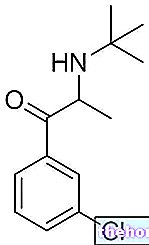
DICYNONE ® est un médicament à base d'éthamsylate.
GROUPE THÉRAPEUTIQUE : Antihémorragiques.

Indications DICYNONE® Etamsilate
DICYNONE ® est utilisé dans la prévention et le traitement des saignements en chirurgie générale et spécialisée.
L'étamsilate est également efficace dans le traitement thérapeutique des états de fragilité vasculaire.
Mécanisme d'action DICYNONE® Etamsilate
L'étamsilate, pris par voie orale via DICYNONE ® est efficacement absorbé dans l'intestin et facilement distribué dans tout le corps, lié à plus de 90 % aux protéines plasmatiques.
Les concentrations plasmatiques maximales sont obtenues environ 4 heures après l'administration orale. Le principe actif persiste donc dans la circulation sanguine pendant environ 8 heures, au bout desquelles il est éliminé, sous forme inchangée, principalement par voie urinaire.
L'action anti-hémorragique de l'étamsilate est garantie par l'effet direct sur l'adhésivité plaquettaire, nettement renforcé, par l'activation plaquettaire et par l'expression accrue des molécules d'adhésion induite par le principe actif. Ce mécanisme d'action permet une réduction significative de la durée et du volume des saignements, sans augmenter le risque de thrombose.
Études réalisées et efficacité clinique
ÉTHAMSYLATE ET MORTALITÉ PRÉMATURÉE
Cochrane Database Syst Rév. 20 janvier 2010 : CD004343.
Ethamsylate pour la prévention de la morbidité et de la mortalité chez les nourrissons prématurés ou de très faible poids de naissance.
Chasse R, Hé E.
Plusieurs chercheurs ont émis l'hypothèse que les effets anti-hémorragiques de l'étamsilate pourraient réduire l'incidence des hémorragies intraventriculaires, très fréquentes chez les prématurés, améliorant leur pronostic et leur évolution clinique. Malgré de nombreuses expériences, l'étamsilate ne semble pas affecter significativement la réduction de la mortalité chez ces petits patients.
2. EFFICACITÉ RÉDUITE DE L'ÉTHAMSILATE DANS LE TRAITEMENT DE LA MÉNORRAGIE
BMJ. 7 septembre 1996 ; 313 : 579-82.
Traitement de la ménorragie pendant la menstruation : essai contrôlé randomisé sur l'éthamsylate, l'acide méfénamique et l'acide tranexamique.
Bonnar J, Sheppard BL.
L'administration d'étamsilate chez des patients atteints de ménorragie n'a pas produit les résultats attendus, s'avérant inefficace pour réduire la quantité et la durée des saignements. Cette action modeste est à la base de l'utilisation réduite de ce médicament en obstétrique.
3. MÉCANISME MOLÉCULAIRE DE L'ÉTHAMSYLATE
Thromb Res. 15 sept. 2002, 107 : 329-35.
L'agent hémostatique éthamsylate améliore l'expression membranaire de la sélectine P dans les plaquettes humaines et les cellules endothéliales en culture.
Alvarez-Guerra M, Hernandez MR, Escolar G, Chiavaroli C, Garay RP, Hannaert P
Cette étude, menée sur des cultures cellulaires, a permis de clarifier le mécanisme moléculaire par lequel agit l'étamsilate.Plus précisément, ce principe actif semble être capable de pré-activer les plaquettes, augmentant l'expression des P-sélectines et d'autres molécules d'adhésion. responsable de l'adhésivité plaquettaire et de l'effet anti-hémorragique.
Mode d'emploi et posologie
DICYNONE® Comprimés d'étamsylate à 250 mg ou ampoules à 250 mg pour préparations injectables : dans la prophylaxie des événements hémorragiques peropératoires, la posologie recommandée d'étamsilate est de 1,5 g/jour (soit 6 comprimés) pendant les 3 jours précédant l'intervention et de 2 ampoules dans l'heure suivant l'intervention.
Dans la phase post-opératoire, le traitement doit être prolongé avec 1 - 1,5 gr / jour d'étamsilate pendant le même nombre de jours.
La prise de DICYNONE®, la posologie relative et le moment de la prise, doivent être évalués par votre médecin en tenant compte de l'état physiopathologique de l'adulte.
Le schéma de prévention chez l'enfant est naturellement soumis à des variations importantes, qui n'ont d'intérêt que pour les spécialistes.
DANS TOUS LES CAS, AVANT DE PRENDRE DICYNONE ® Etamsilate - VOUS AVEZ BESOIN DE LA PRESCRIPTION ET DU CONTRLE DE VOTRE MEDECIN.
Avertissements DICYNONE® Etamsilate
Étant donné l'importante sécrétion rénale d'étamsilate, DICYNONE ® doit être pris avec une prudence particulière chez tous les patients présentant une insuffisance rénale.
Le bon moment d'administration, à la fois dans la période préopératoire et postopératoire, est très important dans la prévention des événements hémorragiques associés à la chirurgie.
La présence dans les comprimés de sulfite de sodium et les flacons de sulfite monosodique peut provoquer des crises d'asthme sévères et des réactions allergiques chez les patients sensibles ou asthmatiques.
Dans ces cas et en présence de fièvre, d'érythème et d'éruption cutanée, DICYNONE ® doit être arrêté immédiatement.
LA GROSSESSE ET L'ALLAITEMENT
L'absence d'études relatives à la pharmacocinétique de l'étamsilate pendant la grossesse ne permet pas d'établir le profil de sécurité de DICYNONE®, il est donc déconseillé de prendre ce médicament pendant toute la période de la grossesse et de l'allaitement.
Interactions
Il n'y a actuellement aucune interaction connue avec d'autres ingrédients actifs qui pourraient compromettre la santé du patient.
Une augmentation de l'effet anti-hémorragique peut survenir suite à l'administration concomitante d'autres médicaments ayant une activité anti-hémorragique.
Contre-indications DICYNONE ® Etamsilate
DICYNONE ® est contre-indiqué en cas d'hypersensibilité à l'un de ses composants, et en cas d'insuffisance rénale sévère.
Effets indésirables - Effets secondaires
La présence de sulfites dans DICYNONE ® pourrait être associée à des réactions allergiques accompagnées de symptômes anaphylactiques.
Cependant, DICYNONE ® semble bien toléré, avec des réactions secondaires modestes telles que fièvre, maux de tête, éruptions cutanées, nausées, vomissements et diarrhée, qui ont tendance à régresser une fois le traitement arrêté.
Noter
DICYNONE ® ne peut être vendu que sur prescription médicale.
Les informations sur DICYNONE ® Etamsilato publiées sur cette page peuvent être obsolètes ou incomplètes. Pour une utilisation correcte de ces informations, consultez la page Avis de non-responsabilité et informations utiles.