Qu'est-ce que Kadcyla et à quoi sert-il ?
Kadcyla est un médicament anticancéreux qui contient le principe actif trastuzumab emtansine. Il est utilisé pour traiter les adultes atteints d'un cancer du sein avancé ou métastatique (cancer qui s'est propagé à d'autres parties du corps) qui ont déjà reçu un traitement par trastuzumab et un taxane (un type de médicament anticancéreux). Kadcyla ne peut être utilisé que lorsqu'il a été démontré que le cancer « exprime » des quantités excessives de HER2, c'est-à-dire lorsque la cellule cancéreuse produit une grande quantité d'une protéine à sa surface, appelée HER2 (facteur de croissance épidermique humain), qui stimule la croissance de la cellule elle-même.
Comment Kadcyla est-il utilisé - trastuzumab emtansine ?
Kadcyla n'est délivré que sur ordonnance et le traitement doit être prescrit par un médecin et administré sous la supervision d'un professionnel de santé expérimenté dans le traitement des patients cancéreux. Il est disponible sous forme de poudre à reconstituer en une solution pour perfusion (goutte-à-goutte) dans une veine. La dose à administrer dépend du poids corporel du patient et la perfusion est répétée toutes les trois semaines. Si la première perfusion de 90 minutes a été bien tolérée, les doses suivantes peuvent être administrées en perfusions de 30 minutes. Le traitement peut être poursuivi moins longtemps. que la maladie ne s'aggrave pas ou que le patient ne tolère plus le traitement.
Pendant la perfusion et immédiatement après la perfusion, les patients doivent être surveillés afin de détecter toute réaction liée à la perfusion, notamment rougeur, frissons et fièvre. Chez les patients qui développent des réactions allergiques ou des effets secondaires, le médecin peut être amené à réduire la dose ou à arrêter le traitement par Kadcyla. Pour plus d'informations, consultez la notice.
Comment agit Kadcyla - trastuzumab emtansine ?
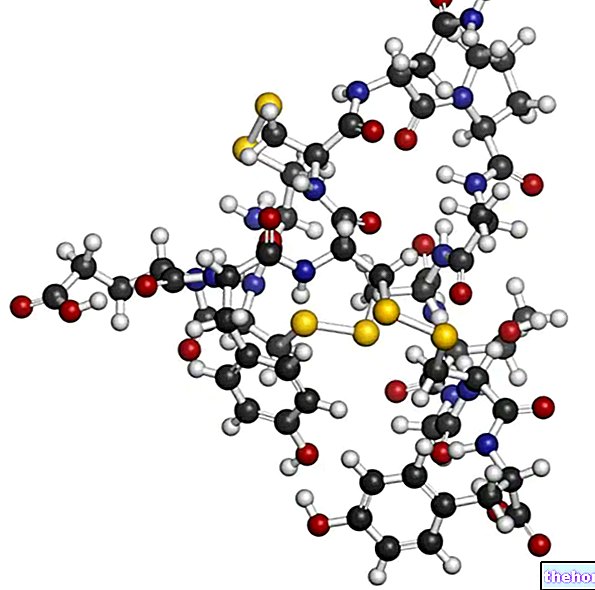
Le principe actif de Kadcyla, le trastuzumab emtansine, est constitué de deux composants actifs associés :
- trastuzumab, un anticorps monoclonal (un type de protéine) conçu pour reconnaître et se fixer à la protéine HER2, présente en grande quantité à la surface de certaines cellules cancéreuses. En se fixant à HER2, le trastuzumab active les cellules du système immunitaire, qui attaquent ensuite les cellules cancéreuses. Le trastuzumab empêche également HER2 de stimuler la croissance des cellules cancéreuses. Environ un quart des cancers du sein expriment des quantités excessives de HER2 ;
- DM1, une substance toxique qui tue les cellules lorsqu'elles tentent de se diviser et de croître. DM1 est activé lorsque Kadcyla pénètre dans la cellule cancéreuse. Il se lie à une protéine présente dans les cellules ("tubuline") qui est importante pour former le "squelette" interne dont les cellules ont besoin pour se reconstituer lorsqu'elles se divisent. En adhérant à la tubuline dans les cellules cancéreuses, le DM1 bloque la formation du squelette, empêchant les cellules cancéreuses de se diviser et de se développer.
Quel est le bénéfice démontré par Kadcyla - trastuzumab emtansine au cours des études ?
Il a été démontré que Kadcyla ralentit considérablement l'aggravation de la maladie et prolonge la survie des patientes atteintes d'un cancer du sein avancé et métastatique exprimant HER2 qui ont déjà reçu un traitement par trastuzumab et un taxane. Dans une étude principale portant sur 991 patients, les sujets traités par Kadcyla ont survécu en moyenne 9,6 mois sans progression de la maladie contre 6,4 mois pour les patients traités par deux autres médicaments anticancéreux, la capécitabine et le lapatinib. De plus, les patients traités par Kadcyla ont survécu 31 mois contre 25 mois chez les patients traités par capécitabine et lapatinib.
Quel est le risque associé à Kadcyla - trastuzumab emtansine ?
Les effets indésirables les plus fréquents avec Kadcyla (pouvant affecter plus de 25 % des patients) sont les saignements (y compris les saignements de nez), l'augmentation des taux sanguins de transaminases (enzymes hépatiques), la fatigue, les douleurs musculaires et osseuses et les maux de tête. Les effets indésirables graves les plus fréquents sont la pyrexie (fièvre), la thrombocytopénie (réduction du nombre de plaquettes), les vomissements, les douleurs abdominales, les nausées, la constipation, la diarrhée, la dyspnée (difficulté à respirer) et la pneumonie (inflammation des poumons). Pour la liste complète des effets indésirables rapportés avec Kadcyla, voir la notice.
Pourquoi Kadcyla - trastuzumab emtansine a-t-il été approuvé ?
Le comité des médicaments à usage humain (CHMP) de l'Agence a estimé que les bénéfices de Kadcyla sont supérieurs à ses risques et a recommandé son autorisation pour une utilisation dans l'UE.Le CHMP a pris en compte l'amélioration significative de la survie observée chez les patients traités par Kadcyla. par rapport au traitement standard En ce qui concerne la sécurité de Kadcyla, les effets secondaires globaux ont été considérés comme gérables et le profil de sécurité global a été considéré comme favorable par rapport aux médicaments actuellement disponibles.
Quelles mesures sont prises pour garantir l'utilisation sûre et efficace de Kadcyla - trastuzumab emtansine ?
Un plan de gestion des risques a été élaboré pour garantir que Kadcyla est utilisé de la manière la plus sûre possible. Sur la base de ce plan, des informations de sécurité ont été ajoutées au résumé des caractéristiques du produit et à la notice de Kadcyla, y compris les précautions appropriées à suivre par les professionnels de santé et les patients. En raison d'un risque potentiel de confusion entre Kadcyla et Herceptin, en raison de la similitude de nom des principes actifs (trastuzumab emtansine et trastuzumab), la société fournira du matériel pédagogique à tous les professionnels de santé susceptibles d'utiliser Kadcyla ou Herceptin, afin de préciser que les médicaments ils ne doivent pas être utilisés de manière interchangeable et de communiquer les mesures qui doivent être prises pour éviter les erreurs de médication.
Plus d'informations sur Kadcyla - trastuzumab emtansine
Le 15 novembre 2013, la Commission européenne a délivré une « autorisation de mise sur le marché » pour Kadcyla, valable dans toute l'Union européenne. Pour la version complète de l'EPAR Kadcyla, veuillez consulter le site Internet de l'Agence : ema.Europa.eu / Find medicine / Human Medicines / Rapports publics européens d'évaluation Pour plus d'informations sur le traitement par Kadcyla, veuillez lire la notice (jointe à l'EPAR) ou contacter votre médecin ou votre pharmacien. Dernière mise à jour de ce résumé : 11-2013.
Les informations sur Kadcyla - trastuzumab emtansine publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.