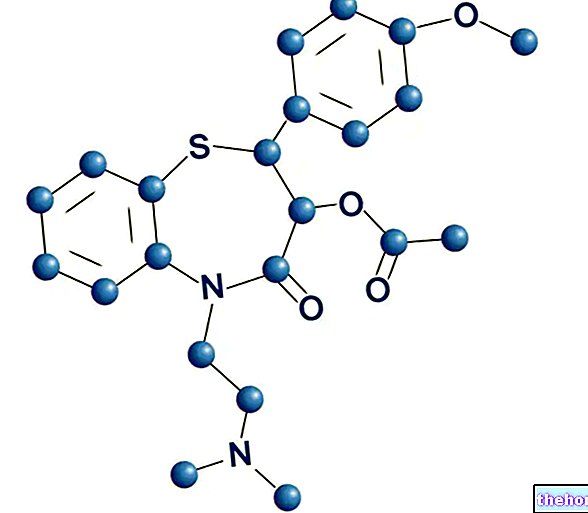
FOSTER ® est un médicament à base de dipropionate de béclométasone et de fumarate de formotérol dihydraté.
GROUPE THÉRAPEUTIQUE : Adrénergiques pour aérosols et autres médicaments pour les syndromes obstructifs des voies respiratoires.

Indications FOSTER ® - Béclométasone + Formotérol
FOSTER ® est indiqué dans le traitement de l'asthme insuffisamment réactif à la corticothérapie seule ou associée à des agonistes B2 de courte durée d'action.
Mécanisme d'action FOSTER ® - Béclométasone + Formotérol
FOSTER ® est un médicament utilisé dans le traitement de l'asthme, composé d'un corticostéroïde Beclometasone et d'un agoniste bêta 2 adrénergique de longue durée d'action, le Formotérol.
Les principes actifs susmentionnés, bien qu'ayant des propriétés biologiques résolument différentes, contribuent au contrôle des symptômes respiratoires obstructifs présents au cours de la maladie asthmatique.
Plus précisément, la Béclométasone, en contrôlant activement la sécrétion de cytokines inflammatoires par la muqueuse respiratoire, réduit le degré d'inflammation des voies respiratoires, entraînant une régression rapide des symptômes obstructifs et congestifs qui caractérisent cette pathologie ; le Formotérol, en revanche, en se liant aux récepteurs bêta 2 adrénergiques, exprimés par les muscles lisses bronchiques, il limite le degré contractile, rétablissant la perméabilité normale des voies respiratoires.
Les caractéristiques pharmacocinétiques différentes des deux principes actifs permettent l'établissement d'une activité biologique en un temps très court, quelques minutes après administration, qui dure plusieurs heures après inhalation.
Études réalisées et efficacité clinique
BECLOMETHASONE FORMOTEROL DANS LA PROTECTION DES DOMMAGES OXYDANTS
Eur J Pharmacol. 15 octobre 2013 ; 718 (1-3) : 418-27. doi: 10.1016 / j.ejphar.2013.08.001. Publication en ligne du 19 août 2013.
Le dipropionate de béclométhasone et le formotérol réduisent le stress oxydatif/nitrosatif généré par les extraits de fumée de cigarette et l'IL-17A dans les cellules épithéliales bronchiques humaines.
Montalbano AM, Anzalone G, Albano GD, Sano CD, Gagliardo R, Bonanno A, Riccobono L, Nicolini G, Ingrassia E, Gjomarkaj M, Profita M.
Étude expérimentale intéressante qui démontre comment l'association entre la béclométasone et le formotérol peut réduire l'activation des mécanismes oxydatifs dans les cellules épithéliales bronchiques soumises à l'exposition à la fumée de cigarette, les protégeant de tout dommage oxydatif.
FORMOTEROL ET BECLOMETASONE EXTRA FINE
Pulm Pharmacol Ther. 14 juin 2013. pii : S1094-553900132-6.
Conception d'étude clinique en vie réelle soutenant l'efficacité de l'inhalation extra-fine de béclométhasone / formotérol au niveau des petites voies respiratoires des asthmatiques.
Popov TA, Petrova D, Kralimarkova TZ, Ivanov Y, Popova T, Peneva M, Odzhakova T, Ilieva Y, Yakovliev P, Lazarova T, Georgiev O, Hodzhev V, Hodzheva E, Staevska MT, Dimitrov VD.
Étude qui démontre comment l'administration de formotérol et de béclométasone en particules extra fines peut déterminer une nette amélioration des symptômes présents dans les conditions d'asthme, résultant en une meilleure capacité de pénétration dans l'épithélium bronchique.
BECLOMETHASONE FORMOTEROL DANS LE TRAITEMENT DE LA BPCO
Int J Chron Obstruct Pulmon Dis. 2011 ; 6 : 503-9. doi: 10.2147 / COPD.S23746. Publication en ligne du 4 octobre 2011
Effets de l'association fixe béclométhasone/formotérol sur l'hyperinflation pulmonaire et la dyspnée chez les patients atteints de BPCO.
Tzani P, Crisafulli E, Nicolini G, Aiello M, Chetta A, Clini EM, Olivieri D.
Un travail intéressant qui démontre comment le traitement d'entretien avec Formotérol et Beclométasone extra fine, peut déterminer une nette amélioration de la dyspnée chez les patients atteints de bronchopneumopathie chronique obstructive, déterminant ainsi une nette amélioration des symptômes.
Mode d'emploi et posologie
FOSTER®
Solution pressurisée pour inhalation de 100 mcg de dipropionate de béclométasone et 6 mcg de fumarate de formotérol dihydraté.
Le schéma thérapeutique varie considérablement d'un patient à l'autre en fonction des caractéristiques de l'état de santé général et de la sévérité de son tableau clinique.
Le médecin doit donc définir cette thérapie de manière très spécifique, en prenant soin de définir la posologie minimale nécessaire pour chaque cas pour assurer un bon contrôle des symptômes.
Dans tous les cas, la posologie maximale ne doit jamais dépasser 4 inhalations quotidiennes.
Avertissements FOSTER ® - Béclométasone + Formotérol
La thérapie avec FOSTER ® doit nécessairement être précédée et supervisée par du personnel médical, afin d'évaluer la pertinence de la prescription, l'efficacité et la sécurité de celui-ci.
Il est également nécessaire de considérer que le traitement par FOSTER ® n'est pas adéquat pour le traitement de la crise d'asthme aiguë.
Une prudence particulière doit évidemment être réservée aux patients souffrant de pathologies cardiovasculaires, de glaucome, d'hyperthyroïdie, de phéochromocytome, de diabète et d'hypertrophie prostatique, pour lesquels une aggravation du tableau clinique pourrait être observée.
Afin de limiter l'apparition d'effets secondaires cliniquement pertinents, il serait conseillé au médecin d'identifier la dose minimale efficace pour assurer le contrôle des symptômes et d'informer le patient de tous les effets secondaires potentiels liés au traitement, afin que le ce dernier peut les reconnaître rapidement et alerter votre médecin à temps.
Bien que l'exposition systémique aux deux principes actifs soit limitée, il serait souhaitable que le médecin vérifie également la survenue éventuelle d'effets indésirables systémiques.
FOSTER ® est interdit en et hors compétition.
Il est recommandé de conserver le médicament hors de la portée des enfants.
LA GROSSESSE ET L'ALLAITEMENT
Les contre-indications susmentionnées à l'utilisation de FOSTER ® s'étendent également à la grossesse et à la période d'allaitement ultérieure, étant donné l'absence d'études capables de caractériser pleinement le profil d'innocuité des deux principes actifs contenus dans le médicament pour la santé du fœtus et du nourrisson.
Interactions
Le patient recevant FOSTER ® doit éviter la prise simultanée de bêta-2 agonistes et de bêtabloquants non sélectifs, ainsi que de diurétiques, de stéroïdes et de dérivés de la xanthine, potentiellement responsables d'hypokaliémie.
Contre-indications FOSTER ® - Béclométasone + Formotérol
L'utilisation de FOSTER® est contre-indiquée chez les patients présentant une hypersensibilité à la substance active ou à l'un de ses excipients, chez les patients présentant une maladie cardiaque sévère, un glaucome, une hypertrophie prostatique et un syndrome de rétention urinaire ou d'obstruction intestinale :
Effets indésirables - Effets secondaires
Le traitement par FOSTER ®, en particulier lorsqu'il est prolongé de manière prolongée ou effectué chez des patients particulièrement sensibles, peut provoquer l'apparition de pharyngites, maux de tête, toux, hypokaliémie, spasmes musculaires, irritation de la gorge, dysphonie et candidose buccale.
Heureusement, l'incidence d'effets secondaires cliniquement plus pertinents tels qu'une éruption cutanée, une hypersensibilité, une urticaire, un œdème de Quincke et des manifestations cardiaques est plus rare.
Noter
FOSTER ® est un médicament soumis à prescription médicale obligatoire.
Les informations sur FOSTER ® - Béclométasone + Formotérol publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.