Ingrédients actifs : Terbinafine
LAMISILMONO 1% solution cutanée
Pourquoi le Lamisilmono est-il utilisé ? Pourquoi est-ce?
LAMISILMONO est un traitement à dose unique pour le tinea pedis (pied d'athlète).
LAMISILMONO agit en tuant les champignons qui causent le tinea pedis (pied d'athlète) Lorsqu'il est appliqué sur les pieds, il laisse un film lisse et à peine visible qui reste sur la peau en libérant l'ingrédient actif dans la peau.
Comment savoir si vous avez un tinea pedis (pied d'athlète)
Tinea pedis (pied d'athlète) n'apparaît que sur les pieds. Il apparaît souvent entre les orteils mais peut s'étendre à la plante et aux côtés des pieds.
Le type le plus courant de tinea pedis (pied d'athlète) provoque des gerçures ou une desquamation de la peau. Vous pouvez également avoir un léger gonflement, des cloques ou des plaies humides. Cela peut souvent être associé à des démangeaisons ou des sensations de brûlure.
Si vous n'êtes pas sûr que vos symptômes soient vraiment dus à la teigne des pieds (pied d'athlète), consultez votre médecin ou votre pharmacien avant d'utiliser LAMISILMONO.
Contre-indications Quand Lamisilmono ne doit pas être utilisé
Ne pas utiliser LAMISILMONO
Si vous êtes allergique à la terbinafine ou à l'un des autres composants contenus dans ce médicament
Informez votre médecin ou votre pharmacien si cela vous concerne et que vous n'utilisez pas LAMISILMONO.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Lamisilmono
Adressez-vous à votre médecin ou pharmacien avant de prendre LAMISILMONO
- N'utilisez pas ce médicament si vous souffrez d'infections fongiques à long terme de la plante des pieds et des talons avec un épaississement associé et/ou une desquamation prononcée de la peau. Si vous pensez avoir cette condition, vous devriez consulter votre médecin car vous pourriez avoir besoin d'un autre médicament.
- LAMISILMONO est à usage externe uniquement. Ne l'utilisez pas dans votre bouche et ne l'avalez pas.
- Évitez tout contact avec votre visage, vos yeux ou une peau endommagée car l'alcool peut être irritant.
- Si la solution pénètre accidentellement dans vos yeux, rincez abondamment à l'eau courante. Si des problèmes persistent, consultez votre médecin.
- LAMISILMONO contient de l'alcool. Tenir à l'écart des flammes nues.
Enfants et adolescents
Les enfants et les adolescents de moins de 18 ans ne doivent pas utiliser LAMISILMONO.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Lamisilmono
Informez votre médecin ou pharmacien si vous utilisez, avez récemment utilisé ou pourriez utiliser tout autre médicament.
N'appliquez aucun autre médicament ou traitement sur votre pied (y compris ceux que vous avez peut-être achetés sans ordonnance) en même temps que LAMISILMONO.
Avertissements Il est important de savoir que :
Grossesse, allaitement et fertilité
- Si vous êtes enceinte, pensez l'être ou envisagez une grossesse, demandez conseil à votre médecin ou votre pharmacien avant d'utiliser ce médicament. LAMISILMONO ne doit pas être utilisé pendant la grossesse, sauf en cas de stricte nécessité.
- Ne pas utiliser LAMISILMONO pendant l'allaitement.Ne pas laisser les nourrissons entrer en contact avec les zones traitées, y compris les seins.
Conduire et utiliser des machines
- L'utilisation de LAMISILMONO n'affecte pas l'aptitude à conduire des véhicules et à utiliser des machines.
Dose, méthode et moment d'administration Comment utiliser Lamisilmono : Posologie
Utilisez ce médicament exactement comme décrit dans cette notice ou comme votre médecin vous l'a dit. En cas de doute, consultez votre médecin ou votre pharmacien.
La dose recommandée est d'appliquer la solution une fois, comme indiqué ci-dessous
Mode d'emploi:
Adultes
- Il est préférable d'appliquer LAMISILMONO après la douche ou le bain.
- Il s'agit d'un traitement unique.
- Lavez les deux pieds et séchez-les soigneusement.
- Lavez et séchez vos mains.
- Retirez le capuchon du tube.
- Vous l'appliquez sur les deux pieds - vous utilisez environ la moitié du tube pour chaque pied, au besoin pour couvrir la peau. Complétez un pied avant de traiter l'autre.
- Appliquez avec vos doigts sur chaque pied comme indiqué ci-dessous. Étaler uniformément sur la peau entre les doigts, en dessous et tout autour. Appliquez ensuite sur la plante et les côtés de son pied.
- Ne pas frotter ni masser.
- Traitez l'autre pied de la même manière, même si la peau a l'air saine, afin de vous assurer de vous débarrasser complètement du champignon - celui-ci peut être présent sur l'autre pied même sans marques.
- Laissez le produit sécher pendant 1 à 2 minutes avant de mettre vos chaussures habituelles.
- Lavez-vous les mains à l'eau chaude savonneuse après application.
- Ne pas laver ou mouiller vos pieds pendant 24 heures après l'application de LAMISILMONO.Tapez légèrement vos pieds pour les sécher après les avoir lavés délicatement.
- Ne pas appliquer sur la peau une deuxième fois.
À quelle fréquence et pendant combien de temps utiliser LAMISILMONO
Appliquer une seule fois. Ne pas appliquer une deuxième fois.
LAMISILMONO commence à tuer les champignons immédiatement. Le film formé indique que le principe actif pénètre sa peau où il continue d'agir pendant plusieurs jours.
Votre peau devrait commencer à s'améliorer en quelques jours, mais cela peut prendre jusqu'à 4 semaines pour que votre peau guérisse complètement.
Si vous ne constatez aucun signe d'amélioration dans la semaine suivant l'application de LAMISILMONO, consultez votre médecin ou votre pharmacien qui vous recommandera.
Ne pas utiliser le produit une deuxième fois au cours du même épisode de pied d'athlète s'il n'a pas fonctionné après la première application.
Vous devez appliquer LAMISILMONO sur les deux pieds, même si les marques ne sont visibles que sur un pied. Cela garantit une élimination complète du champignon : il peut être présent dans d'autres parties du pied même si aucune lésion n'est visible.
Une fois appliqué sur les pieds, le médicament sèche rapidement pour devenir un film transparent.
- Le tube contient suffisamment de médicament pour traiter les deux pieds
LAMISILMONO libère l'ingrédient actif dans la peau où il reste pendant plusieurs jours pour éliminer le champignon qui cause le pied d'athlète.Pour de meilleurs résultats, les pieds ne doivent pas être lavés ou mouillés pendant 24 heures après l'application.
Pour promouvoir le traitement
Gardez la zone affectée propre en la lavant régulièrement après les premières 24 heures. Séchez-la soigneusement sans masser. Essayez de ne pas rayer la zone même si elle vous démange, car cela pourrait causer des dommages supplémentaires et ralentir le processus de guérison ou propager l'infection.
Comme ces infections peuvent être transmises à d'autres personnes, n'oubliez pas de ne garder que votre serviette et vos vêtements pour vous et de ne pas les partager avec d'autres. Lavez vos vêtements et vos serviettes fréquemment pour vous protéger d'une réinfection.
Surdosage Que faire si vous avez pris trop de Lamisilmono
Si vous avalez accidentellement un produit
Contactez votre médecin qui vous dira quoi faire. La teneur en alcool doit être prise en compte
Si le produit entre en contact avec le visage ou les yeux
Rincez-vous le visage ou les yeux à l'eau courante. Consultez votre médecin si vous ressentez encore une gêne.
[Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.]
Effets secondaires Quels sont les effets secondaires du Lamisilmono
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Certaines personnes peuvent être allergiques à LAMISILMONO, ce qui peut provoquer un gonflement et une douleur, des éruptions cutanées ou de l'urticaire. Cela a été rapporté très rarement (peut affecter jusqu'à 1 personne sur 100).
Si vous ressentez une réaction allergique ou l'un des symptômes ci-dessus lors de l'utilisation de ce produit, retirez le papier d'aluminium avec de l'alcool dénaturé (en vente en pharmacie), lavez-vous les pieds à l'eau tiède savonneuse, rincez-les et séchez-les et consultez votre médecin ou votre pharmacien .
Les effets secondaires suivants ont été rapportés :
rare (ils peuvent affecter jusqu'à 1 personne sur 100)
Réactions au site d'application pouvant inclure une peau sèche, une irritation de la peau ou une sensation de brûlure.
Ces réactions sont généralement légères et transitoires.
Si vous ressentez un quelconque effet indésirable, veuillez en informer votre médecin ou votre pharmacien. Cela inclut tous les effets secondaires possibles non mentionnés dans cette notice.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants.
Ne pas utiliser LAMISILMONO après la date de péremption mentionnée sur la boîte et le tube.La date de péremption fait référence au dernier jour du mois indiqué.
A conserver dans l'emballage d'origine à l'abri de la lumière. Ne pas conserver au dessus de 30°C.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Ce que contient LAMISILMONO
- L'ingrédient actif est la terbinafine (sous forme de chlorhydrate). Chaque gramme de solution cutanée contient 10 mg de terbinafine (sous forme de chlorhydrate).
- Les autres composants sont : copolymère acrylates/octylacrylamide, hydroxypropylcellulose, triglycérides saturés à chaîne moyenne et éthanol.
Description de l'apparence de LAMISILMONO et contenu de l'emballage extérieur
LAMISILMONO est une solution visqueuse limpide ou légèrement opaque. Il est disponible en tubes de 4g.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus à jour, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
LAMISILMONO 1% SOLUTION POUR LA PEAU
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
Chaque gramme de solution cutanée contient 10 mg de terbinafine (sous forme de chlorhydrate).
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE
Solution pour la peau.
Solution visqueuse claire ou légèrement opaque.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Traitement du tinea pedis (pied d'athlète) (voir rubrique 4.4).
04.2 Posologie et mode d'administration
Usage cutané.
Dosage
Adultes : une seule application.
Lamisilmono ne doit être appliqué qu'une seule fois sur les deux pieds, même si les lésions sont visibles sur un seul pied. Cela garantit l'élimination des champignons (dermatophytes) qui peuvent être présents dans les zones du pied où aucune lésion n'est visible.
Mode d'administration
Les patients doivent se laver et se sécher les pieds et les mains avant d'appliquer le médicament. Ils doivent traiter un pied, puis l'autre.
En commençant par les orteils, les patients doivent appliquer uniformément une fine couche entre et autour des orteils, ainsi que couvrir la plante et les côtés du pied jusqu'à 1,5 cm. Le médicament doit être appliqué de la même manière sur l'autre pied, même si la peau semble saine. Le médicament doit être laissé sécher pendant 1 à 2 minutes jusqu'à ce qu'il forme un film. Les patients doivent ensuite se laver les mains. Lamisilmono ne doit pas être utilisé. massé dans la peau.
Pour de meilleurs résultats, les zones traitées ne doivent pas être lavées pendant 24 heures après l'application. Il est donc recommandé d'appliquer Lamisilmono après une douche ou un bain et d'attendre le lendemain à la même heure avant de se laver à nouveau délicatement les pieds. ; après le lavage, les pieds doit être séché en tamponnant doucement.
Les patients doivent utiliser la quantité requise pour couvrir les deux pieds comme illustré ci-dessus. Tout médicament restant doit être jeté.
Le soulagement des symptômes cliniques est généralement observé en quelques jours.
Si aucun signe d'amélioration n'est observé après une semaine, le diagnostic doit être réexaminé et les patients doivent alors consulter un médecin. Il n'y a pas de données sur le traitement répété avec Lamisilmono. Par conséquent, un deuxième traitement ne peut pas être recommandé pendant le même épisode de pied d'athlète.
Posologie dans des populations particulières :
Population pédiatrique
Lamisilmono n'a pas été étudié dans la population pédiatrique. Par conséquent, son utilisation est déconseillée chez les patients de moins de 18 ans.
Patients âgés
Aucun ajustement posologique n'est nécessaire.
04.3 Contre-indications
Hypersensibilité à la substance active ou à l'un des excipients mentionnés à la rubrique 6.1.
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Lamisilmono doit être utilisé avec prudence chez les patients présentant des blessures où l'alcool peut être irritant. Il ne doit pas être utilisé sur le visage.
Lamisilmono est à usage externe uniquement. Cela peut être irritant pour les yeux.
En cas de contact accidentel avec les yeux, rincer abondamment les yeux à l'eau courante.
Lamisilmono doit être conservé hors de la portée des enfants.
Lamisilmono n'est pas recommandé pour le traitement de la teigne plantaire hyperkératosique chronique (type "mocassin").
En cas de réaction allergique, le film doit être retiré avec un solvant organique tel que l'alcool dénaturé et les pieds lavés à l'eau tiède savonneuse.
Informations concernant les excipients
Lamisilmon contient de l'éthanol; tenir à l'écart des flammes nues.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Il n'y a pas d'interactions médicamenteuses connues avec Lamisilmono.
04.6 Grossesse et allaitement
Grossesse
Il n'y a pas d'expérience clinique avec la terbinafine chez la femme enceinte.
Les études de toxicité fœtale chez l'animal n'ont pas indiqué d'effets indésirables (voir rubrique 5.3).Lamisilmono ne doit pas être utilisé pendant la grossesse sauf en cas de nécessité absolue.
L'heure du repas
La terbinafine est excrétée dans le lait maternel. Lamisilmono ne doit pas être utilisé pendant l'allaitement.
La fertilité
Les études chez l'animal n'ont montré aucun effet de la terbinafine sur la fertilité (voir rubrique 5.3).
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Lamisilmono n'affecte pas l'aptitude à conduire des véhicules et à utiliser des machines.
04.8 Effets indésirables
Les effets indésirables comprennent des réactions légères et transitoires au site d'application. Dans de très rares cas, des réactions allergiques peuvent survenir.
Affections de la peau et du tissu sous-cutané:
Très rare (prurit, dermatite bulleuse et urticaire.
Troubles généraux et anomalies au site d'administration:
Peu fréquent (> 1 / 1 000,
04.9 Surdosage
En cas d'ingestion accidentelle, la teneur en alcool (81,05 % w/w) du Lamisilmono doit être prise en compte.
Un surdosage est très peu probable car le médicament est à application unique, à usage cutané et le tube ne contient que la quantité nécessaire pour une application.L'ingestion accidentelle du produit contenu dans un tube de 4 g contenant 40 mg de terbinafine est très réduite par rapport à la prise d'un comprimé de Lamisil à 250 mg (unité posologique orale pour adulte). Cependant, si plusieurs tubes de Lamisilmono sont ingérés par inadvertance, des effets indésirables similaires à ceux observés après un surdosage avec les comprimés de Lamisil sont à prévoir. Ceux-ci comprennent des maux de tête, des nausées, des douleurs épigastriques et des étourdissements.
Traitement du surdosage
En cas d'ingestion accidentelle, le traitement recommandé du surdosage est d'éliminer la substance active, principalement par l'administration de charbon activé, et si nécessaire, d'apporter un traitement symptomatique d'appoint.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : autres antifongiques à usage topique.
Code ATC : D01AE15.
La terbinafine est une allylamine qui interfère spécifiquement avec la biosynthèse des stérols fongiques dans une première étape.Cela conduit à une carence en ergostérol et à une accumulation intracellulaire de squalène, entraînant la mort de la cellule fongique.La terbinafine agit en inhibant la cellule fongique.squalène époxydase dans la membrane cellulaire fongique. L'enzyme squalène époxydase n'est pas liée au système du cytochrome P450. La terbinafine n'affecte pas le métabolisme des hormones ou d'autres médicaments.
La terbinafine possède un large spectre d'activité antifongique dans les infections fongiques de la peau causées par des dermatophytes tels que Trichophyton (par exemple.T. rubrum, T. mentagrophytes, T. verrucosum, T. violaceum), Microsporum canis et Epidermophyton floccosum. A faible concentration, la terbinafine est un fongicide contre les dermatophytes.
Des études de patients ont montré que l'application d'une dose unique de Lamisilmono 1% solution cutanée sur les deux pieds s'est avérée efficace chez les patients atteints de tinea pedis (pied d'athlète) qui présentaient des lésions interdigitales, et qui s'étendaient aux zones adjacentes. côtés et plante des pieds.
Cependant, une comparaison directe de l'efficacité avec d'autres formes topiques de Lamisil n'a pas été réalisée, par conséquent aucun jugement ne peut être porté à l'heure actuelle concernant l'efficacité relative de Lamisilmono 1% solution cutanée par rapport à d'autres formes topiques.
05.2 Propriétés pharmacocinétiques
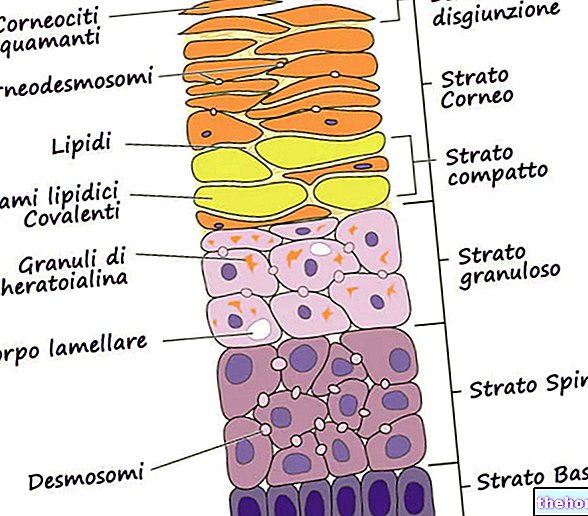
Une fois appliquée sur la peau, Lamisilmono 1% solution cutanée forme un film sur la peau. La terbinafine est susceptible d'être liée ou retenue dans les composants lipidiques du stratum corneum, ce qui peut jouer un rôle dans la longue demi-vie d'élimination de ce médicament du stratum corneum. La terbinafine reste dans la couche cornée jusqu'à 13 jours, à des niveaux supérieurs à la concentration minimale inhibitrice in vitro de la terbinafine sur les dermatophytes.
Le lavage du film diminue la teneur en terbinafine dans le stratum corneum et donc le lavage doit être évité pendant les 24 premières heures suivant l'application pour permettre une pénétration maximale de la terbinafine dans le stratum corneum.
L'occlusion, susceptible d'être envisagée dans le cadre d'une utilisation thérapeutique, entraînera une augmentation de 2,7 fois de l'exposition du stratum corneum à la terbinafine après une seule application de Lamisilmon 1% solution cutanée.Bien que non étudiée, la pénétration dans le stratum corneum de la terbinafine est probablement plus importante dans Cela est dû à l'effet occlusif trouvé dans les espaces interdigitaux, qui est probablement accru par "le port de chaussures, plutôt qu'à tout effet causé par une altération" de l'intégrité de la couche cornée.
La biodisponibilité systémique est très faible tant chez les volontaires sains que chez les patients.
Une "application de Lamisilmono 1% solution cutanée sur le dos, sur une" zone 3 fois plus grande que la zone des deux pieds, a entraîné une "exposition estimée à la terbinafine inférieure à 0,5% de l'exposition suite à l'administration par voie orale de un comprimé de 250 mg.
05.3 Données de sécurité précliniques
Dans les études à long terme (jusqu'à 1 an) chez le rat et le chien, aucun effet toxique marqué n'a été observé chez les deux espèces jusqu'à des doses orales d'environ 100 mg/kg par jour. À des doses orales élevées, le foie et peut-être aussi les reins ont été identifiés comme des organes cibles potentiels.
Dans une étude de cancérogénicité orale de 2 ans chez la souris, il n'y a eu aucun signe néoplasique ou autre anomalie attribuable au traitement avec des doses allant jusqu'à 130 (mâles) et 156 (femelles) mg/kg par jour. Dans une étude de cancérogénicité orale de 2 ans chez le rat à la dose la plus élevée, 69 mg/kg par jour, une augmentation de l'incidence des tumeurs du foie a été observée chez les mâles. spécifiques à l'espèce puisqu'ils n'ont pas été observés dans l'étude de cancérogénicité chez la souris ou dans d'autres études chez la souris, le chien ou le singe.
Au cours des études avec des doses orales élevées de terbinafine chez le singe, des irrégularités de réfraction de la rétine ont été observées à des doses plus élevées (le niveau non toxique était de 50 mg/kg). Ces irrégularités étaient associées à la présence d'un métabolite de la terbinafine dans le tissu oculaire et ont disparu après l'arrêt du médicament. Ils n'étaient pas associés à des changements histologiques.
Un ensemble standard de tests pour évaluer in vitro Et in vivo la génotoxicité n'a montré aucun potentiel mutagène ou clastogène pour le médicament.
Aucun effet indésirable sur la fertilité ou d'autres paramètres de reproduction n'a été observé dans les études chez le rat et le lapin.
L'application répétée sur la peau de Lamisilmono 1% solution cutanée sur des rats et des cochons miniatures produit des niveaux plasmatiques de terbinafine qui sont au moins 50 à 100 fois inférieurs à ceux établis comme niveaux sans effet indésirable dans les études de toxicité animale de la terbinafine, donc utilisation du médicament ne devrait pas produire d'effets indésirables systémiques.
Lamisilmon 1% solution cutanée a été bien toléré dans de nombreuses études de tolérance et n'a pas induit de sensibilisation.
La sécurité du copolymère acrylates/octylacrylamide, un excipient récemment utilisé dans les médicaments dermatologiques topiques, a été établie sur la base d'études conventionnelles de toxicité à dose unique et répétée, de génotoxicité et de tolérabilité locale.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Copolymère acrylates/octylacrylamide; hydroxypropylcellulose; triglycérides saturés à chaîne moyenne; éthanol.
06.2 Incompatibilité
Non pertinent.
06.3 Durée de validité
3 années.
06.4 Précautions particulières de conservation
A conserver dans l'emballage d'origine à l'abri de la lumière. Ne pas conserver au dessus de 30°C.
06.5 Nature du conditionnement primaire et contenu de l'emballage
Tube en aluminium laminé de 4 g (polyéthylène-aluminium-polyéthylène) avec bouchon à vis en polyéthylène.
06.6 Instructions d'utilisation et de manipulation
Pas d'instructions particulières.
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Novartis Farma S.p.A., Largo U. Boccioni 1 - Origgio (VA).
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
Tube de 4 g - A.I.C. n.m. 038282012 / M.
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
Première autorisation : janvier 2008.
Renouvellement : 4 novembre 2010.
10.0 DATE DE RÉVISION DU TEXTE
20 janvier 2013.