Qu'est-ce que Tesavel ?
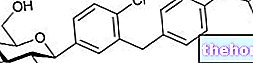
Tesavel est un médicament qui contient le principe actif sitagliptine. Il se présente sous forme de comprimés ronds (rose : 25 mg ; beige : 50 et 100 mg).
A quoi sert Tesavel ?
Tesavel est utilisé chez les patients atteints de diabète de type 2 pour améliorer le contrôle de la glycémie (sucre). Il est utilisé comme suit, en plus du régime alimentaire et de l'exercice :
• seul, chez les patients dont l'équilibre alimentaire et l'exercice ne sont pas satisfaisants et pour lesquels la metformine (un médicament antidiabétique) ne convient pas ;
• en association avec la metformine ou un agoniste du PPAR gamma (un type d'antidiabétique), tel qu'une thiazolidinedione, chez les patients dont le contrôle n'est pas satisfaisant avec la metformine ou l'agoniste du PPAR gamma utilisé seul ;
• en association avec un sulfamide hypoglycémiant (un autre type de médicament antidiabétique) chez les patients dont le contrôle n'est pas satisfaisant sous sulfamide hypoglycémiant seul et pour lesquels la metformine n'est pas adaptée ;
• en association à la fois avec la metformine et un sulfamide hypoglycémiant ou un agoniste du PPAR gamma, chez les patients dont le contrôle n'est pas satisfaisant avec ces deux médicaments ;
• en association avec l'insuline, avec ou sans metformine, chez les patients dont le contrôle n'est pas satisfaisant aux doses stables d'insuline.
Le médicament ne peut être obtenu que sur ordonnance.
Comment Tesavel est-il utilisé ?
Tesavel est pris à une dose de 100 mg une fois par jour, avec ou sans nourriture. Si Tesavel doit être pris en association avec un sulfamide hypoglycémiant ou de l'insuline, il peut être nécessaire de réduire la dose du sulfamide hypoglycémiant ou de l'insuline afin de réduire le risque d'hypoglycémie (faible taux de sucre dans le sang).
Comment fonctionne Tesavel ?
Le diabète de type 2 est une maladie dans laquelle le pancréas ne produit pas suffisamment d'insuline pour contrôler le taux de glucose dans le sang ou dans laquelle le corps est incapable d'utiliser l'insuline efficacement. Le principe actif de Tesavel, la sitagliptine, est un inhibiteur de la dipeptidyl-peptidase-4 (DPP-4). Il agit en inhibant la dégradation des hormones "incrétines" dans le corps. Ces hormones, qui sont libérées après un repas, stimulent le pancréas à produire de l'insuline. En augmentant le niveau d'incrétines dans le sang, la sitagliptine stimule le pancréas à produire plus d'insuline. lorsque le taux glycémique est élevé, alors qu'elle est inefficace lorsque la concentration de glucose dans le sang est faible.La sitagliptine réduit également la quantité de glucose produite par le foie en augmentant les niveaux d'insuline et en diminuant les niveaux de l'hormone glucagon. Ensemble, ces processus réduisent le taux de glucose dans le sang et contribuent au contrôle du diabète de type 2.
Comment Tesavel a-t-il été étudié ?
Tesavel a fait l'objet de neuf études portant sur près de 6 000 patients atteints de diabète de type 2 dont la glycémie n'était pas suffisamment contrôlée :
• dans quatre de ces études, Tesavel a été comparé à un placebo (un traitement fictif). Tesavel ou le placebo ont été utilisés seuls dans deux études portant sur 1 262 patients, en complément de la metformine dans une étude portant sur 701 patients et en complément de la pioglitazone (un agoniste du PPAR gamma) dans une étude portant sur 353 patients ;
• Tesavel a été comparé à d'autres médicaments antidiabétiques dans deux études. Dans la première étude, Tesavel a été comparé au glipizide (un sulfamide hypoglycémiant), lorsqu'il a été utilisé en complément de la metformine chez 1 172 patients. Dans la deuxième étude, Tesavel a été comparé à la metformine,
utilisé seul, chez 1 058 patients ;
• dans trois autres études, Tesavel a été comparé à un placebo lorsqu'ils ont été ajoutés à d'autres
médicaments antidiabétiques : au glimépiride (autre sulfamide hypoglycémiant), avec ou sans metformine, chez 441 patients ; à l'association metformine et rosiglitazone (un agoniste du PPAR gamma) chez 278 patients ; et un dosage stable d'insuline, avec ou sans metformine, chez 641 patients.
Dans toutes les études, le principal critère d'évaluation de l'efficacité était la modification de la concentration sanguine d'une substance appelée hémoglobine glycosylée (HbA1c), qui donne une indication du degré de contrôle de la glycémie.
Quel bénéfice Tesavel a-t-il démontré au cours des études ?
Tesavel s'est avéré plus efficace que le placebo à la fois seul et en association avec d'autres médicaments antidiabétiques. Chez les patients prenant Tesavel seul, les taux d'HbA1c ont diminué de 0,48 % (d'environ 8,0 % au début des études) après 18 semaines et de 0,61 % après 24 semaines. A l'inverse, ils ont augmenté de 0,12 % et 0,18 %, respectivement, chez les patients sous placebo. L'ajout de Tesavel à la metformine a réduit les taux d'HbA1c de 0,67% après 24 semaines, par rapport à une réduction de 0,02% chez les patients ayant reçu un placebo. L'ajout de Tesavel à la pioglitazone a réduit les taux d'HbA1c de 0,85% après 24 semaines, contre une réduction de 0,15% chez les patients placebo ajouté.
Dans les études où Tesavel a été comparé à d'autres médicaments, l'efficacité de l'ajout de Tesavel à la metformine était similaire à celle de l'ajout de glipizide. Lorsqu'ils sont pris seuls, Tesavel et la metformine ont obtenu des réductions similaires des taux d'HbA1c. que celui de la metformine.
Dans les autres études, lorsque Tesavel a été ajouté au glimépiride (avec ou sans metformine), les taux d'HbA1c ont diminué de 0,45% après 24 semaines, contre une augmentation de 0,28% chez les patients ayant reçu un placebo. Les taux d'HbA1c ont diminué de 1,03 % après 18 semaines chez les patients ajoutant Tesavel à la metformine et à la rosiglitazone, contre une diminution de 0,31 % chez ceux ajoutant le placebo ; enfin, ils ont diminué de 0,59 % chez les patients ayant ajouté Tesavel (avec ou sans metformine) à l'insuline , par rapport à une diminution de 0,03 % chez ceux qui ont ajouté un placebo.
Quel est le risque associé à Tesavel ?
Les effets indésirables les plus fréquemment observés avec Tesavel (généralement observés chez plus de 5 % des patients) sont les infections des voies respiratoires supérieures (rhume) et la rhinopharyngite (inflammation du nez et de la gorge). Pour la liste complète des effets indésirables rapportés avec Tesavel, voir la notice.
Tesavel ne doit pas être utilisé chez les personnes pouvant présenter une hypersensibilité (allergie) à la sitagliptine ou à l'un des autres composants.
Pourquoi Tesavel a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) a estimé que les bénéfices de Tesavel sont supérieurs à ses risques et a recommandé son autorisation de mise sur le marché.
Autres informations sur Tesavel :
Le 10 janvier 2008, la Commission européenne a accordé à Merck Sharp & Dohme Ltd. une « autorisation de mise sur le marché » pour Tesavel. Cette autorisation était fondée sur l'autorisation accordée à Januvia en 2007 ("consentement éclairé").L'autorisation de mise sur le marché est valable cinq ans et peut être renouvelée au-delà de cette période.
Pour la version complète de l'EPAR de Tesavel, cliquez ici.
Dernière mise à jour de ce résumé : 10-2009
Les informations sur Tesavel - sitagliptina publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.