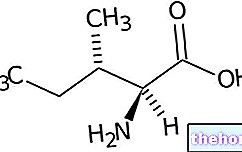
Ingrédients actifs : Barnidipine (chlorhydrate de barnidipine)
Vasexten 10 mg gélules à libération modifiée
Vasexten 20 mg gélules à libération modifiée
Pourquoi Vasexten est-il utilisé ? Pourquoi est-ce?
Le principe actif de Vasexten appartient à un groupe de médicaments appelés inhibiteurs calciques. Vasexten provoque la dilatation des vaisseaux sanguins, ce qui entraîne une baisse de la pression artérielle. Les capsules Vasexten sont à "libération prolongée". Cela signifie que le principe actif est absorbé progressivement par l'organisme et que son effet se prolonge dans le temps, c'est pourquoi une seule administration par jour suffit.
Vasexten est utilisé pour traiter l'hypertension artérielle.
Contre-indications Quand Vasexten ne doit pas être utilisé
Ne prenez jamais Vasexten
- si vous êtes allergique à la barnidipine ou à l'un des autres composants contenus dans ce médicament (mentionnés dans la rubrique 6)
- si vous êtes allergique aux dihydropyridines (substances présentes dans les médicaments utilisés pour traiter l'hypertension)
- si vous souffrez d'une maladie du foie
- si vous souffrez d'une maladie rénale grave
- si vous souffrez de l'une des maladies cardiaques suivantes : insuffisance cardiaque non traitée, certaines formes de douleurs thoraciques (dues à une angine de poitrine instable) ou un arrêt cardiaque aigu
- si vous utilisez l'un des médicaments suivants : inhibiteurs de protéase (médicaments utilisés pour traiter le SIDA), kétoconazole ou itraconazole (médicaments utilisés pour traiter les infections fongiques), érythromycine ou clarithromycine (antibiotiques).
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Vasexten ?
Adressez-vous à votre médecin ou pharmacien avant de prendre Vasexten
- si vous avez une maladie rénale
- si vous souffrez d'une maladie cardiaque
Enfants et adolescents
Vasexten ne doit pas être administré aux enfants ou aux adolescents de moins de 18 ans.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Vasexten
Autres médicaments et Vasexten
Informez votre médecin ou pharmacien si vous prenez, avez récemment pris ou pourriez prendre tout autre médicament.
Ceci est particulièrement important si vous utilisez l'un des médicaments suivants, car ils ne doivent pas être pris avec Vasexten :
- inhibiteurs de la protéase (médicaments utilisés pour traiter le SIDA)
- kétoconazole ou itraconazole (médicaments utilisés pour traiter les infections fongiques)
- érythromycine ou clarithromycine (antibiotiques)
Informez également votre médecin si vous prenez :
- d'autres médicaments pour traiter l'hypertension, ce qui peut entraîner une baisse supplémentaire de la pression artérielle
- cimétidine (médicament utilisé pour traiter les maladies de l'estomac) car elle peut augmenter l'effet de Vasexten
- la phénytoïne ou la carbamazépine (médicaments utilisés pour traiter l'épilepsie) ou la rifampicine (un antibiotique), car vous pourriez avoir besoin d'une dose plus élevée de Vasexten. Si vous arrêtez de prendre l'un de ces médicaments, votre médecin peut réduire votre dose de Vasexten.
Vasexten avec de la nourriture, des boissons et de l'alcool
Soyez particulièrement prudent lorsque vous buvez de l'alcool ou du jus de pamplemousse, car ces boissons peuvent augmenter l'effet de Vasexten.
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Si vous êtes enceinte ou si vous allaitez, si vous pensez être enceinte ou prévoyez une grossesse, demandez conseil à votre médecin ou votre pharmacien avant de prendre ce médicament.
Vous ne devez pas utiliser Vasexten si vous êtes enceinte, sauf en cas de nécessité absolue. N'utilisez pas Vasexten si vous allaitez. Il peut être excrété dans le lait maternel.
Conduire et utiliser des machines
Il n'existe aucune donnée suggérant que Vasexten puisse altérer l'aptitude à conduire des véhicules et à utiliser des machines. Cependant, Vasexten peut provoquer des vertiges, vous devez donc être sûr de l'effet que ce médicament a sur vous avant de conduire ou d'utiliser des machines.
Les capsules Vasexten contiennent du saccharose.
Si votre médecin vous a dit que vous aviez « une intolérance à certains sucres, contactez votre médecin avant de prendre ce médicament.
Posologie et mode d'utilisation Comment utiliser Vasexten : Posologie
Dosage
Prenez toujours ce médicament en suivant exactement les indications de votre médecin. En cas de doute, consultez votre médecin ou votre pharmacien.
La dose initiale habituelle est de 1 gélule de Vasexten 10 mg une fois par jour. Votre médecin peut augmenter cette dose à 1 gélule de Vasexten 20 mg une fois par jour ou à deux gélules de 10 mg une fois par jour.
Si vous êtes âgé, vous pouvez utiliser la posologie normale. Votre médecin est plus susceptible de vous suivre de plus près au début du traitement.
Instructions pour une utilisation correcte
- Prendre la gélule une fois par jour, le matin. Il est préférable d'associer la prise de la gélule à une action quotidienne, comme se brosser les dents ou prendre son petit-déjeuner.
- Avalez les gélules entières, de préférence avec un verre d'eau.Vous pouvez prendre Vasexten avant, pendant ou après un repas, selon vos préférences.
- Même si vous ne présentez aucun signe ou symptôme d'hypertension, il est important que vous continuiez à prendre Vasexten tous les jours pour profiter pleinement des avantages de l'abaissement de la tension artérielle.
Surdosage Que faire si vous avez pris trop de Vasexten
Si vous avez pris plus de Vasexten que vous n'auriez dû
Si vous avez accidentellement pris une grande quantité de gélules en une seule fois, vous devez contacter immédiatement votre médecin ou demander à être emmené aux urgences d'un hôpital. Les symptômes pouvant survenir à la suite d'un surdosage sont une faiblesse, une diminution ou une augmentation de la fréquence cardiaque, de la somnolence, de la confusion, des nausées, des vomissements et des convulsions.
Si vous oubliez de prendre Vasexten
Si vous oubliez de prendre Vasexten à l'heure habituelle de la journée, prenez la gélule dès que possible le même jour. Si vous ne vous en souvenez que le lendemain, ne prenez pas de dose double pour compenser une gélule oubliée, mais continuez votre dose quotidienne régulièrement.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Effets secondaires Quels sont les effets secondaires de Vasexten
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Si vous présentez une réaction allergique grave entraînant des difficultés respiratoires ou des étourdissements, vous devez en informer immédiatement votre médecin ou votre infirmier/ère.
Vasexten peut causer :
Très fréquent : affecte plus de 1 patient sur 10
- mal de tête
- rougeur du visage
- accumulation de liquide (œdème) dans les bras et les jambes
Fréquent : affecte jusqu'à 1 patient sur 10 :
- vertiges
- palpitations
Fréquence indéterminée : la fréquence ne peut être estimée à partir des données disponibles
- rythme cardiaque rapide
- tests sanguins montrant des changements dans la fonction hépatique
- éruption
Ces effets secondaires diminuent ou disparaissent généralement au cours du traitement (dans un délai d'un mois pour l'accumulation de liquide et dans les deux semaines pour les rougeurs du visage, les maux de tête et les palpitations).
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants.
Conservez les gélules de Vasexten à une température inférieure à 25°C.
Ne pas utiliser Vasexten après la date de péremption indiquée sur la boîte après « EXP ». La date d'expiration fait référence au dernier jour de ce mois.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Composition et forme pharmaceutique
Ce que contient Vasexten
- Chaque gélule de Vasexten contient 10 mg ou 20 mg de chlorhydrate de barnidipine, équivalant respectivement à 9,3 mg et 18,6 mg de barnidipine par gélule.
- Les autres composants sont : Contenu de la gélule : carboxyméthyléthylcellulose, polysorbate 80, saccharose, éthylcellulose et talc. Enveloppe de la gélule : dioxyde de titane (E171), oxyde de fer jaune (E172) et gélatine. Encre d'impression : gomme laque, propylène glycol (E 1520), oxyde de fer noir (E 172), ammoniac.
Qu'est-ce que Vasexten et contenu de l'emballage extérieur
Capsules jaunes.
Les gélules de Vasexten 10 mg sont marquées du code 155 10
Les gélules de Vasexten 20 mg sont marquées du code 155 20
Les gélules Vasexten sont conditionnées dans des plaquettes thermoformées en aluminium/aluminium (avec revêtement en PVC et polyamide) contenues dans des boîtes de 10, 14, 20, 28, 30, 50, 56, 98 ou 100 gélules. Toutes les présentations peuvent ne pas être commercialisées.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
VASEXTEN 10 MG GÉLULES À LIBÉRATION MODIFIÉE.
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Vasexten contient du chlorhydrate de barnidipine.
Les gélules à libération modifiée Vasexten® 10 mg contiennent 10 mg de chlorhydrate de barnidipine, équivalent à 9,3 mg de barnidipine par gélule.
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Gélules à libération modifiée, dures.
Les gélules de Vasexten 10 mg à libération modifiée sont jaunes et marquées 155 10.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Hypertension essentielle légère à modérée.
04.2 Posologie et mode d'administration
Dosage
La dose initiale recommandée est de 10 mg une fois par jour le matin, mais elle peut être augmentée à 20 mg une fois par jour si nécessaire. La décision d'augmenter la dose ne doit être prise qu'après avoir atteint une stabilité complète des valeurs de pression artérielle avec la dose de départ, ce qui prend généralement au moins 3 à 6 semaines.
Enfants
Aucune donnée n'étant disponible chez les enfants (moins de 18 ans), la barnidipine ne doit pas être administrée aux enfants.
Patients âgés
La dose n'a pas besoin d'être ajustée chez les patients âgés. Il est conseillé de faire plus attention au début du traitement.
Patients atteints d'insuffisance rénale
Chez les patients présentant une insuffisance rénale légère à modérée, des précautions doivent être prises lors de l'augmentation de la dose de 10 à 20 mg une fois par jour. Voir les rubriques « Contre-indications » et « Mises en garde spéciales et précautions d'emploi ».
Patients souffrant d'insuffisance hépatique
Voir la rubrique "Contre-indications".
Mode d'administration
Prenez les gélules de préférence avec un verre d'eau. Vasexten peut être pris avant, pendant ou après un repas.
04.3 Contre-indications
Hypersensibilité à la substance active (ou à d'autres dihydropyridines) ou à l'un des excipients.
Insuffisance hépatique. Insuffisance rénale sévère (clairance de la créatinine instable, angine de poitrine et infarctus aigu du myocarde (dans les 4 premières semaines). Insuffisance cardiaque non traitée.
Les taux sanguins de barnidipine peuvent augmenter lorsqu'ils sont utilisés en association avec des inhibiteurs puissants du CYP3A4 (comme le montrent les études d'interaction in vitro). Par conséquent, les antiprotéasiques, le kétoconazole, l'itraconazole, l'érythromycine et la clarithromycine ne doivent pas être associés.
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Vasexten doit être utilisé avec prudence chez les patients présentant une insuffisance rénale légère à modérée (clairance de la créatinine comprise entre 10 et 80 ml/min) (voir rubrique 4.2 « Posologie et mode d'administration »).
L'association d'un inhibiteur calcique avec un médicament qui exerce un effet inotrope négatif peut provoquer une insuffisance cardiaque, une hypotension ou un (autre) infarctus du myocarde chez les patients à haut risque (par exemple les patients ayant des antécédents d'infarctus du myocarde).
Comme avec tous les dérivés de la dihydropyridine, Vasexten doit être utilisé avec prudence chez les patients présentant une dysfonction ventriculaire gauche, chez les patients présentant une obstruction du canal d'éjection ventriculaire gauche et chez les patients présentant une décompensation cardiaque droite isolée, par ex. coeur pulmonaire.
La barnidipine n'a pas été étudiée chez les patients de classe NYHA III ou IV.
La prudence est également recommandée lors de l'administration de barnidipine aux patients atteints d'une maladie des sinus (en l'absence de stimulateur cardiaque).
Éducation in vitro indiquent que la barnidipine est métabolisée par le cytochrome P450 3A4 (CYP3A4). Aucune étude d'interaction n'a été réalisée in vivo sur l'effet des médicaments qui inhibent ou induisent l'enzyme cytochrome P450 3A4 sur la pharmacocinétique de la barnidipine. Sur la base des résultats d'études d'interaction in vitro, des précautions doivent être prises lorsque la barnidipine est prescrite en association avec des inhibiteurs ou inducteurs faibles de l'enzyme CYP3A4 (voir rubrique « Interactions avec d'autres médicaments et autres formes d'interactions »).
Le produit contient du saccharose; par conséquent, les patients présentant des problèmes héréditaires rares d'intolérance au fructose, un syndrome de malabsorption du glucose/galactose ou une insuffisance en sucrase-somaltase ne doivent pas prendre ce médicament.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
L'administration concomitante de barnidipine et d'autres agents antihypertenseurs peut entraîner un effet antihypertenseur supplémentaire.
Vasexten peut être utilisé en association avec des bêta-bloquants ou des inhibiteurs de l'ECA.
Le profil d'interaction pharmacocinétique de la barnidipine n'a pas fait l'objet d'études approfondies. Éducation in vitro montrent que la barnidipine est métabolisée par le cytochrome P450 3A4 (CYP3A4).
Aucune étude approfondie d'interaction n'a été réalisée in vivo l'effet des médicaments qui inhibent ou induisent l'enzyme CYP3A4 sur la pharmacocinétique de la barnidipine.
Données obtenues à partir d'études in vitro montrent que la cyclosporine peut inhiber le métabolisme de la barnidipine. Jusqu'à ce que les informations issues des études soient disponibles in vivo, la barnidipine ne doit pas être prescrite en association avec des inhibiteurs puissants du CYP3A4, tels que les antiprotéases, le kétoconazole, l'itraconazole, l'érythromycine et la clarithromycine (voir rubrique 4.3 « Contre-indications »). La prudence est recommandée en cas d'utilisation concomitante d'inhibiteurs ou d'inducteurs faibles du CYP3A4. En cas d'utilisation concomitante d'inhibiteurs du CYP3A4, il n'est pas recommandé d'augmenter la dose de barnidipine à 20 mg.
L'administration concomitante de cimétidine dans une étude d'interaction spécifique a entraîné, en moyenne, un doublement des taux plasmatiques de barnidipine. La prudence est donc recommandée lors de l'utilisation concomitante de barnidipine et de cimétidine.
Une dose plus élevée de barnidipine peut être nécessaire lorsque la barnidipine est administrée en même temps que des médicaments inducteurs enzymatiques, tels que la phénytoïne, la carbamazépine et la rifampicine. Si le patient arrête d'utiliser un médicament inducteur enzymatique, il faut envisager de réduire la dose de barnidipine.
Sur la base des résultats d'études d'interaction in vitro avec (entre autres) la simvastatine, le métoprolol, le diazépam et la terfénadine, il est peu probable que la barnidipine affecte la pharmacocinétique d'autres médicaments métabolisés par les isoenzymes du cytochrome P450.
Une étude d'interaction in vivo ont démontré que la barnidipine n'affecte pas la pharmacocinétique de la digoxine.
Dans une étude d'interaction, l'alcool a entraîné une augmentation des taux plasmatiques de barnidipine (40 %), ce qui n'est pas considéré comme cliniquement pertinent.
Bien que la cinétique de la barnidipine n'ait pas été modifiée de manière significative par l'administration de jus de pamplemousse, un effet modeste a été observé.
04.6 Grossesse et allaitement
Grossesse
Il n'y a pas d'expérience clinique avec la barnidipine pendant la grossesse ou l'allaitement. Les études chez l'animal ne suggèrent pas d'effets nocifs directs sur la grossesse, l'embryon/le fœtus ou le développement postnatal. Seuls des effets indirects ont été observés (voir 5.3). La classe des dihydropyridines a montré le potentiel pour prolonger le travail et l'accouchement, ce qui n'a pas été observé avec la barnidipine, la barnidipine ne doit donc être utilisée pendant la grossesse que si le bénéfice justifie le risque potentiel pour le fœtus.
L'heure du repas
Les résultats des tests sur les animaux ont montré que la barnidipine (ou ses métabolites) est excrétée dans le lait maternel. Par conséquent, l'allaitement n'est pas recommandé pendant l'utilisation de la barnidipine.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Il n'y a pas de données disponibles indiquant une possible influence négative de Vasexten sur l'aptitude à conduire des véhicules et à utiliser des machines. Cependant, la prudence est recommandée, car des étourdissements ou des vertiges peuvent survenir pendant le traitement antihypertenseur.
04.8 Effets indésirables
Les symptômes ont tendance à s'atténuer ou à disparaître au cours du traitement (œdème périphérique en un mois et bouffées de chaleur, maux de tête et palpitations en moins de deux semaines).
Les éruptions cutanées et une augmentation (réversible) de la phosphatase alcaline et des transaminases sériques sont des effets indésirables connus des autres dihydropyridines. Bien que des élévations transitoires et réversibles des enzymes hépatiques aient été rapportées à de rares occasions avec la barnidipine, elles n'ont pas été considérées comme cliniquement pertinentes.
Bien qu'il n'ait jamais été observé, l'événement indésirable suivant peut être intéressant, comme cela se produit avec l'utilisation d'autres dihydropyridines : hyperplasie gingivale.
Certaines dihydropyridines peuvent rarement provoquer des douleurs précordiales et une angine de poitrine. Très rarement, les patients souffrant d'angine de poitrine préexistante peuvent présenter une augmentation de la fréquence, de la durée et de la gravité de telles crises. Il peut y avoir des cas isolés d'infarctus du myocarde.
04.9 Surdosage
Aucun cas de surdosage n'a été rapporté.
Symptômes d'intoxication
En général, les symptômes cliniques après un surdosage d'inhibiteurs calciques se développent dans les 30 à 60 minutes suivant l'administration d'une dose 5 à 10 fois supérieure à la dose thérapeutique.
Les effets secondaires suivants peuvent théoriquement être prédits : hypotension, effets électrophysiologiques (bradycardie sinusale, allongement de la conduction AV, bloc AV du 2e et du 3e degré), effets sur le système nerveux central (étourdissements, confusion et, rarement, convulsions), symptômes gastro-intestinaux (nausées et vomissements ) et des effets métaboliques (hyperglycémie).
Traitement de l'intoxication
Un traitement hospitalier est nécessaire en « cas » d'intoxication. Un traitement symptomatique et une surveillance ECG continue sont indiqués.
En cas de surdosage, un lavage gastrique doit être effectué dès que possible.
Une injection intraveineuse (à la dose de 0,2 ml/kg de poids corporel) de calcium (de préférence 10 ml d'une solution de chlorure de calcium à 10 %) doit être administrée en 5 minutes, jusqu'à une dose totale de 10 ml à 10 % Contractilité myocardique , le rythme sinusal et la conduction auriculo-ventriculaire seront donc améliorés.
Le traitement peut être répété toutes les 15 à 20 minutes (jusqu'à un total de 4 doses) en fonction de la réponse du patient. Les niveaux de calcium doivent être surveillés.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : Antihypertenseurs. Code ATC C08CA12.
Mécanisme d'action :
La barnidipine (isomère S pur, S) est un inhibiteur lipophile des canaux calciques 1,4-dihydropyridine qui présente une « haute affinité avec les canaux calciques des cellules musculaires lisses de la paroi vasculaire. La cinétique des récepteurs de la barnidipine est caractérisée par une apparition de d "action lente et un lien fort et durable. La réduction de la résistance périphérique causée par la barnidipine provoque une baisse de la pression artérielle. Lors de l'utilisation de Vasexten, l'effet antihypertenseur persiste pendant toute la période de 24 heures.
L'utilisation de Vasexten dans le traitement chronique n'entraîne pas d'augmentation de la fréquence cardiaque de base.
L'impact de la barnidipine sur la morbidité et la mortalité cardiovasculaires n'a pas été étudié.
Cependant, des études contrôlées récemment conclues avec d'autres dihydropyridines à action prolongée ont indiqué des effets bénéfiques sur la morbidité et la mortalité similaires à ceux d'autres agents antihypertenseurs dans l'hypertension des personnes âgées.
Effets métaboliques :
La barnidipine n'a aucun effet négatif sur le profil lipémique, la glycémie ou les électrolytes du sang.
05.2 "Propriétés pharmacocinétiques
Absorption:
Après l'administration répétée de Vasexten® 20 mg à des sujets sains, la consommation concomitante de nourriture n'a eu aucun effet statistiquement significatif sur l'ASC, la Cmax, le Tmax ou la t½.
Les concentrations plasmatiques maximales sont atteintes 5 à 6 heures après l'administration orale de Vasexten® 20 mg.
Vasexten a une biodisponibilité absolue de 1,1%.
Les concentrations plasmatiques de barnidipine peuvent présenter une variabilité interindividuelle considérable.
Distribution:
Éducation in vitro montrent que la barnidipine se lie de 26 à 32 % aux érythrocytes humains et, dans une large mesure (89 à 95 %), aux protéines plasmatiques. L'analyse in vitro des composants protéiques indique que la barnidipine se lie principalement à l'albumine sérique, suivie par la glycoprotéine acide alpha1 et la lipoprotéine de haute densité. La liaison aux gammaglobulines se produit dans une bien moindre mesure.
Dans les études in vitro aucune interaction médicamenteuse basée sur l'élimination de la liaison aux protéines plasmatiques n'a été observée.
Biotransformation :
La barnidipine est métabolisée dans une large mesure en métabolites inactifs. Il n'y a pas d'inversion chirale in vivo de l'isomère pur S, S. Les principales réactions sont la Ndébenzilisation de la chaîne latérale, l'hydrolyse de l'ester de N-benzylpyrrolidine, l'oxydation du cycle 1,4-dihydropyridine, l'hydrolyse de l'ester méthylique et la réduction de groupe nitro Le métabolisme de la barnidipine semble être principalement médié par la famille d'isoenzymes CYP3A.
Excrétion:
La demi-vie d'élimination terminale médiane du plasma de Vasexten était de 20 heures après administration répétée selon un modèle analytique à deux compartiments.
L'élimination se fait principalement par métabolisme.La barnidipine et/ou ses métabolites sont excrétés dans les fèces (60%), les urines (40%) et l'air expiré (moins de 1%).La barnidipine n'est pas excrétée dans les urines.
Groupes de patients particuliers :
Après une dose unique, les taux plasmatiques de barnidipine sont 3 à 4 fois plus élevés chez les patients présentant une insuffisance hépatique légère à modérée que chez les volontaires sains. La variabilité des taux plasmatiques est également augmentée.
Les taux plasmatiques de barnidipine sont en moyenne le double chez les patients atteints d'insuffisance rénale qui n'ont pas besoin de subir d'hémodialyse par rapport aux volontaires sains. Le taux plasmatique moyen chez les patients hémodialysés est plus de 3 fois plus élevé que chez les volontaires sains, accompagné d'une "variabilité accrue".
05.3 Données de sécurité précliniques
Les données non cliniques ne révèlent aucun risque particulier pour l'homme sur la base des études conventionnelles de pharmacologie de sécurité, de toxicité à doses répétées, de génotoxicité, de potentiel cancérigène, de toxicité pour la reproduction.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Les autres composants de Vasexten sont les suivants :
Contenu de la gélule :
carboxyméthyléthylcellulose, polysorbate 80, saccharose, éthylcellulose, talc.
Capsule:
dioxyde de titane (E171), oxyde de fer jaune (E172) et gélatine.
Encre à imprimer:
gomme laque, alcool dénaturé, propylène glycol (E1520), eau purifiée, n-butanol, alcool isopropylique, oxyde de fer noir (E172).
06.2 Incompatibilité
Non pertinent.
06.3 Durée de validité
2 ans.
06.4 Précautions particulières de conservation
Ne pas conserver au-dessus de 25°C.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Les gélules à libération modifiée de Vasexten sont conditionnées dans des boîtes contenant 10,14,20,28,30,50,56,98 ou 100 gélules sous plaquettes thermoformées en aluminium-aluminium (avec revêtement en PVC et polyamide).
Une plaquette contient 7, 10 ou 14 capsules.
Toutes les présentations peuvent ne pas être commercialisées.
06.6 Instructions d'utilisation et de manipulation
Ne retirez pas les granulés des gélules.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Italfarmaco S.p.A. - Viale Fulvio Testi, 330 - 20126 Milan
Sous licence d'Astellas Pharma S.p.A.
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
Vasexten® 10 mg gélules à libération modifiée - 28 gélules est enregistré sous le numéro 035144029/M
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
19 novembre 2001 / Renouvellement 17 avril 2009
10.0 DATE DE RÉVISION DU TEXTE
mai 2010