux adultes participant à un programme de prédon autologue
Binocrit doit être administré par voie intraveineuse.
Traitement des patients adultes devant subir une chirurgie orthopédique majeure élective
Binocrit doit être administré par injection sous-cutanée.
Traitement de l'anémie symptomatique chez les patients pédiatriques atteints d'insuffisance rénale chronique sous hémodialyse
Chez les patients pédiatriques insuffisants rénaux chroniques pour lesquels un accès intraveineux est régulièrement disponible (patients hémodialysés), l'administration intraveineuse de Binocrit est préférable.
Administration intraveineuse
Administrer pendant au moins une à cinq minutes, selon la dose totale. Chez les patients hémodialysés, un bolus peut être administré pendant la séance de dialyse par un accès veineux adapté dans la ligne de dialyse. Alternativement, l'injection peut être administrée à la fin de la séance de dialyse par le tube de l'aiguille de fistule, suivie de 10 ml de sérum physiologique isotonique pour rincer le tube et s'assurer que le produit est injecté dans la circulation (voir Posologie, « Patients adultes en hémodialyse. " ).
Chez les patients réagissant au traitement avec des symptômes pseudo-grippaux, une administration plus lente est préférable (voir rubrique 4.8).
Ne pas administrer Binocrit par perfusion intraveineuse ou en association avec d'autres médicaments en solution (voir rubrique 6.6 pour plus d'informations).
Administration sous-cutanée
Ne pas dépasser le volume maximum de 1 ml à chaque site d'injection. Pour injecter des volumes plus importants, utiliser plusieurs sites d'injection.
Administrer l'injection dans les membres ou la paroi abdominale antérieure.
Dans les cas où le médecin détermine que le patient ou son soignant peut administrer Binocrit par voie sous-cutanée de manière sûre et efficace, des conseils sur la posologie et la méthode d'administration correctes doivent être fournis.
Les « instructions d'auto-injection de Binocrit » se trouvent à la fin de la notice.
04.3 Contre-indications -
• Hypersensibilité à la substance active ou à l'un des excipients mentionnés à la rubrique 6.1.
• Ne pas administrer Binocrit ou toute autre érythropoïétine aux patients qui développent une aplasie érythrocytaire pure (PRCA) après un traitement par une érythropoïétine (voir rubrique 4.4).
• Hypertension non contrôlée.
• Toutes les contre-indications associées aux programmes de prédon de sang autologue doivent être observées chez les patients qui reçoivent un supplément de Binocrit.
L'utilisation de Binocrit chez les patients devant subir une chirurgie orthopédique élective majeure, qui ne participent pas à un programme de prédon autologue, est contre-indiquée chez les patients présentant une maladie coronarienne, artérielle périphérique, carotidienne ou cérébrale sévère, y compris les patients ayant subi une crise cardiaque, un accident myocardique ou cérébrovasculaire récent. .
• Patients chirurgicaux qui, pour une raison quelconque, ne peuvent pas recevoir une prophylaxie antithrombotique adéquate.
04.4 Mises en garde spéciales et précautions d'emploi appropriées -
Considération générale
Chez les patients traités par époétine alfa, la pression artérielle doit être étroitement surveillée et contrôlée si nécessaire. L'époétine alfa doit être utilisée avec prudence en présence d'hypertension non traitée, insuffisamment traitée ou mal contrôlée. Des médicaments peuvent être nécessaires. o augmenter la dose du traitement antihypertenseur Arrêtez le traitement par époétine alfa si la pression artérielle ne peut pas être contrôlée.
Des crises hypertensives avec encéphalopathie et des convulsions nécessitant des soins médicaux immédiats et des soins intensifs sont également survenues au cours du traitement par époétine alfa chez des patients ayant une pression artérielle auparavant normale ou basse de type migraineux, ce qui peut être un signe avant-coureur (voir rubrique 4.8).
L'époétine alfa doit être utilisée avec prudence chez les patients souffrant d'épilepsie, d'antécédents de convulsions ou d'affections médicales associées à une prédisposition aux crises convulsives, telles que les infections du SNC et les métastases cérébrales.
L'époétine alfa doit être utilisée avec prudence chez les patients présentant une insuffisance hépatique chronique.La sécurité de l'époétine alfa n'est pas établie chez les patients présentant une insuffisance hépatique.
Une « incidence accrue d'événements thrombotiques vasculaires (TEV) a été observée chez les patients traités par ASE (voir rubrique 4.8). Ceux-ci incluent des thromboses veineuses et artérielles et des embolies (y compris certains cas d'issue fatale), telles que thrombose veineuse profonde, embolie pulmonaire. , thrombose rétinienne et infarctus du myocarde De plus, des accidents vasculaires cérébraux (y compris infarctus cérébral, hémorragie cérébrale et accidents ischémiques transitoires) ont été rapportés.
Le risque rapporté de ces TEV doit être soigneusement évalué par rapport aux bénéfices attendus du traitement par l'époétine alfa, en particulier chez les patients présentant des facteurs de risque préexistants de TEV, y compris l'obésité et des antécédents de TEV (par exemple, thrombose veineuse profonde, embolie pulmonaire et accident vasculaire cérébral). .
Chez tous les patients, les taux d'hémoglobine doivent être étroitement surveillés en raison du risque potentiellement plus élevé d'événements thromboemboliques et d'issue fatale si les patients sont traités à des taux d'hémoglobine supérieurs à la plage de concentration de l'indication.
Une augmentation dose-dépendante modérée de la numération plaquettaire dans la plage normale peut survenir pendant le traitement par l'époétine alfa. Cette augmentation régresse au cours du traitement. De plus, des cas de thrombocytémie au-dessus de la plage normale ont été rapportés. Il est recommandé d'avoir les plaquettes. numération contrôlée régulièrement pendant les huit premières semaines de traitement.
Toutes les autres causes d'anémie (carence en fer, en acide folique ou en vitamine B12, intoxication à l'aluminium, infection ou inflammation, perte de sang, hémolyse et fibrose de la moelle osseuse de toute origine) doivent être évaluées et traitées avant de commencer le traitement par époétine alfa et au moment de décider d'un augmentation de la dose. Dans la plupart des cas, les valeurs de ferritine sérique diminuent en même temps que l'hématocrite augmente. Pour assurer une réponse optimale à l'époétine alfa, il est nécessaire de s'assurer que des réserves de fer suffisantes existent et, si nécessaire, d'administrer un supplément de fer (voir rubrique 4.2) :
• Chez les patients insuffisants rénaux chroniques, l'administration de fer est recommandée (fer élémentaire, 200 à 300 mg/jour par voie orale chez l'adulte et 100 à 200 mg/jour par voie orale chez l'enfant) si les taux sériques de ferritine sont inférieurs à 100 ng/mL.
• Chez les patients cancéreux, l'administration de fer (fer élémentaire, 200 à 300 mg/jour par voie orale) est recommandée si la saturation de la transferrine est inférieure à 20 %.
• Chez les patients participant à un programme de prédon autologue, l'administration de fer (fer élémentaire, 200 mg/jour par voie orale) doit avoir lieu plusieurs semaines avant le début du prédon autologue, afin de former des dépôts de fer abondants avant « l'initiation du traitement par l'époétine alfa et pendant toute la durée du traitement par époétine alfa.
• Chez les patients devant subir une chirurgie orthopédique élective majeure, l'administration de fer (fer élémentaire, 200 mg/jour par voie orale) doit avoir lieu pendant toute la durée du traitement par époétine alfa. Si possible, l'administration de fer doit être débutée avant le début du traitement par époétine alfa. de sorte que des réserves de fer adéquates se forment.
Très rarement, l'apparition ou l'exacerbation d'une porphyrie a été observée chez des patients traités par époétine alfa. L'époétine alfa doit être utilisée avec prudence chez les patients atteints de porphyrie.
Afin d'améliorer la traçabilité des agents stimulant l'érythropoïèse (ASE), le nom de l'agent administré doit être inscrit (ou indiqué) sans ambiguïté dans le dossier de santé du patient.
Les patients ne doivent passer d'un ASE à un autre que sous une surveillance adéquate.
Aplasie pure des globules rouges (PRCA)
Un PRCA médié par des anticorps a été rapporté après des mois ou des années de traitement par époétine sous-cutanée, principalement chez des patients atteints d'insuffisance rénale chronique. Des cas ont également été rapportés chez des patients atteints d'hépatite C traités par interféron et ribavirine, en présence d'un traitement concomitant par ASE. L'époétine alfa n'est pas approuvée pour le traitement de l'anémie associée à l'hépatite C.
Chez les patients chez qui il y a soudainement un manque d'efficacité du traitement, défini par une diminution de l'hémoglobine (1-2 g/dL ou 0,62-1,25 mmol/L par mois) avec des besoins transfusionnels accrus, il convient de déterminer la numération réticulocytaire et typique les causes de non-réponse doivent être analysées (carence en fer, en folate ou en vitamine B12, intoxication à l'aluminium, infection ou inflammation, perte de sang, hémolyse et fibrose médullaire de toute origine).
En cas de diminution paradoxale du taux d'hémoglobine et d'apparition d'une anémie sévère associée à une faible numération des réticulocytes, le traitement par époétine alfa doit être interrompu et des anticorps anti-érythropoïétine doivent être déterminés. Un examen de la moelle osseuse doit également être envisagé pour un « diagnostic éventuel de PRCA ».
Les autres traitements par ASE ne doivent pas être instaurés en raison du risque de réaction croisée.
Traitement de l'anémie symptomatique chez les patients adultes et pédiatriques atteints d'insuffisance rénale chronique
Chez les patients insuffisants rénaux chroniques traités par époétine alfa, les taux d'hémoglobine doivent être mesurés régulièrement jusqu'à ce qu'un taux stable soit atteint, puis à intervalles réguliers.
Chez les patients atteints d'insuffisance rénale chronique, l'augmentation du taux d'hémoglobine doit être d'environ 1 g/dL (0,62 mmol/L) par mois et ne doit pas dépasser 2 g/dL (1,25 mmol/L) par mois, afin de minimiser le risque d'aggravation. de l'hypertension.
Chez les patients atteints d'insuffisance rénale chronique, la concentration d'hémoglobine d'entretien ne doit pas dépasser la limite supérieure de la plage de concentration d'hémoglobine, comme recommandé dans la rubrique 4.2. Un risque accru de décès et d'événements cardiovasculaires graves a été observé dans les essais cliniques. taux de concentration en hémoglobine supérieur à 12 g/dL (7,5 mmol/L).
Les essais cliniques contrôlés n'ont montré aucun bénéfice significatif attribuable à l'administration d'époétines une fois que la concentration d'hémoglobine a dépassé les niveaux nécessaires pour contrôler les symptômes de l'anémie et éviter les transfusions sanguines.
Des précautions doivent être prises lors de l'augmentation des doses de Binocrit chez les patients atteints d'insuffisance rénale chronique, car des doses cumulées élevées d'époétine peuvent être associées à un risque accru de mortalité et d'événements cardiovasculaires et cérébrovasculaires graves. la mauvaise réponse doit être prise en compte (voir rubriques 4.2 et 5.1).
Les patients insuffisants rénaux chroniques traités par époétine alfa par voie sous-cutanée doivent être surveillés périodiquement pour détecter une perte d'efficacité, définie comme une absence ou une réponse réduite au traitement par époétine alfa chez les patients ayant précédemment répondu à ce traitement. Cette affection est caractérisée par une diminution prolongée du taux d'hémoglobine malgré une augmentation de la dose d'époétine alfa (voir rubrique 4.8).
Certains patients utilisant des intervalles plus longs entre les doses d'époétine alfa (supérieurs à une fois par semaine) peuvent ne pas maintenir des taux d'hémoglobine adéquats (voir rubrique 5.1) et peuvent nécessiter une augmentation de la dose d'époétine alfa. Les taux d'hémoglobine doivent être surveillés régulièrement.
Une thrombose du shunt est survenue chez des patients hémodialysés, en particulier chez des patients ayant une tendance à l'hypotension ou avec des complications de fistules artérioveineuses (par exemple sténose, anévrisme, etc.) Chez ces patients, une reprise précoce du shunt et une reprise précoce du shunt sont recommandées. .prophylaxie antithrombotique, par exemple avec de l'acide acétylsalicylique.
Une hyperkaliémie a été observée dans des cas isolés, bien qu'un lien de causalité n'ait pas été établi. Chez les patients atteints d'insuffisance rénale chronique, les électrolytes sériques doivent être surveillés. En présence d'un taux de potassium sérique élevé ou en augmentation, en plus d'un traitement approprié de l'hyperkaliémie, l'arrêt de l'administration d'époétine alfa doit être envisagé jusqu'à ce que le taux de potassium sérique soit corrigé.
En raison de l'augmentation de l'hématocrite, une augmentation de la dose d'héparine pendant l'hémodialyse est souvent nécessaire pendant le traitement par époétine alfa. Si l'héparinisation n'est pas optimale il est possible qu'une occlusion du système de dialyse se produise.
Sur la base des informations actuellement disponibles, la correction de l'anémie par l'époétine alfa chez les patients adultes insuffisants rénaux non encore dialysés n'accélère pas la progression de l'insuffisance rénale.
Traitement des patients atteints d'anémie induite par la chimiothérapie
Chez les patients cancéreux traités par époétine alfa, les taux d'hémoglobine doivent être mesurés régulièrement jusqu'à ce qu'un taux stable soit atteint, puis à intervalles réguliers.
Les époétines sont des facteurs de croissance qui stimulent principalement la production d'érythrocytes. Les récepteurs de l'érythropoïétine peuvent être exprimés à la surface d'une variété de cellules cancéreuses. Comme pour tous les facteurs de croissance, on craint que les époétines puissent stimuler la croissance tumorale. Le rôle des ASE sur la progression tumorale ne peut être exclu sur la survie sans progression réduite. Dans les essais cliniques contrôlés, l'utilisation d'époétine alfa et d'autres ASE a été associée à une réduction du contrôle tumoral locorégional ou à une réduction de la survie globale :
• contrôle locorégional réduit chez les patients atteints d'un cancer avancé de la tête et du cou traités par radiothérapie, lorsqu'il est administré pour atteindre un taux d'hémoglobine supérieur à 14 g/dL (8,7 mmol/L),
• une réduction de la survie globale et une augmentation des décès attribués à la progression tumorale à 4 mois chez les patientes atteintes d'un cancer du sein métastatique traitées par chimiothérapie lorsqu'elle est administrée pour atteindre une plage de concentration d'hémoglobine de 12-14 g/dL (7,5- 8,7 mmol/L) ,
• un risque accru de décès lorsqu'il est administré pour atteindre un taux d'hémoglobine de 12 g/dL (7,5 mmol/L) chez les patients atteints de tumeurs malignes actives, non traités par chimiothérapie ou radiothérapie. L'utilisation d'ASE n'est pas indiquée dans cette population de patients.
• un risque accru de 9 % de progression de la maladie (MP) ou de décès dans le groupe époétine alfa et traitement standard (SOC) et un risque accru de 15 %, qui ne peut être statistiquement exclu chez les patientes atteintes d'un cancer du sein métastatique traitées par chimiothérapie lorsqu'elles sont administrées pour atteindre une plage de concentration en hémoglobine de 10-12 g/dL (6,2-7,5 mmol/L).
Sur la base de ce qui précède, dans certaines conditions cliniques, la transfusion sanguine doit être le traitement privilégié pour la prise en charge de l'anémie chez les patients cancéreux. La décision de traiter par l'érythropoïétine recombinante doit être fondée sur une évaluation du rapport bénéfice/risque avec la participation du patient et doit prendre en considération le contexte clinique spécifique. Les facteurs à prendre en considération dans cette évaluation doivent inclure le type de cancer et son stade, le degré d'anémie, « l'espérance de vie », l'environnement dans lequel le patient est traité et l'état de santé du patient. préférences (voir section 5.1).
Chez les patients cancéreux recevant une chimiothérapie, l'intervalle de 2-3 semaines entre l'administration d'ASE et l'apparition d'érythrocytes induits par l'érythropoïétine doit être pris en compte lors de l'évaluation de la pertinence du traitement par époétine alfa (patients à risque de transfusion).
Patients chirurgicaux participant à des programmes de prédon autologue
Tous les avertissements et précautions spéciaux relatifs aux programmes de prédon autologue doivent être observés ; en particulier, le remplacement du volume doit être effectué régulièrement.
Patients devant subir une chirurgie orthopédique élective majeure
Suivez toujours de bonnes pratiques de gestion du sang pendant la période périopératoire.
Les patients devant subir une chirurgie orthopédique élective majeure doivent recevoir une "prophylaxie antithrombotique adéquate, car des événements thrombotiques et vasculaires peuvent survenir chez les patients chirurgicaux, en particulier chez les patients présentant une maladie cardiovasculaire sous-jacente. Une attention particulière doit également être portée chez les patients prédisposés à développer une TVP De plus, chez les patients ayant un taux d'hémoglobine initial > 13 g/dL (> 8,1 mmol/L), la possibilité ne peut être exclue que le traitement par époétine alfa puisse être associé à un risque accru d'événements thrombotiques/vasculaires postopératoires. , l'époétine alfa ne doit pas être utilisée chez les patients ayant un taux d'hémoglobine initial > 13 g/dL (> 8,1 mmol/L).
Excipients
Ce médicament contient moins de 1 mmol (23 mg) de sodium par seringue préremplie, c'est-à-dire qu'il est essentiellement « sans sodium ».
04.5 Interactions avec d'autres médicaments et autres formes d'interactions -
Il n'existe aucune preuve que le traitement par l'époétine alfa modifie le métabolisme d'autres médicaments.
Les médicaments qui réduisent l'érythropoïèse peuvent réduire la réponse à l'époétine alfa.
Étant donné que la cyclosporine est liée aux érythrocytes, il existe un potentiel d'interaction médicamenteuse. Si l'époétine alfa est administrée en même temps que la ciclosporine, les taux sanguins de ciclosporine doivent être surveillés et la dose de ciclosporine ajustée à mesure que l'hématocrite augmente.
Il n'existe aucune preuve indiquant une « interaction entre l'époétine alfa et le facteur de stimulation des colonies de granulocytes (G-CSF) ou le facteur de stimulation des colonies de granulocytes et de macrophages (GM-CSF) en ce qui concerne la différenciation ou la prolifération hématologique dans les échantillons de biopsie tumorale. in vitro.
Chez les patientes adultes atteintes d'un cancer du sein métastatique, la co-administration sous-cutanée de 40 000 UI/mL d'époétine alfa avec 6 mg/kg de trastuzumab n'a eu aucun effet sur la pharmacocinétique du trastuzumab.
04.6 Grossesse et allaitement -
Grossesse
Il n'y a pas ou peu de données sur l'utilisation de l'époétine alfa chez la femme enceinte.Des études chez l'animal ont montré une toxicité pour la reproduction (voir rubrique 5.3).
Par conséquent, l'époétine alfa ne doit être utilisée pendant la grossesse que si le bénéfice potentiel l'emporte sur le risque potentiel pour le fœtus.
L'heure du repas
On ne sait pas si l'époétine alfa exogène est excrétée dans le lait maternel. L'époétine alfa doit être utilisée avec prudence chez les femmes qui allaitent. La décision d'interrompre l'allaitement ou d'interrompre/de s'abstenir du traitement par l'époétine alfa doit être prise en tenant compte du bénéfice de l'allaitement pour l'enfant et du bénéfice du traitement pour la femme.
L'utilisation d'époétine alfa n'est pas recommandée chez les patientes chirurgicales allaitantes participant à un programme de prédon autologue.
La fertilité
Aucune étude n'a permis de déterminer l'effet potentiel de l'époétine alfa sur la fertilité masculine ou féminine.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines -
Aucune étude sur l'aptitude à conduire des véhicules et à utiliser des machines n'a été réalisée.Binocrit n'a aucun effet ou qu'un effet négligeable sur l'aptitude à conduire des véhicules ou à utiliser des machines.
04.8 Effets indésirables -
Résumé du profil de sécurité
L'effet indésirable le plus fréquent au cours du traitement par époétine alfa est une augmentation dose-dépendante de la pression artérielle ou une aggravation d'une hypertension préexistante.La pression artérielle doit être surveillée, en particulier au début du traitement (voir rubrique 4.4).
Les effets indésirables les plus fréquents observés dans les études cliniques avec l'époétine alfa sont les suivants : diarrhée, nausées, vomissements, fièvre et céphalées. Des syndromes grippaux peuvent survenir en particulier au début du traitement.
Une congestion des voies respiratoires, y compris des événements de congestion des voies respiratoires supérieures, de congestion nasale et de rhinopharyngite, a été rapportée dans des études avec des intervalles de dose prolongés chez des patients adultes atteints d'insuffisance rénale et non encore dialysés.
Une « incidence accrue d'événements thrombotiques vasculaires (ETV) a été observée chez les patients traités par ASE (voir rubrique 4.4).
Tableau des effets indésirables
Sur un total de 3 262 sujets inclus dans 23 études randomisées, en double aveugle, contrôlées contre placebo ou avec des soins standard, le profil de sécurité global de l'époétine alfa a été évalué chez 1 992 sujets anémiques. 228 sujets atteints d'insuffisance rénale chronique traités ont été inclus. avec l'époétine alfa dans 4 études sur l'insuffisance rénale chronique (2 études en pré-dialyse [N = 131 sujets exposés présentant une insuffisance rénale chronique] et 2 sous dialyse [N = 97 sujets exposés présentant une insuffisance rénale chronique] ; 1 404 sujets cancéreux exposés dans 16 études sur l'anémie due à la chimiothérapie ; 147 sujets exposés dans 2 études de don de sang autologue et 213 sujets exposés dans 1 étude périopératoire. Les effets indésirables rapportés par ≥ 1 % des sujets traités par l'époétine alfa dans ces études sont indiqués dans le tableau ci-dessous.
Estimation de la fréquence : très fréquent (≥ 1/10) ; commun (≥ 1/100,
1 Identifié dans l'expérience post-commercialisation et catégorie de fréquence estimée sur la base des taux de notification spontanée
² Commun en dialyse
³ Comprend les événements artériels et veineux, mortels et non mortels, tels que thrombose veineuse profonde, embolie pulmonaire, thrombose rétinienne, thrombose artérielle (y compris infarctus du myocarde), accidents vasculaires cérébraux (y compris infarctus cérébral et hémorragie cérébrale), accidents ischémiques transitoires, shunt thrombose (y compris équipement de dialyse) et thrombose dans les anévrismes de shunt artérioveineux
4 Discuté dans la sous-section ci-dessous et/ou la section 4.4.
Description des effets indésirables sélectionnés
Des réactions d'hypersensibilité, y compris des cas d'éruption cutanée (y compris urticaire), de réactions anaphylactiques et d'œdème de Quincke ont été rapportées (voir rubrique 4.4).
Des crises hypertensives avec encéphalopathie et des convulsions nécessitant des soins médicaux immédiats et des soins intensifs sont également survenues au cours du traitement par époétine alfa chez des patients dont la pression artérielle était auparavant normale ou basse.
Aplasie érythrocytaire pure médiée par les anticorps (en
Population pédiatrique avec insuffisance rénale chronique sous hémodialyse
Exposition de patients pédiatriques insuffisants rénaux chroniques sous hémodialyse dans les essais cliniques et dans l'expérience post-commercialisation c'est limité. Aucun effet indésirable spécifique à l'enfant n'a été rapporté dans cette population qui ne soit pas mentionné dans le tableau ci-dessus ou qui soit incompatible avec la maladie sous-jacente.
Déclaration des effets indésirables suspectés
La déclaration des effets indésirables suspectés survenant après autorisation du médicament est importante car elle permet un suivi continu du rapport bénéfice/risque du médicament. Les professionnels de santé sont invités à déclarer tout effet indésirable suspecté via le système national de déclaration. agenziafarmaco.gov.it/it/responsabili.
04.9 Surdosage -
La marge thérapeutique de l'époétine alfa est très large. Un surdosage d'époétine alfa peut provoquer des effets qui représentent une extension des effets pharmacologiques de l'hormone. En présence de taux d'hémoglobine trop élevés, une phlébotomie peut être utilisée. doit recourir à des traitements de soutien complémentaires. .
05.0 PROPRIÉTÉS PHARMACOLOGIQUES -
05.1 "Propriétés pharmacodynamiques -
Classe pharmacothérapeutique : antianémiques, érythropoïétine, code ATC : B03XA01
Binocrit est un médicament biosimilaire. Des informations plus détaillées sont disponibles sur le site Internet de l'Agence européenne des médicaments : http://www.ema.europa.eu.
Mécanisme d'action
L'érythropoïétine (EPO) est une hormone glycoprotéique produite principalement par le rein en réponse à l'hypoxie et est le régulateur central de la production d'érythrocytes. « L'EPO est impliquée dans toutes les phases du développement érythroïde et son effet principal s'exprime au niveau des précurseurs érythroïdes. Après s'être lié à son récepteur à la surface cellulaire, l'EPO active les voies de transmission des signaux qui interfèrent avec « l'apoptose et stimule la prolifération. de cellules érythroïdes.
L'EPO humaine recombinante (époétine alfa), exprimée dans les cellules d'ovaire de hamster chinois, a une séquence de 165 acides aminés identique à celle de l'EPO urinaire humaine ; les deux substances ne se distinguent pas des analyses fonctionnelles. Le poids moléculaire apparent de l'érythropoïétine est compris entre 32 000 et 40 000 daltons.
L'érythropoïétine est un facteur de croissance qui stimule principalement la production de globules rouges.Les récepteurs de l'érythropoïétine peuvent être exprimés à la surface de différents types de cellules cancéreuses.
Effets pharmacodynamiques
Volontaires sains
Après des doses uniques (20 000 à 160 000 UI par voie sous-cutanée) d'époétine alfa, une réponse dose-dépendante a été observée pour les marqueurs pharmacodynamiques étudiés, notamment les réticulocytes, les érythrocytes et l'hémoglobine. Un profil concentration-temps défini, avec pic et retour à la ligne de base, a été observé pour les changements dans le pourcentage de réticulocytes. Un profil moins défini a été observé pour les érythrocytes et l'hémoglobine.En général, tous les marqueurs pharmacodynamiques ont augmenté en proportion linéaire avec la dose et une réponse maximale a été obtenue à des doses plus élevées.
Des études pharmacodynamiques supplémentaires ont porté sur 40 000 UI une fois par semaine contre 150 UI/kg 3 fois par semaine. Malgré les différences dans les profils concentration-temps, la réponse pharmacodynamique (mesurée par les variations du pourcentage de réticulocytes, d'hémoglobine et d'érythrocytes totaux) était similaire entre ces schémas thérapeutiques. Dans d'autres études, le schéma thérapeutique de 40 000 UI d'époétine alfa une fois par semaine a été comparé avec des doses allant de 80 000 à 120 000 UI administrées par voie sous-cutanée toutes les deux semaines. Globalement, sur la base des résultats de ces études pharmacodynamiques chez des sujets sains, le régime de 40 000 UI une fois par semaine semble être plus efficace en termes de production d'érythrocytes par rapport aux régimes bihebdomadaires, bien qu'une production similaire de réticulocytes ait été observée dans les régimes une fois par semaine et toutes les deux semaines.
L'insuffisance rénale chronique
Il a été démontré que l'époétine alfa stimule l'érythropoïèse chez les patients anémiques atteints d'insuffisance rénale chronique, y compris les patients dialysés et prédialysés. La première réponse notable à l'époétine alfa est une augmentation du nombre de réticulocytes dans les 10 jours, suivie d'une augmentation du nombre d'érythrocytes, de l'hémoglobine et de l'hématocrite, généralement dans les 2 à 6 semaines.La réponse de l'hémoglobine varie d'un patient à l'autre et peut être influencé par les dépôts de fer et la présence de maladies concomitantes.
Anémie induite par la chimiothérapie
Il a été démontré que l'époétine alfa, administrée 3 fois par semaine ou une fois par semaine, augmente l'hémoglobine et réduit le besoin de transfusions après le premier mois de traitement chez les patients atteints d'un cancer anémique sous chimiothérapie.
Dans une étude comparant le schéma thérapeutique à 150 UI/kg 3 fois par semaine et le schéma thérapeutique à 40 000 UI une fois par semaine chez des sujets sains et chez des sujets cancéreux anémiques, les profils temporels des modifications du pourcentage de réticulocytes, d'hémoglobine et de total Les érythrocytes étaient similaires dans les deux schémas chez les sujets sains et les sujets anémiques atteints de cancer.Les ASC des paramètres pharmacodynamiques respectifs étaient similaires dans le schéma à 150 UI/kg 3 fois par semaine et le schéma à 40 000 UI une fois par semaine chez les sujets sains et également chez les sujets sains. sujets atteints de cancer anémique.
Patients chirurgicaux adultes participant à un programme de prédon autologue
Il a été démontré que l'époétine alfa stimule la production d'érythrocytes, permettant une augmentation de la collecte de sang autologue et limitant la réduction du taux d'hémoglobine chez les patients adultes en attente d'une chirurgie élective majeure, pour lesquels on pense que le prédépôt ne répond pas pleinement aux besoins périopératoires en sang. Les effets les plus évidents sont observés chez les patients ayant de faibles valeurs d'hémoglobine (≤ 13 g / dL).
Traitement des patients adultes devant subir une chirurgie orthopédique majeure élective
Chez les patients devant subir une chirurgie orthopédique majeure programmée avec des taux d'hémoglobine avant traitement > 10 et ≤ 13 g/dL, il a été démontré que l'époétine alfa réduit le risque de recevoir des transfusions allogéniques et accélère la récupération érythroïde (augmentation des taux d'hémoglobine, des taux d'hématocrite et du nombre de réticulocytes).
Efficacité et sécurité cliniques
L'insuffisance rénale chronique
L'époétine alfa a fait l'objet d'études cliniques chez des patients adultes anémiques atteints d'insuffisance rénale chronique, y compris des patients en hémodialyse et en pré-dialyse, pour le traitement de l'anémie et le maintien de l'hématocrite sur une plage de concentrations cibles de 30 et 36 %.
Dans les essais cliniques avec des doses initiales allant de 50 à 150 UI/kg trois fois par semaine, environ 95 % des patients ont répondu par une augmentation cliniquement significative de l'hématocrite. Après environ deux mois de traitement, presque tous les patients étaient indépendants des transfusions. l'hématocrite cible a été atteint, la dose d'entretien a été fixée individuellement pour chaque patient.
Dans les trois plus grands essais cliniques menés chez des patients dialysés adultes, la dose d'entretien médiane nécessaire pour maintenir l'hématocrite entre 30 et 36 % était d'environ 75 UI/kg administrée 3 fois par semaine.
Dans une étude multicentrique, en double aveugle, contrôlée par placebo, sur la qualité de vie chez des patients insuffisants rénaux chroniques sous hémodialyse, il y a eu une amélioration cliniquement et statistiquement significative chez les patients traités par époétine alfa par rapport au placebo en termes de fatigue, de symptômes physiques, de relations et de dépression (questionnaire sur la maladie rénale) après six mois de traitement. Les patients du groupe époétine alfa ont également été inclus dans une étude d'extension en ouvert dans laquelle des améliorations de la qualité de vie ont été démontrées et maintenues pendant 12 mois supplémentaires.
Patients adultes insuffisants rénaux non encore dialysés
Dans les études cliniques menées chez des patients insuffisants rénaux chroniques non dialysés et traités par époétine alfa, la durée moyenne du traitement était de près de cinq mois. Ces patients ont répondu au traitement par époétine alfa d'une manière similaire à celle observée chez les patients dialysés. Chez les patients insuffisants rénaux chroniques non dialysés, une augmentation prolongée et dose-dépendante de l'hématocrite a été observée suite à l'administration intraveineuse ou sous-cutanée d'époétine alfa. Les taux d'augmentation de l'hématocrite étaient similaires avec les deux voies d'administration d'époétine alfa. Il a été démontré que l'époétine alfa entre 75 et 150 UI / kg par semaine maintient l'hématocrite à des valeurs comprises entre 36 et 38 % jusqu'à six mois.
Dans deux études avec des intervalles de dosage prolongés pour l'époétine alfa (3 fois par semaine, une fois par semaine, une fois toutes les 2 semaines et une fois toutes les 4 semaines), certains patients avec des intervalles de dosage plus longs n'ont pas maintenu des taux d'hémoglobine adéquats et ont répondu aux critères de retrait du protocole pour l'hémoglobine. (0 % dans le groupe une fois par semaine, 3,7 % dans le groupe une fois toutes les 2 semaines et 3,3 % dans le groupe une fois toutes les 4 semaines).
Dans une étude prospective randomisée, 1 432 patients anémiques atteints d'insuffisance rénale chronique non dialysés ont été évalués. Les patients ont été assignés à un traitement par époétine alfa pour maintenir un taux d'hémoglobine de 13,5 g/dL (au-dessus du niveau de concentration d'hémoglobine recommandé) ou 11,3 g/dL. Un événement cardiovasculaire majeur (décès, infarctus du myocarde, accident vasculaire cérébral ou hospitalisation pour insuffisance cardiaque congestive) est survenu chez 125 (18 %) des 715 patients du groupe présentant des taux d'hémoglobine plus élevés par rapport à 97 (14 %) des 717 patients du groupe avec des taux d'hémoglobine inférieurs (rapport de risque [HR] 1,3 ; IC à 95 % : 1,0, 1,7 ; p = 0,03).
Des analyses post-hoc groupées d'études cliniques sur les ASE chez des patients atteints d'insuffisance rénale chronique (sous dialyse, pas sous dialyse, diabétiques et non diabétiques) ont été menées. Une tendance a été observée à une augmentation du risque estimé de mortalité toutes causes confondues et d'événements cardiovasculaires et cérébrovasculaires associés à des doses cumulées plus élevées d'ASE, indépendamment de la présence ou de l'absence de diabète ou de dialyse (voir rubriques 4.2 et rubrique 4.4).
Traitement des patients atteints d'anémie induite par la chimiothérapie
L'époétine alfa a fait l'objet d'études cliniques chez des patients atteints d'un cancer anémique présentant des tumeurs lymphoïdes et solides et chez des patients suivant divers schémas de chimiothérapie, y compris des schémas avec ou sans platine. Dans ces études, il a été démontré que l'époétine alfa, administrée 3 fois par semaine et par semaine, augmente l'hémoglobine et réduit le besoin de transfusions après le premier mois de traitement chez les patients atteints d'un cancer anémique. Dans certaines études, la phase en double aveugle a été suivie d'une phase en ouvert au cours de laquelle tous les patients ont reçu de l'époétine alfa et le l'effet a été observé.
Les preuves disponibles indiquent que les patients atteints d'hémopathies malignes et de tumeurs solides répondent de manière équivalente au traitement par époétine alfa et que les patients avec ou sans infiltration tumorale de la moelle osseuse répondent de manière équivalente au traitement par époétine alfa. L'intensité similaire de la chimiothérapie dans les groupes époétine alfa et placebo dans les études de chimiothérapie a été démontrée par une « aire sous la courbe du temps des neutrophiles similaire chez les patients traités par époétine alfa et chez les patients traités par placebo, ainsi que par une proportion similaire de patients des groupes époétine alfa et placebo dont le nombre absolu de neutrophiles était inférieur à 1 000 et 500 cellules/mcL.
Dans une étude prospective, randomisée, en double aveugle, contrôlée contre placebo menée auprès de 375 patients anémiques atteints de diverses tumeurs malignes non myéloïdes et traités par chimiothérapie sans platine, une réduction significative des séquelles liées à l'anémie (fatigue) a été observée. , énergie et activité réduite) sur la base des mesures instrumentales et des échelles d'évaluation suivantes : échelle FACT-An (General Functional Assessment of Cancer Therapy-Anemia), FACT-An fatigue scale et Cancer Linear Analogue Scale (CLAS). -études contrôlées, aucune amélioration significative des paramètres de qualité de vie n'a été observée sur les échelles EORTC-QLQ-C30 ou CLAS, respectivement.
La survie et la progression tumorale ont été analysées dans cinq grandes études contrôlées portant sur un total de 2 833 patients, dont quatre études en double aveugle contrôlées par placebo et une étude en ouvert. Ces études ont inclus des patients sous chimiothérapie (deux études) ou des populations de patients pour lesquelles les ASE ne sont pas indiqués : patients cancéreux anémiques ne subissant pas de chimiothérapie et patients présentant un cancer de la tête et du cou subissant une radiothérapie. Dans deux études, le niveau de concentration d'hémoglobine souhaité était > 13 g/dL (8,1 mmol/L) ; dans les autres études, elle variait de 12 à 14 g/dL (7,5 à 8,7 mmol/L). Dans l'étude ouverte, il n'y avait pas de différence dans la survie globale des patients traités par l'érythropoïétine humaine recombinante et les témoins.Dans les quatre études contrôlées contre placebo, le risque relatif de survie globale variait de 1,25 à 2,47, en faveur des témoins. Par rapport aux témoins, ces études ont observé une augmentation statistiquement significative, constante et inexpliquée de la mortalité chez les patients atteints d'anémie associée à plusieurs tumeurs malignes courantes et traités par l'érythropoïétine humaine recombinante. Le résultat global de la survie n'était pas suffisamment expliqué par les différences d'incidence de thrombose et de complications associées chez les sujets traités par érythropoïétine humaine recombinante et chez les sujets témoins.
Une « analyse des données individuelles des patients a également été menée chez plus de 13 900 patients atteints de cancer (recevant une chimiothérapie, une radiothérapie, une chimio-radiothérapie ou ne subissant aucun traitement) participant à 53 essais cliniques contrôlés impliquant différentes époétines. La méta-analyse de la survie globale des patients. les données ont fourni une estimation ponctuelle du rapport de risque (Taux de dangerosité) de 1,06 en faveur des témoins (IC à 95 % : 1,00 ; 1,12 ; 53 études et 13 933 patients) et pour les patients cancéreux traités par chimiothérapie, le risque relatif pour la survie globale était de 1,04 (IC à 95 % : 0,97 ; 1,11 ; 38 études et 10 441 les patients). Les méta-analyses ont également montré systématiquement une augmentation significative du risque relatif d'événements thromboemboliques chez les patients cancéreux traités par l'érythropoïétine humaine recombinante (voir rubrique 4.4).
Une étude ouverte, randomisée et multicentrique a été menée auprès de 2 098 femmes atteintes d'anémie avec cancer du sein métastatique qui ont reçu une chimiothérapie de première ou de deuxième ligne. Il s'agissait d'une étude de non-infériorité conçue pour exclure une augmentation de 15 % du risque de progression tumorale ou de décès pour l'époétine alfa plus le traitement standard (SOC) par rapport au SOC seul. Survie médiane sans progression (survie sans progression, PFS) selon l'évaluation de l'investigateur de la progression de la maladie était de 7,4 mois dans chaque bras (HR 1,09, IC à 95 % : 0,99, 1,20), indiquant que l'objectif de l'étude n'a pas été atteint couper 1337 décès ont été signalés. La survie globale médiane dans le groupe recevant l'époétine alfa plus le SOC était de 17,2 mois contre 17,4 mois dans le groupe recevant le SOC seul (HR 1,06, IC à 95 % : 0,95, 1,18). Dans le bras recevant époétine alfa plus SOC, significativement moins de patients ont reçu des transfusions d'érythrocytes (5,8 % contre 11,4 %) ; cependant, dans le bras recevant époétine alfa plus SOC, un nombre significativement plus élevé de patients (2,8 % contre 1,4 %) ont présenté des événements vasculaires thrombotiques.
Programme de prédon autologue
L'effet de l'époétine alfa sur la facilitation du don de sang autologue chez les patients à faible hématocrite (≤ 39 % en l'absence d'anémie ferriprive sous-jacente) en attente d'une chirurgie orthopédique majeure a été évalué dans une étude en double aveugle contre placebo menée dans 204 patients et une étude en simple aveugle contrôlée contre placebo menée chez 55 patients.
Dans l'étude en double aveugle, les patients ont été traités par l'époétine alfa 600 UI/kg ou un placebo par voie intraveineuse une fois par jour tous les 3 ou 4 jours pendant 3 semaines (pour un total de 6 doses). En moyenne, les patients traités par époétine alfa ont pu donner significativement plus d'unités de sang pour le prédépôt (4,5 unités) que les patients traités par placebo (3,0 unités).
Dans l'étude en simple aveugle, les patients ont été traités par l'époétine alfa 300 UI/kg ou 600 UI/kg ou un placebo par voie intraveineuse une fois par jour tous les 3 ou 4 jours pendant 3 semaines (pour un total de 6 doses). Ces patients traités par époétine alfa ont également pu donner significativement plus d'unités de sang au prédépôt (époétine alfa 300 UI/kg = 4,4 unités ; époétine alfa 600 UI/kg = 4,7 unités) par rapport aux patients traités par placebo (2,9 unités ).
Le traitement par époétine alfa a réduit le risque d'exposition au sang allogénique de 50 % par rapport aux patients ne recevant pas d'époétine alfa.
Chirurgie orthopédique majeure élective
L'effet de l'époétine alfa (300 UI/kg ou 100 UI/kg) sur l'exposition aux transfusions sanguines allogéniques a été évalué dans une étude clinique en double aveugle, contrôlée versus placebo chez des patients adultes non ironopéniques en attente d'une chirurgie orthopédique majeure élective sur la hanche ou le genou. L'époétine alfa a été administrée par voie sous-cutanée dans les 10 jours précédant la chirurgie, le jour de la chirurgie et pendant les quatre jours suivant la chirurgie. Les patients ont été stratifiés en fonction de l'hémoglobine de base (≤10 g/dL,> 10 à ≤13 g/dL, et> 13 g/dL).
L'époétine alfa 300 UI/kg a significativement réduit le risque de transfusion allogénique chez les patients ayant un taux d'hémoglobine avant traitement allant de > 10 à ≤ 13 g/dL. Seize pour cent des patients traités par époétine alfa 300 UI/kg, 23 % des patients traités par époétine alfa 100 UI/kg et 45 % des patients traités par placebo ont nécessité des transfusions.
Dans une étude ouverte en groupes parallèles chez des sujets adultes non carencés en fer avec un taux d'hémoglobine avant traitement compris entre ≥10 et ≤13 g/dL en attente d'une chirurgie orthopédique majeure de la hanche ou du genou, l'époétine alfa a été comparée. dans les 10 jours précédant l'intervention chirurgicale, le jour de l'intervention chirurgicale et dans les quatre jours suivant l'intervention chirurgicale avec de l'époétine alfa 600 UI/kg par voie sous-cutanée une fois par semaine pendant les 3 semaines précédant l'intervention chirurgicale et le jour de l'intervention.
De la phase pré-traitement à la phase pré-opératoire, l'augmentation moyenne de l'hémoglobine dans le groupe 600 UI/kg par semaine (1,44 g/dL) était le double de celle du groupe 300 UI/dL kg par jour (0,73 g /dL). Les taux moyens d'hémoglobine étaient similaires dans les deux groupes de traitement tout au long de la période postopératoire.
La réponse érythropoïétique observée dans les deux groupes de traitement a entraîné des taux de transfusion similaires (16 % dans le groupe 600 UI/kg par semaine et 20 % dans le groupe 300 UI/kg par jour).
Population pédiatrique
L'insuffisance rénale chronique
L'époétine alfa a été évaluée dans une étude clinique ouverte, non randomisée, à échelle ouverte, de 52 semaines chez des patients pédiatriques atteints d'insuffisance rénale chronique sous hémodialyse. L'âge médian des patients inclus dans l'étude était de 11,6 ans (intervalle de 0,5 à 20,1 ans). années).
L'époétine alfa a été administrée à des doses de 75 UI/kg/semaine par voie intraveineuse, réparties en 2 ou 3 prises après dialyse, titrées à 75 UI/kg/semaine à 4 semaines d'intervalle (jusqu'à un maximum de 300 UI/kg/semaine) pour obtenir une augmentation de l'hémoglobine de 1 g/dL/mois. La plage de concentration d'hémoglobine souhaitée était de 9,6 à 11,2 g / dL. Quatre-vingt-un pour cent des patients ont atteint ce niveau de concentration d'hémoglobine. Le délai médian jusqu'à l'objectif était de 11 semaines et la dose médiane jusqu'à l'objectif était de 150 UI/kg/semaine. Parmi les patients qui ont atteint l'objectif, 90 % l'ont atteint avec le régime 3 fois par semaine.
Après 52 semaines, 57 % des patients sont restés dans l'étude, recevant une dose médiane de 200 UI/kg/semaine.
Les données cliniques relatives à l'administration sous-cutanée chez l'enfant sont limitées. Dans 5 études ouvertes non contrôlées avec un petit nombre de patients (le nombre de patients variait de 9 à 22, pour un total N = 72), l'époétine alfa a été administrée par voie sous-cutanée à des enfants avec une dose unique à partir de 100 UI/kg /semaine à 150 UI/kg/semaine, avec possibilité de l'augmenter jusqu'à 300 UI/kg/semaine.Dans ces études, la majorité des patients étaient pré-dialysés (N=44), 27 patients étaient sous dialyse péritonéale et deux étaient sous hémodialyse, l'âge des patients variait de 4 mois à 17 ans. Dans l'ensemble, ces études ont des limites méthodologiques, mais le traitement a été associé à des tendances positives vers des taux d'hémoglobine plus élevés. Aucun événement indésirable inattendu n'a été rapporté (voir rubrique 4.2).
Anémie induite par la chimiothérapie
L'époétine alfa 600 UI/kg (administrée par voie intraveineuse ou sous-cutanée une fois par semaine) a été évaluée dans une étude randomisée, en double aveugle, contrôlée versus placebo de 16 semaines et dans une étude randomisée, contrôlée, en ouvert et d'une durée de 20 semaines chez des patients pédiatriques atteints de anémie subissant une chimiothérapie myélosuppressive pour le traitement de diverses tumeurs malignes non myéloïdes infantiles.
Dans l'étude de 16 semaines (n = 222), chez les patients traités par époétine alfa, aucun effet significatif sur la qualité de vie des patients pédiatriques n'a été rapporté par les patients eux-mêmes ou par leurs parents ou dans les scores du module Cancer par rapport au placebo ( Par ailleurs, il n'y avait pas de différence statistique entre le pourcentage de patients ayant nécessité des transfusions de globules rouges dans le groupe recevant l'époétine alfa et celui recevant le placebo.
Dans l'étude de 20 semaines (n = 225), il n'y avait pas de différence significative sur le critère principal d'efficacité, c'est-à-dire dans la proportion de patients ayant nécessité une transfusion de globules rouges après le 28e jour (62 % des patients ayant reçu de l'époétine alfa vs 69 % de patients recevant un traitement standard).
05.2 "Propriétés pharmacocinétiques -
Absorption
Après injection sous-cutanée, les concentrations sériques d'époétine alfa ont atteint un pic entre 12 et 18 heures après l'administration de la dose. Il n'y a pas eu d'accumulation après administration de doses multiples de 600 UI/kg par voie sous-cutanée une fois par semaine.
La biodisponibilité absolue de l'époétine alfa injectable par voie sous-cutanée est d'environ 20 % chez les sujets sains.
Distribution
Le volume moyen de distribution était de 49,3 mL/kg après des doses intraveineuses de 50 et 100 UI/kg chez des sujets sains. Après administration intraveineuse d'époétine alfa chez des sujets atteints d'insuffisance rénale chronique, le volume de distribution variait de 57 à 107 mL/kg après doses uniques (12 UI/kg) et de 42 à 64 mL/kg après doses multiples, respectivement (48 -192 UI/kg). Par conséquent, le volume de distribution est légèrement supérieur à l'espace plasma.
Élimination
La demi-vie de l'époétine alfa après administration intraveineuse de doses multiples est d'environ 4 heures chez des sujets sains.
La demi-vie après administration sous-cutanée est estimée à environ 24 heures chez les sujets sains.
La CL/F moyenne pour les schémas thérapeutiques de 150 UI/kg 3 fois par semaine et 40 000 UI une fois par semaine chez les sujets sains était respectivement de 31,2 et 12,6 mL/h/kg. La CL/F moyenne pour les schémas thérapeutiques de 150 UI/kg 3 fois par semaine et 40 000 UI une fois par semaine chez les sujets anémiques cancéreux était de 45,8 et 11,3 mL/h/kg, respectivement. Chez la plupart des sujets cancéreux anémiques subissant une chimiothérapie cyclique, la CL/F était plus faible après des doses sous-cutanées de 40 000 UI une fois par semaine et de 150 UI/kg 3 fois par semaine par rapport aux valeurs observées chez les sujets sains.
Linéarité / Non-linéarité
Chez les sujets sains, une augmentation proportionnelle à la dose des concentrations sériques d'époétine alfa a été observée après administration intraveineuse de 150 et 300 UI/kg 3 fois par semaine. L'administration de doses uniques comprises entre 300 et 2 400 UI/kg d'époétine alfa par voie sous-cutanée a entraîné une corrélation linéaire entre la Cmax moyenne et la dose et entre l'ASC moyenne et la dose. Une corrélation inverse entre la clairance apparente et la dose a été observée chez les sujets sains.
Dans les études visant à examiner l'allongement de l'intervalle posologique (40 000 UI une fois par semaine et 80 000, 100 000 et 120 000 UI toutes les deux semaines), une corrélation linéaire, mais non proportionnelle à la dose, a été observée entre la Cmax moyenne et la dose et entre l'ASC moyenne et la dose à l'état d'équilibre.
Relations pharmacocinétiques / pharmacodynamiques
L'époétine alfa présente un effet dose-dépendant sur les paramètres hématologiques, indépendant de la voie d'administration.
Population pédiatrique
Une demi-vie d'environ 6,2 à 8,7 heures a été observée chez des sujets pédiatriques atteints d'insuffisance rénale chronique après administration intraveineuse de doses multiples d'époétine alfa.Le profil pharmacocinétique de l'époétine alfa chez les enfants et les adolescents semble similaire à celui des adultes.
Les données pharmacocinétiques chez les nouveau-nés sont limitées.
Une étude portant sur 7 nouveau-nés prématurés de très faible poids de naissance et 10 adultes en bonne santé ayant reçu de l'érythropoïétine par voie intraveineuse. ont suggéré que le volume de distribution était environ 1,5 à 2 fois plus élevé chez les prématurés que chez les adultes en bonne santé et que la clairance était environ 3 fois plus élevée chez les prématurés que chez les adultes en bonne santé.
Insuffisance rénale
Chez les patients atteints d'insuffisance rénale chronique, la demi-vie de l'époétine alfa administrée par voie intraveineuse est légèrement plus longue, environ 5 heures, par rapport aux sujets sains.
05.3 Données de sécurité préclinique -
Dans les études de toxicologie à doses répétées chez le chien et le rat, mais pas chez le singe, le traitement par époétine alfa a été associé à une fibrose médullaire subclinique. La fibrose médullaire est une complication connue de l'insuffisance rénale chronique chez l'homme et peut être liée à une hyperparathyroïdie secondaire ou à des facteurs inconnus. groupe témoin compatible de patients dialysés non traités par époétine alfa.
L'époétine alfa n'induit pas de mutation génique chez les bactéries (test d'Ames), d'aberrations chromosomiques dans les cellules de mammifères, de micronoyaux chez la souris ou de mutation génique au locus HGPRT.
Aucune étude de cancérogénicité à long terme n'a été menée. Données discordantes existant dans la littérature, basées sur les résultats obtenus in vitro avec des échantillons de tumeurs humaines, suggèrent un rôle possible des érythropoïétines dans la prolifération tumorale. La signification clinique est incertaine.
Dans les cultures de cellules de moelle osseuse humaine, l'époétine alfa stimule spécifiquement l'érythropoïèse et n'affecte pas la leucopoïèse. Il n'y a pas eu d'effet cytotoxique de l'époétine alfa sur les cellules de la moelle osseuse.
Dans les études animales, il a été démontré que l'époétine alfa induit une réduction du poids corporel du fœtus, un retard de l'ossification et une augmentation de la mortalité fœtale lorsqu'elle est administrée à des doses hebdomadaires environ 20 fois supérieures à la dose hebdomadaire recommandée chez l'homme. gain de poids corporel réduit et leur importance pour les humains est inconnue aux niveaux de dose thérapeutique.
06.0 INFORMATIONS PHARMACEUTIQUES -
06.1 Excipients -
Phosphate monosodique dihydraté
Phosphate disodique dihydraté
Chlorure de sodium
Glycine
Polysorbate 80
Eau pour préparations injectables
Acide chlorhydrique (pour ajuster le pH)
Hydroxyde de sodium (pour ajuster le pH)
06.2 Incompatibilité "-
En l'absence d'études de compatibilité, ce médicament ne doit pas être mélangé avec d'autres médicaments.
06.3 Durée de validité "-
2 ans
06.4 Précautions particulières de conservation -
A conserver et transporter réfrigéré (2°C - 8°C). Cette plage de température doit être strictement respectée jusqu'à l'administration au patient.
Pour un usage ambulatoire, le médicament peut être sorti du réfrigérateur, sans le remettre, pendant une durée maximale de 3 jours à une température n'excédant pas 25°C. Si le médicament n'a pas été utilisé à l'issue de cette période, il doit être jeté. .
Ne pas congeler ni secouer.
Conserver dans l'emballage d'origine pour protéger le médicament de la lumière.
06.5 Nature du conditionnement primaire et contenu de l'emballage -
Seringues préremplies (verre de type I), avec ou sans dispositif de sécurité pour aiguille, avec bouchon piston (caoutchouc Téflon) scellées sous blister.
Binocrit 1000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,5 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 2 000 UI/1 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 1 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 3000 UI/0,3 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,3 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 4000 UI/0,4 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,4 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 5 000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,5 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 6 000 UI / 0,6 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,6 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 7 000 UI/0,7 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,7 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 8 000 UI / 0,8 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,8 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 9 000 UI/0,9 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,9 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 10 000 UI/1 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 1 ml de solution injectable.
Boîtes de 1 ou 6 seringues.
Binocrit 20 000 UI / 0,5 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,5 ml de solution injectable.
Boîtes de 1, 4 ou 6 seringues.
Binocrit 30 000 UI/0,75 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 0,75 ml de solution injectable.
Boîtes de 1, 4 ou 6 seringues.
Binocrit 40 000 UI/1 mL solution injectable en seringue préremplie
Chaque seringue préremplie contient 1 ml de solution injectable.
Boîtes de 1, 4 ou 6 seringues.
Toutes les présentations peuvent ne pas être commercialisées.
06.6 Instructions d'utilisation et de manipulation -
Binocrit ne doit pas être utilisé et doit être jeté
• si le liquide est coloré ou si vous voyez des particules flotter dedans
• si le sceau est brisé
• s'il est connu ou supposé qu'il peut avoir été accidentellement congelé ou
• si une panne de réfrigérateur s'est produite
Les seringues préremplies sont prêtes à l'emploi (voir rubrique 4.2). La seringue préremplie ne doit pas être agitée. Les seringues sont marquées de graduations en relief, ce qui permet une utilisation partielle si nécessaire. Chaque graduation correspond à un volume de 0, 1 mL. Le produit est à usage unique. Ne prélever qu'une dose de Binocrit de chaque seringue et jeter toute solution inutile avant l'injection.
Utilisation de la seringue préremplie avec protection de l'aiguille
La protection de sécurité de l'aiguille recouvre l'aiguille après l'injection et empêche l'opérateur de se blesser. Le dispositif n'interfère pas avec l'utilisation normale de la seringue. Appuyez lentement et uniformément sur le piston jusqu'à ce que la dose complète soit libérée et que le piston ne puisse plus être poussé. Tout en continuant d'appuyer sur le piston, éloignez la seringue du patient. Le dispositif de sécurité recouvre l'aiguille dès que le piston est relâché.
Utilisation de la seringue préremplie sans protection de l'aiguille
Administrer la dose selon la procédure standard.
Les médicaments non utilisés et les déchets dérivés de ce médicament doivent être éliminés conformément aux réglementations locales.
07.0 TITULAIRE DE « L'AUTORISATION DE MISE SUR LE MARCHE » -
Sandoz GmbH
Biochimie dix
A-6250 Kundl
L'Autriche
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ -
Binocrit 1000 UI / 0,5 mL solution injectable en seringue préremplie
EU/1/07/410/001 - 1000 UI/0,5 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,5 ml (2000 UI/ml) 1 seringue préremplie - AIC n. 038190017 / F
EU/1/07/410/002 - 1000 UI/0,5 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,5 ml (2000 UI/ml) 6 seringues préremplies - AIC n. 038190029 / E
EU/1/07/410/027 - 1000 UI/0,5 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,5 ml (2000 UI/ml) 1 seringue préremplie de 0,5 ml avec dispositif de sécurité pour aiguille - AIC n. 038190272 / E
EU/1/07/410/028 - 1000UI/0,5 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,5 ml (2000 UI/ml) - 6 seringues préremplies de 0,5 ml avec aiguille pour dispositif sécurité - AIC N. 038190284 / E
Binocrit 2 000 UI/1 mL solution injectable en seringue préremplie
EU/1/07/410/003 - 2000 UI/1,0 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 1,0 ml (2000 UI/ml) 1 seringue préremplie - AIC n. 038190031 / E
EU/1/07/410/004 - 2000 UI/1,0 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 1,0 ml (2000 UI/ml) 6 seringues préremplies - AIC n. 038190043 / E
EU/1/07/410/029 - 2000UI/1ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 1ml (2000UI/ml) 1 seringue préremplie de 1ml avec dispositif de sécurité pour aiguille - AIC N. 038190296 / E
EU/1/07/410/030- 2000UI/0,5 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 1 ml (2000 UI/ml) - 6 seringues préremplies de 1 ml avec aiguille de sécurité appareil - AIC N. 038190308 / E
Binocrit 3000 UI/0,3 mL solution injectable en seringue préremplie
EU/1/07/410/005 - 3000 UI/0,3 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,3 ml (10000 UI/ml) 1 seringue préremplie - AIC n. 038190056 / E
EU/1/07/410/006 - 3000 UI/0,3 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,3 ml (10000 UI/ml) 6 seringues préremplies - AIC n. 038190068 / E
EU/1/07/410/031- 3000UI/0,3ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,3ml (10000UI/ml) 1 seringue préremplie de 0,3ml avec dispositif de sécurité par aiguille - N° AIC 038190310 / E
EU/1/07/410/032 - 3000UI/0,3ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,3ml (10000 UI/ml) - 6 seringues préremplies de 0,3ml avec dispositif aiguille de sécurité - AIC N° 038190322 / E
Binocrit 4000 UI/0,4 mL solution injectable en seringue préremplie
EU/1/07/410/007 - 4000 UI/0,4 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,4 ml (10000 UI/ml) 1 seringue préremplie - AIC n. 038190070 / E
EU/1/07/410/008 - 4000 UI/0,4 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,4 ml (10000 UI/ml) 6 seringues préremplies - AIC n. 038190082 / E
EU/1/07/410/033 - 4000UI/0,4ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,4ml (10000UI/ml) 1 seringue préremplie de 0,4ml avec dispositif de sécurité par aiguille - N° AIC 038190334 / E
EU/1/07/410/034 - 4000UI/0,4 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,4 ml (10000 UI/ml) - 6 seringues préremplies de 0,4 ml avec dispositif de sécurité aiguille - AIC N. 038190346 / E
Binocrit 5 000 UI / 0,5 mL solution injectable en seringue préremplie
EU/1/07/410/009 - 5000 UI/0,5 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,5 ml (10000 UI/ml) 1 seringue préremplie - AIC n. 038190094 / E
EU/1/07/410/010 - 5000 UI/0,5 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,5 ml (10000 UI/ml) 6 seringues préremplies - AIC n. 038190106 / F
EU/1/07/410/035 - 5000UI/0,5ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,5ml (10000 UI/ml) - 1 seringue préremplie de 0,5ml avec dispositif aiguille de sécurité - N° AIC 038190359 / E
EU/1/07/410/036 - 5000UI/0,5 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,5 ml (10000 UI/ml) - 6 seringues préremplies de 0,5 ml avec dispositif de sécurité pour aiguille -AIC N. 038190361 / E
Binocrit 6 000 UI / 0,6 mL solution injectable en seringue préremplie
EU/1/07/410/011 - 6000 UI/0,6 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,6 ml (10 000 UI/ml) 1 seringue préremplie - AIC n . 038190118 / E
EU/1/07/410/012 - 6000 UI/0,6 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,6 ml (10 000 UI/ml) 6 seringues préremplies - AIC n . 038190120 / E
EU/1/07/410/037 - 6000UI/0,6 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,6 ml (10000 UI/ml) - 1 seringue préremplie de 0,6 ml avec dispositif de sécurité aiguille - AIC N. 038190373 / E
EU/1/07/410/038 - 6000UI/0,6 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,6 ml (10 000 UI/ml) - 6 seringues préremplies de 0 ,6 ml avec dispositif aiguille de sécurité - AIC N. 038190385 / E
Binocrit 7 000 UI/0,7 mL solution injectable en seringue préremplie
EU/1/07/410/017 - 7000UI/0,7 ml solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 0,7 ml (10000UI/ml) 1 seringue préremplie - AIC Non 038190171 / ET
EU/1/07/410/018 - 7000UI/0,7 ml solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 0,7 ml (10000UI/ml) 6 seringues préremplies - AIC Non 038190183 / ET
EU/1/07/410/039 - 7000UI/0,7ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,7ml (10000UI/ml) 1 seringue préremplie de 0,7ml avec dispositif de sécurité par aiguille - AIC N. 038190397 / E
EU/1/07/410/040 - 7000UI/0,7 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,7 ml (10000UI/ml) 6 seringues préremplies de 0,7 ml avec dispositif de sécurité par aiguille - AIC N. 038190409 / E
Binocrit 8 000 UI / 0,8 mL solution injectable en seringue préremplie
EU/1/07/410/013 - 8000 UI / 0,8 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,8 ml (10 000 UI/ml) 1 seringue préremplie - AIC n . 038190132 / E
EU/1/07/410/014 - 8000 UI / 0,8 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 0,8 ml (10 000 UI/ml) 6 seringues préremplies - AIC n . 038190144 / E
EU/1/07/410/041 - 8000UI/0,8 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,8 ml (10000 UI/ml) - 1 seringue préremplie de 0,8 ml avec dispositif de sécurité aiguille - AIC N. 038190411 / E
EU/1/07/410/042 - 8000UI/0,8 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 0,8 ml (10000 UI/ml) - 6 seringues préremplies de 0,8 ml avec dispositif de sécurité aiguille - AIC N. 038190423 / E
Binocrit 9 000 UI/0,9 mL solution injectable en seringue préremplie
EU/1/07/410/019 - 9000UI/0,9 ml solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse seringue préremplie (verre) 0,9 ml (10000UI/ml) 1 seringue préremplie préremplie - N° AIC 038190195 / ET
EU/1/07/410/020 - 9000UI/0,9 ml solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 0,9 ml (10000UI/ml) 6 seringues préremplies - AIC Non 038190207 / ET
EU/1/07/410/043 - 9000UI/0,9 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,9 ml (10000UI/ml) 1 seringue préremplie de 0,9 ml avec dispositif de sécurité par aiguille - AIC N. 038190435 / E
EU/1/07/410/044 - 9000UI/0,9 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,9 ml (10000UI/ml) 6 seringues préremplies de 0,9 ml avec dispositif de sécurité par aiguille - AIC N. 038190447 / E
Binocrit 10 000 UI/1 mL solution injectable en seringue préremplie
EU/1/07/410/015 - 10000 UI/1,0 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 1,0 ml (10000 UI/ml) 1 seringue préremplie - AIC n. 038190157 / E
EU/1/07/410/016 - 10000 UI/1,0 ml solution injectable en seringue préremplie (verre) voie sous-cutanée ou intraveineuse 1,0 ml (10000 UI/ml) 6 seringues préremplies - AIC n. 038190169 / E
EU/1/07/410/045 - 10000UI/1 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 1 ml (10000 UI/ml) - 1 seringue préremplie de 1 ml avec dispositif de sécurité pour aiguille - AIC N. 038190450 / E
EU/1/07/410/046 - 10000UI/1 ml solution injectable en seringue préremplie (verre) - voie intraveineuse sous-cutanée - 1 ml (10000 UI/ml) - 6 seringues préremplies de 1 ml avec dispositif de sécurité pour aiguille - AIC N. 038190462 / E
Binocrit 20 000 UI / 0,5 mL solution injectable en seringue préremplie
EU/1/07/410/021 - 20 000 UI / 0,5 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse 0,5 ml (40 000 UI/ml) 1 seringue préremplie - AIC n. 038190219 / E
EU/1/07/410/022 - 20 000 UI / 0,5 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse 0,5 ml (40 000 UI/ml) 6 seringues préremplies - AIC n. 038190221 / E
EU/1/07/410/047 - 20000UI/0,5ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,5ml (40000UI/ml) 1 seringue préremplie de 0,5ml avec dispositif de sécurité par aiguille - N° AIC 038190474 / E
EU/1/07/410/053 - 20 000 UI / 0,5 ML - solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 0,5 ML (40 000 UI/ML) - 4 préremplies seringues avec dispositif de sécurité pour aiguille AIC : 038190536 / E
EU/1/07/410/048 - 20000UI/0,5ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,5ml (40000UI/ml) 6 seringues préremplies de 0,5ml avec dispositif de sécurité par aiguille - AIC N. 038190486 / E
Binocrit 30 000 UI/0,75 mL solution injectable en seringue préremplie
EU/1/07/410/023 - 30 000 UI/0,75 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse 0,75 ml (40 000 UI/ml) 1 seringue préremplie - AIC n. 038190233 / E
EU/1/07/410/024 - 30 000 UI/0,75 ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse 0,75 ml (40 000 UI/ml) 6 seringues préremplies - AIC n. 038190245 / E
EU/1/07/410/049 - 30000UI/0.75ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0,75ml (40000UI/ml) 1 seringue préremplie de 0,75ml avec dispositif de sécurité pour Août - AIC N. 038190498 / E
UE/1/07/410/054 - 30 000 UI/0,75 ML - solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 0,75 ML (40 000 UI/ML) - 4 préremplies seringues avec dispositif de sécurité pour aiguille AIC : 038190548 / E
EU/1/07/410/050 - 30000UI/0.75ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 0.75ml (40000UI/ml) 6 seringues préremplies de 0.75ml avec dispositif de sécurité par aiguille - AIC N. 038190500 / E
Binocrit 40 000 UI/1 mL solution injectable en seringue préremplie
EU/1/07/410/025 - 40 000 UI/1,0 ml - solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 1,0 ml (40 000 UI/ml) 1 seringue préremplie - AIC n. 038190258 / E
EU/1/07/410/026 - 40 000 UI/1,0 ml - solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 1,0 ml (40 000 UI/ml) 6 seringues préremplies - AIC n. 038190260 / E
EU/1/07/410/051 - 40000UI/1ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 1ml (40000UI/ml) 1 seringue préremplie de 1ml avec dispositif de sécurité pour aiguille - AIC N. 038190512 / E
EU/1/07/410/055 - 40 000 UI / 1,0 ML - solution injectable en seringue préremplie - voie sous-cutanée ou intraveineuse - seringue préremplie (verre) 1,0 ML (40 000 UI/ML) - 4 préremplies seringues avec dispositif aiguille de sécurité pour aiguille AIC : 038190551 / E
EU/1/07/410/052 - 40000UI/1ml solution injectable en seringue préremplie (verre) - voie sous-cutanée ou intraveineuse - 1ml (40000UI/ml) 6 seringues préremplies de 1ml avec dispositif de sécurité pour aiguille - AIC N° 038190524 / E
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION -
Dès la première autorisation : 28 août 2007
Date du dernier renouvellement : 18 juin 2012
10.0 DATE DE REVISION DU TEXTE -
10/2016












.jpg)