VEUILLEZ NOTER: MÉDICAMENT N'EST PLUS AUTORISÉ
Qu'est-ce que Teysuno - tegafur / gimeracil / oteracil ?
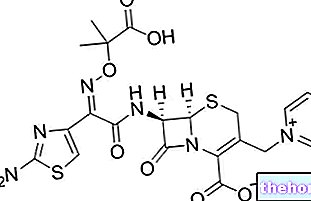
Teysuno est un médicament qui contient les principes actifs tégafur, giméracil et otéracil. Il est disponible sous forme de gélules blanches et brunes contenant 15 mg de tégafur avec 4,35 mg de giméracil et 11,8 mg d'otéracil, et sous forme de gélules blanches contenant 20 mg de tégafur avec 5,8 mg de giméracil et 15,8 mg d'otéracil.
Dans quel cas Teysuno est-il utilisé - tegafur / gimeracil / oteracil?
Teysuno est indiqué chez l'adulte dans le traitement du cancer gastrique (cancer de l'estomac). Le médicament est administré en association avec le cisplatine (un autre médicament anticancéreux).
Le médicament ne peut être obtenu que sur ordonnance.
Comment Teysuno est-il utilisé - tegafur / gimeracil / oteracil ?
Teysuno ne doit être prescrit que par un médecin spécialiste expérimenté dans le traitement des patients cancéreux avec des médicaments anticancéreux.
Teysuno est utilisé en association avec le cisplatine dans un cycle de traitement qui est répété toutes les 4 semaines, en commençant le jour de l'administration du cisplatine. La dose à prendre est calculée en fonction du principe actif tégafur et de la surface corporelle du patient (c'est-à-dire par rapport à la taille et au poids du patient).La dose recommandée pour une cure est de 25 mg/m2 deux fois par jour le matin. et le soir, pendant trois semaines, suivies de 7 jours de repos. Le cycle est répété toutes les 4 semaines, même après l'arrêt de l'administration de cisplatine au bout de six cycles. Les gélules de Teysuno sont prises avec de l'eau au moins une "maintenant avant ou après les repas. Pour plus d'informations sur l'utilisation de Teysuno, y compris sur la manière de le prendre en association avec le cisplatine, veuillez vous référer au Résumé des Caractéristiques du Produit (inclus dans l'EPAR).
Comment fonctionne Teysuno - tegafur / gimeracil / oteracil ?
Le principe actif de Teysuno, le tegafur, est un médicament cytotoxique (une substance qui tue les cellules en division active, telles que les cellules cancéreuses) appartenant au groupe des « antimétabolites ». Le tégafur est un "promédicament" qui est un médicament qui, lorsqu'il est administré dans le corps, se transforme en un produit chimique appelé 5-fluorouracile (5-FU).Le 5-FU est un analogue de la pyrimidine, une substance présente dans le matériel génétique des cellules (ADN et ARN). Dans l'organisme, le 5-FU remplace la pyrimidine et interfère avec les enzymes impliquées dans la synthèse de l'ADN, empêchant ainsi les cellules cancéreuses de se développer jusqu'à ce qu'elles soient détruites.
Les deux autres principes actifs de Teysuno permettent au tegafur d'être efficace à faibles doses et avec moins d'effets secondaires : le giméracil en empêchant la dégradation du 5-FU et l'otéracil en réduisant l'activité du 5-FU dans les tissus intestinaux normaux de nature non naturelle. .tumeur.
Comment a été étudié Teysuno - tegafur / gimeracil / oteracil ?
Les effets de Teysuno ont d'abord été testés dans des modèles expérimentaux avant d'être étudiés chez l'homme. Dans l'étude principale, Teysuno a été comparé au médicament anticancéreux 5-FU administré par perfusion chez 1 053 adultes atteints d'un cancer gastrique avancé. Les deux médicaments ont été administrés en association avec le cisplatine. Le principal critère d'évaluation de l'efficacité était la durée de vie des patients.
Quel bénéfice Teysuno - tegafur / gimeracil / oteracil a-t-il montré au cours des études ?
Le traitement avec les capsules de Teysuno était aussi efficace que la thérapie par perfusion avec le 5-FU. Les patients traités par Teysuno et cisplatine ont survécu en moyenne 8,6 mois contre 7,9 mois pour les patients traités par 5-FU et cisplatine.
Quel est le risque associé à Teysuno - tegafur / giméracil / oteracil ?
Chez les patients traités par Teysuno en association avec le cisplatine, les effets indésirables graves les plus fréquents (observés chez plus de 1 patient sur 10) sont la neutropénie (diminution du nombre de neutrophiles, un type de globules blancs), l'anémie (diminution du nombre de de globules rouges dans le sang) et la fatigue. Pour la liste complète des effets indésirables rapportés avec Teysuno, voir la notice.
Teysuno ne doit pas être utilisé chez les personnes pouvant présenter une hypersensibilité (allergie) au principe actif ou à l'un des autres composants. Teysuno ne doit pas non plus être utilisé dans les groupes de patients suivants :
- les patients prenant un autre médicament à base de fluoropyrimidine (un groupe de médicaments anticancéreux auquel appartient Teysuno) ou qui ont eu des réactions sévères et inattendues aux fluoropyrimidines ;
- les sujets présentant un déficit de l'enzyme dihydropyrimidine déshydrogénase (DPD) et les sujets qui, au cours des quatre dernières semaines, ont été traités par un médicament inhibant cette enzyme ;
- les femmes enceintes ou allaitantes ;
- patients atteints de formes sévères de leucopénie, de neutropénie ou de thrombocytopénie (faibles taux de globules blancs ou de plaquettes dans le sang) ;
patients souffrant de graves problèmes rénaux;
patients pour lesquels il n'est pas recommandé d'utiliser le cisplatine.
Pourquoi Teysuno - tegafur / gimeracil / oteracil a-t-il été approuvé ?
Le CHMP a estimé que les bénéfices de Teysuno sont supérieurs à ses risques et a recommandé l'octroi d'une autorisation de mise sur le marché pour ce médicament.
Plus d'informations sur Teysuno - tegafur / gimeracil / oteracil
Le 14 mars 2011, la Commission européenne a accordé à Taiho Pharma Europe Limited une « Autorisation de mise sur le marché » pour Teysuno, valable dans toute l'Union européenne. L'« Autorisation de mise sur le marché » est valable cinq ans, après quoi elle peut être renouvelée.
Dernière mise à jour de ce résumé : 01-2011.
Les informations sur Teysuno - tegafur / gimeracil / oteracil publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.



.jpg)