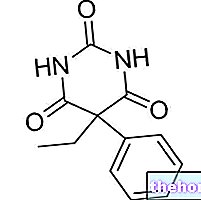
MIKAN ® est un médicament à base de sulfate d'amikacine
GROUPE THÉRAPEUTIQUE : Antibactériens - Aminoglycosides

Indications MIKAN ® Amikacine
MIKAN ® est indiqué dans le traitement des infections bactériennes sévères causées par des micro-organismes sensibles à l'amikacine, même résistants à la gentamicine.
Mécanisme d'action MIKAN ® Amikacine
L'amikacine, principe actif de MIKAN ®, appartient au groupe nombreux des aminosides, antibiotiques largement utilisés en milieu clinique pour le traitement des infections causées par des micro-organismes généralement résistants au traitement par bêta-lactamine.
Caractérisée par une excellente perméabilité membranaire, l'amikacine prise par voie parentérale atteint des concentrations plasmatiques maximales en seulement 30 à 60 minutes, se répartissant de manière homogène entre les différents fluides biologiques.
Pénétrée dans le cytoplasme bactérien, l'amikacine se lie et inhibe la sous-unité ribosomique 30S, nécessaire pour déterminer le bon allongement de la chaîne peptidique, donc pour sauvegarder l'ensemble du processus de synthèse protéique.
La formation et l'accumulation de peptides aberrants, en plus de compromettre les propriétés biochimiques et métaboliques des bactéries, endommagent gravement la membrane plasmique entraînant une perte maximale de potassium qui s'accompagne inévitablement d'une lyse cellulaire.
Malgré l'excellente efficacité d'action de l'amikacine, également dirigée contre les staphylocoques et les pseudomonas, différents microorganismes ont développé au cours du temps des mécanismes de résistance caractérisés par :
- Réduction de la perméabilité bactérienne à l'antibiotique, médiée par la réduction de l'expression des transporteurs membranaires ;
- Variation structurelle des domaines de liaison de l'antibiotique ;
- Expression de protéines membranaires ayant une activité lytique contre les aminosides.
Études réalisées et efficacité clinique
1. L'AMIKACINE DANS LE CONTRLE DES INFECTIONS À PSEUDOMONAS CHEZ LES PATIENTS CANCERÉS
J Paediatr Santé de l'enfant. Fév 2001 ; 37 : 38-43.
Ceftriaxone plus amikacine une fois par jour versus ceftazidime plusamikacine trois fois par jour comme traitement empirique de la neutropénie fébrile chez les enfants atteints de cancer.
Ariffin H, Arasu A, Mahfuzah M, Ariffin WA, Chan LL, Lin HP.
Étude intéressante démontrant l'efficacité de la thérapie combinée amikacine/ceftriaxone dans la réduction de la fièvre induite par Pseudomonas aeruginosa chez des patients pédiatriques cancéreux présentant une neutropénie manifeste.
2 . AMIKACINE DANS LE SEPSIS NÉONATAL
Scand J Infect Dis. 2006 ; 38 : 36-42.
Utilisation de pipéracilline/tazobactam (en association avec l'amikacine) dans le sepsis néonatal : données d'efficacité et de tolérance.
Fldel-Rimon O, Friedman S, Leibovitz E, Shinwell ES.
Des travaux qui démontrent comment l'ajout d'amikacine à une antibiothérapie courante peut être efficace et sûr dans le traitement de la septicémie néonatale, éradiquant les agents pathogènes en seulement 48 à 72 heures de traitement médicamenteux.
3.L"AMIKACINE DANS LA PREVENTION DES INFECTIONS POST-OPERATOIRES
BJU Int. 2011 mars ; 107 : 760-4.
L'ajout d'amikacine à la prophylaxie antimicrobienne à base de fluoroquinolone réduit les taux d'infection par biopsie de la prostate.
Batura D, Rao GG, Bo Nielsen P, Charlett A.
Travail intéressant démontrant l'efficacité des aminosides dans la prévention des infections du site opératoire.Dans cette étude, en effet, l'utilisation de l'amikacine s'est avérée efficace pour réduire le taux d'infections chez les patients subissant une biopsie de la prostate.
Mode d'emploi et posologie
MIKAN®
Solutions injectables à usage parentéral de 500 à 1000 mg d'amikacine.
Le schéma posologique de MIKAN ® doit être établi par le médecin en fonction des caractéristiques physiopathologiques du patient et de la gravité de son tableau clinique.
Toute adaptation doit être évaluée en cas d'infections particulièrement graves ou si le patient a des maladies rénales telles qu'une diminution du taux de filtration glomérulaire, avec pour conséquence une accumulation plasmatique d'amikacine.
Avertissements MIKAN ® Amikacine
La thérapie avec MIKAN ® doit nécessairement être définie et supervisée par le personnel médical, afin d'évaluer la pertinence de la prescription et en même temps contrôler l'apparition possible d'effets secondaires potentiels par des contrôles périodiques.
Une attention particulière doit être accordée aux patients ayant des antécédents de maladies neurologiques, auditif-vestibulaires et néphrologiques étant donné la forte sensibilité aux effets secondaires typiques de l'amikacine.
Si le patient présente des signes et des symptômes d'effets indésirables potentiels, il doit immédiatement en informer son médecin avec qui envisager la possibilité d'interrompre ou de modifier le traitement en cours.
La présence de métabisulfite de sodium parmi les excipients pourrait augmenter le risque de réactions d'hypersensibilité au médicament.
LA GROSSESSE ET L'ALLAITEMENT
L'utilisation de MIKAN ® pendant la grossesse et dans la période ultérieure d'allaitement est généralement contre-indiquée en raison de l'absence d'essais cliniques capables de caractériser l'efficacité thérapeutique et le profil de sécurité pour le fœtus exposé à la prise d'amikacine.
Interactions
Le patient recevant MIKAN ® doit porter une attention particulière à la prise simultanée de :
- Anesthésiques et relaxants musculaires capables de compromettre la fonction respiratoire, provoquant également une paralysie neuromusculaire ;
- Principes actifs présentant un potentiel d'ototoxicité et de néphrotoxicité, en raison du risque accru de survenue de néphropathies et de pathologies audito-vestibulaires ;
- Diurétiques parfois responsables de surdités sévères.
Contre-indications MIKAN ® Amikacine
L'utilisation de MIKAN® est contre-indiquée en cas d'hypersensibilité au principe actif et à ses excipients, pendant la grossesse et l'allaitement.
Effets indésirables - Effets secondaires
L'utilisation systémique des aminosides expose le patient à des effets secondaires typiques, parfois cliniquement pertinents.
L'ototoxicité liée à l'accumulation d'amikacine dans la périlymphe et l'endolymphe pourrait en effet provoquer l'apparition de troubles auditifs-vestibulaires tels que les acouphènes, les vertiges et la surdité tandis que la néphrotoxicité du principe actif pourrait augmenter le risque de troubles urinaires tels que l'albuminurie, l'hématurie, la cylindrurie. et dans les cas les plus graves, une azotémie et une hypercréatininémie.
En plus des effets secondaires mentionnés ci-dessus associés à la prise de MIKAN ®, il en existe d'autres qui sont généralement moins graves et transitoires caractérisés par des nausées, des vomissements, une hypotension, une anémie et une éosinophilie.
Noter
MIKAN ® est un médicament d'ordonnance.
Les informations sur MIKAN ® Amikacina publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.