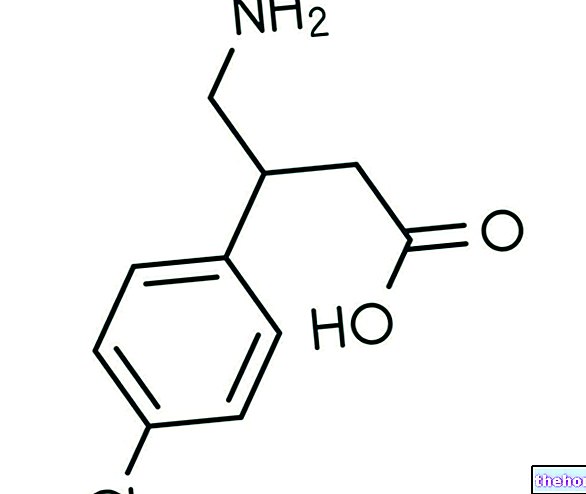
Ingrédients actifs : Mésalazine (5-ASA)
PENTASA 500 mg comprimés à libération modifiée
PENTASA 1 g granulés à libération prolongée
Les notices d'emballage Pentasa sont disponibles pour les tailles d'emballage : - PENTASA 500 mg comprimés à libération modifiée, PENTASA 1 g granulés à libération prolongée
- PENTASA 1 g suppositoires
- PENTASA 4 g/100 ml suspension rectale
Indications Pourquoi Pentasa est-il utilisé ? Pourquoi est-ce?
Pentasa contient le principe actif mésalazine, qui appartient à un groupe de médicaments appelés salicylés, anti-inflammatoires intestinaux utilisés pour traiter la rectocolite hémorragique et la maladie de Crohn.
La colite ulcéreuse est une maladie du côlon (gros intestin) et du rectum dans laquelle la paroi interne de l'intestin devient rouge et enflée (enflammée), avec des symptômes tels que des selles fréquentes et des selles sanglantes, accompagnées de crampes d'estomac.
Lorsqu'il est administré pour un épisode aigu de colite ulcéreuse, Pentasa agit sur l'ensemble du côlon et du rectum pour traiter l'inflammation et réduire les symptômes. Les comprimés peuvent également être pris pour aider à prévenir le retour de la colite ulcéreuse.
Dans la phase active sévère, l'association avec des médicaments à base de cortisone peut être conseillée.
Contre-indications Quand Pentasa ne doit pas être utilisé
Ne pas utiliser Pentasa
- si vous êtes allergique à la substance active ou à l'un des autres composants contenus dans ce médicament (mentionnés dans la rubrique 6)
- si vous êtes allergique à une classe de médicaments appelés salicylates (par exemple l'aspirine)
- chez les enfants de moins de 6 ans
- pendant les dernières semaines de grossesse et pendant l'allaitement (voir "Grossesse, allaitement et fertilité")
- si vous avez de graves problèmes rénaux ou hépatiques.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Pentasa
Adressez-vous à votre médecin ou pharmacien avant d'utiliser Pentasa :
- si vous êtes allergique à la sulfasalazine (allergie aux salicylates)
- si votre foie fonctionne mal ou mal
- si vous souffrez de problèmes rénaux. La mésalazine induit une néphrotoxicité (toxicité rénale), elle doit donc être interrompue chez les patients qui développent un dysfonctionnement rénal pendant le traitement.
- si vous êtes traité avec des médicaments qui peuvent affecter votre fonction rénale, tels que les anti-inflammatoires non stéroïdiens (AINS) et l'azathioprine, car ils peuvent augmenter le risque de réactions rénales
- si vous êtes traité par l'azathioprine ou la 6-mercaptopurine et la thioguanine car ce sont des médicaments qui peuvent augmenter le risque de dyscrasie sanguine (troubles sanguins). Le traitement doit être réduit ou interrompu en présence de ces effets indésirables
- si vous avez une digestion gastrique particulièrement lente ou si vous êtes porteur d'une sténose du pylore (rétrécissement de la partie de l'estomac qui mène à l'intestin grêle)
- si vous avez des problèmes respiratoires tels que l'asthme.
Surtout pendant la phase initiale du traitement, votre médecin peut demander des analyses d'urine et de sang pour évaluer la fonction de vos reins, de votre foie.
Enfants et adolescents
Pentasa n'est pas recommandé pour une utilisation chez les enfants de moins de 6 ans.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Pentasa
Informez votre médecin si vous utilisez, avez récemment utilisé ou pourriez utiliser tout autre médicament, y compris ceux pouvant être pris sans ordonnance.
Ceci est particulièrement important si vous prenez des médicaments tels que :
- Azathioprine, 6-mercaptopurine et thioguanine (connus sous le nom de médicaments « immunosuppresseurs » qui réduisent l'activité de votre système immunitaire).
- Anticoagulants coumariniques (médicaments qui augmentent le temps nécessaire à la coagulation du sang). Par exemple la warfarine.
Pentasa avec nourriture et boisson
Ce médicament peut être pris avec des aliments et des boissons (voir rubrique 3).
Avertissements Il est important de savoir que :
Grossesse, allaitement et fertilité
Si vous êtes enceinte ou si vous allaitez, si vous pensez être enceinte ou prévoyez une grossesse, demandez conseil à votre médecin ou votre pharmacien avant d'utiliser ce médicament.
Pentasa doit être utilisé avec prudence pendant la grossesse ou l'allaitement, et uniquement en cas d'absolue nécessité (voir également la rubrique 2 « N'utilisez jamais Pentasa »).
Des troubles sanguins ont été observés chez les nourrissons de mères traitées par Pentasa.
Des réactions allergiques telles que la diarrhée chez les nouveau-nés ne peuvent être exclues.
Si le nourrisson souffre de diarrhée, l'allaitement doit être interrompu.
Conduire et utiliser des machines
Le traitement par Pentasa n'affecte normalement pas l'aptitude à conduire des véhicules et/ou à utiliser des machines.
Dose, méthode et moment d'administration Comment utiliser Pentasa : Posologie
Utilisez toujours ce médicament en suivant exactement les indications de votre médecin ou pharmacien. En cas de doute, consultez votre médecin ou votre pharmacien.
La dose recommandée est :
Adultes:
Dans la rectocolite hémorragique
Traitement aigu : jusqu'à 4 g de mésalamine une fois par jour ou répartis en 2 à 4 administrations
Traitement d'entretien : il est recommandé de commencer par 1 500 mg de mésalamine par jour, répartis en 3 prises.
Dans la maladie de Crohn
Traitement aigu : jusqu'à 3-4 g de mésalamine par jour, répartis en 2-3 administrations.
Traitement d'entretien : il est recommandé de débuter par 3 g de mésalamine par jour, répartis en 3 prises.
Utilisation chez les enfants
Enfants à partir de 6 ans
Il est généralement recommandé d'administrer la moitié de la dose adulte aux enfants pesant jusqu'à 40 kg et la dose adulte normale à ceux pesant plus de 40 kg.
- Traitement aigu : à partir de 30-50 mg/kg/jour en administrations séparées. Dose maximale : 75 mg/kg/jour en prises séparées. La dose totale ne doit pas dépasser 4 g/jour (dose maximale adulte).
- Traitement d'entretien : à partir de 15-30 mg/kg/jour en prises séparées. La dose totale ne doit pas dépasser 2 g/jour (dose adulte recommandée).
Les comprimés ou sachets peuvent être pris pendant ou peu après un repas. Pentasa peut être pris avec de l'eau ou du yaourt. Les comprimés peuvent être avalés entiers ou, à défaut, ils peuvent également être brisés ou dissous dans un verre ou une cuillère avec un peu d'eau.
Les comprimés ou granulés de Pentasa ne doivent pas être mâchés. Après ouverture du sachet, les granulés doivent être pris dans les 24 heures (voir "Comment conserver Pentasa").
Si vous oubliez de prendre Pentasa
Si vous oubliez de prendre une dose de Pentasa, prenez votre prochaine dose régulière prévue à l'heure habituelle.
Ne prenez pas de dose double pour compenser une dose oubliée.
Si vous arrêtez de prendre Pentasa
Votre médecin vous dira combien de temps durera le traitement. Adressez-vous à votre médecin avant d'arrêter le traitement par ce médicament.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Surdosage Que faire si vous avez pris trop de Pentasa
Si vous avez pris plus de comprimés ou de sachets que vous n'auriez dû, contactez immédiatement votre médecin ou rendez-vous à l'hôpital le plus proche.
Effets secondaires Quels sont les effets secondaires de Pentasa
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Effets indésirables fréquents (pouvant affecter jusqu'à 1 personne sur 10) :
- Mal de tête
- La diarrhée
- Douleur abdominale
- La nausée
- il vomit
- Flatulence (gaz intestinaux)
- Éruptions cutanées, telles que urticaire, érythème.
Effets indésirables rares (pouvant affecter jusqu'à 1 personne sur 1 000) :
- Vertiges
- Mio- et péricardite (inflammation de certaines parties du cœur)
- Pancréatite aiguë (inflammation aiguë du pancréas) et augmentation des amylases (enzymes du pancréas).
Effets indésirables très rares (pouvant affecter jusqu'à 1 personne sur 10 000) :
- Troubles sanguins, tels que l'éosinophilie (augmentation d'un type de globules blancs appelés éosinophiles), l'anémie (diminution des globules rouges), l'anémie aplasique (une forme d'anémie caractérisée par une production insuffisante de cellules sanguines de tous types dans la moelle osseuse) , leucopénie (diminution des globules blancs), thrombocytopénie (diminution des plaquettes), agranulocytose (diminution d'un type de globules blancs appelés granulocytes), pancytopénie (diminution de toutes les cellules sanguines)
- Réactions d'hypersensibilité (allergie), y compris éruption allergique, réaction anaphylactique, réaction médicamenteuse avec éosinophilie et symptômes systémiques (DRESS), érythème polymorphe et syndrome de Steavens-Johnson (SJS)
- Fièvre médicamenteuse
- Neuropathie périphérique (état pathologique affectant les nerfs des mains et des pieds)
- Alvéolite allergique, réactions allergiques et troubles pulmonaires (réactions fibrotiques pulmonaires, y compris difficulté à respirer (dyspnée), toux, bronchospasme, pneumonie à éosinophiles, maladie pulmonaire interstitielle, infiltration pulmonaire, pneumonie)
- Pancolite (inflammation du côlon)
- Troubles hépatiques (augmentation des enzymes hépatiques, des indices cholestatiques et de la bilirubine, hépatotoxicité, y compris hépatite, hépatite cholestatique, cirrhose, insuffisance hépatique)
- Alopécie (chute ou amincissement des cheveux) réversible
- Douleurs musculaires (myalgies), douleurs articulaires (arthralgies), cas de réactions immunitaires de type lupus érythémateux
- Troubles rénaux (néphrite interstitielle aiguë et chronique, syndrome néphrotique, insuffisance rénale, décoloration des urines)
- Oligospermie (faible concentration de spermatozoïdes dans le sperme) réversible.
Certains des effets secondaires peuvent également être attribués à la maladie elle-même.
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice. Vous pouvez également signaler les effets indésirables directement via le système national de notification à l'adresse www.agenziafarmaco.gov.it/it/responsabili. En signalant les effets indésirables, vous pouvez contribuer à fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants.
Après ouverture des sachets, les granulés peuvent être conservés pendant 24 heures, après quoi ils doivent être jetés.
N'utilisez pas ce médicament après la date de péremption indiquée sur l'emballage après « EXP ». La date de péremption fait référence au dernier jour de ce mois.Cette date fait référence au produit dans un emballage intact, correctement stocké.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Délai "> Autres informations
Ce que contient Pentasa
Comprimé à libération modifiée :
- L'ingrédient actif est la mésalamine
- Les autres composants sont : polyvinylpyrrolidone, éthylcellulose, stéarate de magnésium, talc, cellulose microcristalline.
Sachet de granulés à libération prolongée :
- L'ingrédient actif est la mésalamine
- Les autres composants sont : éthylcellulose, cellulose microcristalline.
Description de ce à quoi ressemble Pentasa et contenu de l'emballage
Pentasa 500 mg comprimés à libération modifiée : Les comprimés sont présentés avec de petites taches blanc-gris à brun clair et une ligne sécable. 500 mg est imprimé d'un côté et PENTASA de l'autre Boîte de 50 comprimés.
Sachets Pentasa 1 g de granulés à libération prolongée : granulés blanc-gris à blanc-brun clair. Paquet de 50 sachets.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT -
PENTASA
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE -
Un comprimé à libération modifiée contient : 500 mg de mésalamine (acide 5-amino-salicylique).
Un sachet de granulés à libération prolongée contient : mésalazine (acide 5-amino-salicylique) 1 g.
Pour la liste complète des excipients, voir rubrique 6.1.
03.0 FORME PHARMACEUTIQUE -
Comprimés sécables à libération modifiée avec de petites taches blanc-gris à brun clair, une ligne sécable et une mention : 500 mg d'un côté, PENTASA de l'autre.
Granulés à libération prolongée, de couleur blanc-gris à blanc-brun clair.
04.0 INFORMATIONS CLINIQUES -
04.1 Indications thérapeutiques -
Rectocolite hémorragique. La maladie de Crohn.
Pentasa est indiqué à la fois dans le traitement de la phase active de la maladie et dans la prévention des rechutes. Dans la phase active sévère, l'association avec un traitement par cortisone peut être conseillée.
04.2 Posologie et mode d'administration -
Dosage
La posologie doit être adaptée à chaque patient en fonction de la gravité de la maladie. Dans la rectocolite hémorragique
Traitement aigu : jusqu'à 4 g de mésalamine une fois par jour ou répartis en 2 à 4 administrations.
Traitement d'entretien : il est recommandé de débuter par 1500 mg de mésalamine par jour, répartis en 3 prises.
Dans la maladie de Crohn
Traitement aigu : jusqu'à 3-4 g de mésalamine par jour, répartis en 2-3 administrations.
Traitement d'entretien : il est recommandé de débuter par 3 g de mésalamine par jour, répartis en 3 prises.
Population pédiatrique
Il n'existe qu'une documentation limitée d'un effet chez les enfants (6-18 ans). Enfants à partir de six ans
Traitement aigu : doit être établi individuellement, en commençant par 30-50 mg/kg/jour en administrations séparées. Dose maximale : 75 mg/kg/jour en prises séparées. La dose totale ne doit pas dépasser 4 g/jour (dose maximale adulte).
Traitement d'entretien : doit être établi individuellement, en commençant par 15-30 mg/kg/jour en prises séparées. La dose totale ne doit pas dépasser 2 g/jour (dose adulte recommandée).
Il est généralement recommandé d'administrer la moitié de la dose adulte aux enfants pesant jusqu'à 40 kg et la dose adulte normale à ceux pesant plus de 40 kg.
Mode d'administration
Les comprimés ou granulés de Pentasa ne doivent pas être mâchés.
Les comprimés ou sachets peuvent être pris pendant ou peu après un repas. Vous pouvez également casser les comprimés ou les laisser se déliter dans un verre ou sur une cuillère avec de l'eau. Pentasa peut être pris avec de l'eau ou du yaourt.
04.3 Contre-indications -
Hypersensibilité à la substance active, aux salicylates ou à l'un des excipients mentionnés à la rubrique 6.1.
Enfants de moins de 6 ans.
Dernières semaines de grossesse et de lactation (voir rubrique 4.6). Insuffisance rénale ou hépatique sévère.
04.4 Mises en garde spéciales et précautions d'emploi appropriées -
Des rechutes de symptômes objectifs et subjectifs peuvent survenir à la fois après l'arrêt du traitement et au cours d'un traitement d'entretien inadéquat. La survenue possible de réactions d'hypersensibilité entraîne l'arrêt immédiat du traitement. Chez les patients qui ont une digestion lente ou qui sont porteurs d'une sténose pylorique, il peut parfois être possible d'avoir une libération de 5-ASA déjà dans l'estomac, avec pour conséquence une irritation gastrique et une perte d'efficacité du médicament.
Pentasa doit être utilisé avec prudence chez les patients allergiques à la sulfasalazine (risque d'allergie aux salicylates). En cas de réactions d'intolérance aiguës telles que crampes abdominales, douleurs abdominales aiguës, fièvre, maux de tête sévères et éruption cutanée, le traitement doit être arrêté immédiatement.
La prudence est recommandée chez les patients présentant une insuffisance hépatique. Les paramètres de la fonction hépatique, tels que l'ALAT ou l'ASAT, doivent être évalués au début et pendant le traitement, à la discrétion du médecin.
L'utilisation de Pentasa n'est pas recommandée chez les patients présentant des troubles rénaux. La fonction rénale doit être surveillée régulièrement (par exemple, la créatinine sérique), en particulier pendant la phase initiale du traitement. La mésalazine induit une néphrotoxicité, elle doit donc être interrompue chez les patients qui développent un dysfonctionnement rénal pendant le traitement. En cas d'utilisation concomitante d'autres agents néphrotoxiques, tels que les AINS et l'azathioprine, la fréquence de surveillance de la fonction rénale doit être augmentée.
Les patients atteints de maladies pulmonaires, en particulier d'asthme, doivent être étroitement surveillés pendant le traitement.
Des réactions d'hypersensibilité cardiaque à la mésalamine (mine et péricardite) et très rarement des dyscrasies sanguines sévères ont été rarement rapportées. Des tests sanguins pour des numérations globulaires différentielles sont recommandés au début et pendant le traitement, à la discrétion du médecin. Un traitement concomitant par la mésalamine peut augmenter le risque de dyscrasie sanguine chez les patients recevant de l'azathioprine ou de la 6-mercaptopurine ou de la thioguanine. (voir rubrique 4.5) Le traitement doit être arrêté si ces effets indésirables sont suspectés ou surviennent.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions -
Le traitement concomitant par PENTASA et l'azathioprine ou la 6-mercaptopurine ou la thioguanine a montré une fréquence plus élevée d'effets myélosuppresseurs dans plusieurs études, et bien que le mécanisme d'interaction n'ait pas été complètement établi, une corrélation semble exister.Une surveillance régulière est recommandée. et la posologie de la thiopurine doit être ajustée en conséquence.
La mésalamine peut diminuer l'effet anticoagulant de la warfarine.
04.6 Grossesse et allaitement -
Pentasa doit être utilisé avec prudence pendant la grossesse ou l'allaitement et uniquement si les bénéfices potentiels l'emportent, de l'avis du médecin, sur les risques possibles.La mésalazine traverse la barrière placentaire et sa concentration dans le plasma du cordon ombilical est inférieure à celle du plasma maternel. Le métabolite acétyl-mésalazine a été trouvé à des concentrations similaires dans le plasma du cordon ombilical et du cordon maternel. Dans plusieurs études d'observation, aucun effet tératogène n'a été signalé et il n'y a aucune preuve de risque significatif chez l'homme. Les études animales avec la mésalazine orale n'ont pas indiqué d'effets indésirables directs ou indirects sur la grossesse, le développement embryo-fœtal, la parturition ou le développement postnatal. Des troubles sanguins (pancytopénie, leucopénie, thrombocytopénie, anémie) ont été rapportés chez les nouveau-nés de mères traitées par Pentasa.
La mésalamine est excrétée dans le lait maternel. La concentration de mésalamine dans le lait maternel est plus faible que dans le sang maternel tandis que le métabolite, l'acétyl-mésalazine, est à des concentrations similaires ou supérieures. Il existe une « expérience limitée » de l'utilisation orale de la mésalamine chez les femmes qui allaitent. Aucune étude contrôlée n'a été menée avec Pentasa pendant l'allaitement. Des réactions d'hypersensibilité telles que la diarrhée chez les nouveau-nés ne peuvent pas être exclues. Si le nourrisson souffre de diarrhée, l'allaitement doit être interrompu.
Les études animales sur la mésalamine n'ont montré aucun effet sur la fertilité des mâles et des femelles.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines -
Il est peu probable que le traitement par Pentasa affecte l'aptitude à conduire des véhicules et/ou à utiliser des machines.
04.8 Effets indésirables -
Les effets secondaires sont répertoriés ci-dessous par organe, organe/système et fréquence MedDRA. Les fréquences sont définies comme : très fréquente (1/10) fréquente (≥ 1/100 a
Fréquence des événements indésirables sur la base des essais cliniques et des rapports de surveillance post-commercialisation
(*) le mécanisme par lequel la mésalazine induit la myo- et péricardite, la pancréatite, la néphrite et l'hépatite est inconnu, mais il est probablement de nature allergique.
Il est important de noter que bon nombre des effets secondaires peuvent également être attribués à la MII elle-même.
Déclaration des effets indésirables suspectés.
La déclaration des effets indésirables suspectés survenant après autorisation du médicament est importante car elle permet un suivi continu du rapport bénéfice/risque du médicament. Les professionnels de santé sont invités à déclarer tout effet indésirable suspecté via le système national de déclaration. agenziafarmaco.gov.it/it/responsabili.
04.9 Surdosage -
Expérience aiguë chez l'animal :
Des doses orales de mésalamine allant jusqu'à 5 g/kg chez le porc ou des doses intraveineuses uniques de mésalazine allant jusqu'à 920 mg/kg chez le rat n'ont pas été létales.
Expérience chez l'homme :
L'expérience clinique limitée de surdosage n'indique pas de toxicité rénale ou hépatique. Un antidote spécifique n'est pas connu et le traitement est symptomatique et de soutien. Traitement du surdosage chez l'homme :
Traitement symptomatique en milieu hospitalier. Surveillance attentive de la fonction rénale.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES -
05.1 "Propriétés pharmacodynamiques -
Classe pharmacothérapeutique : anti-inflammatoires intestinaux, acide aminé salicylique et analogues.
ATC : A07EC02.
La mésalamine s'est avérée être l'ingrédient actif de la sulfasalazine, qui est utilisée pour le traitement de la colite ulcéreuse, de la maladie de Crohn et de la rectite.
Des études cliniques montrent que l'intérêt thérapeutique de la mésalazine, après administration orale ou rectale, semble être dû à un effet local sur la muqueuse intestinale enflammée plutôt qu'à un effet systémique.
Les patients atteints de MICI présentent : une migration accrue des leucocytes, une production anormale de cytokines, une production accrue de métabolites de l'acide arachidonique, en particulier le leucotriène B4, et une formation accrue de radicaux libres dans la muqueuse intestinale enflammée. effet qui inhibe la chimiotaxie des leucocytes, diminue la production de cytokines et de leucotriènes et neutralise les radicaux libres.On ignore actuellement lequel de ces mécanismes joue un rôle prépondérant dans l'efficacité clinique de la mésalazine.
05.2 "Propriétés pharmacocinétiques -
Disponibilité locale : l'activité thérapeutique de la mésalazine dépend très probablement du contact local du médicament avec la zone de la muqueuse intestinale malade.
Les sachets et comprimés à libération modifiée Pentasa sont constitués de microgranules de mésalamine enrobés d'éthylcellulose. Après l'administration et la désintégration des comprimés, la mésalazine est libérée en continu des microgranules individuels dans tout le tractus gastro-intestinal à différentes conditions de pH entéral.
Les microgranules arrivent dans le duodénum dans l'"heure suivant l'administration, indépendamment" de la prise alimentaire concomitante. Le taux de transit moyen dans l'intestin grêle est d'environ 3 à 4 heures chez des volontaires sains.
Absorption
D'après les données urinaires de volontaires sains, 30 à 50 % de la dose ingérée est absorbée après administration orale, principalement dans l'intestin grêle.
La mésalazine est détectable dans le plasma dès 15 minutes après l'administration. La concentration plasmatique maximale est obtenue après 1 à 4 heures. Après une diminution progressive, la mésalamine ne sera plus détectable 12 heures après l'administration. La courbe de concentration plasmatique de l'acétyl mésalazine suit le même schéma, mais les concentrations sont généralement plus élevées et l'élimination est plus lente.
Le rapport métabolique de l'acétyl-mésalazine et de la mésalamine dans le plasma après administration orale varie de 3,5 à 1,3 après des doses quotidiennes de 500 mg x3 et 2 g x3, respectivement, impliquant une acétylation dose-dépendante qui peut être sujette à saturation.
Les concentrations plasmatiques de mésalamine à l'état d'équilibre sont d'environ 2 mol/L, 8 mol/L et 12 μmol/L après des doses quotidiennes de 1,5 g, 4 g et 6 g, respectivement. Pour l'acétyl mésalazine les concentrations correspondantes sont de 6 mcmol/l, 13 mcmol/l et 16 mcmol/l.
Distribution
La mésalazine se lie aux protéines plasmatiques pour environ 50 %, l'acétyl-mésalazine pour environ 80 %. Le 5-ASA et l'Ac-5-ASA ne traversent pas la barrière hémato-encéphalique.
Biotransformation
La mésalazine est métabolisée à la fois par voie présystémique par la muqueuse intestinale et par voie systémique dans le foie en N-acétyl-mésalazine (acétylmésalazine). Une partie de l'acétylation se produit également par la flore bactérienne du côlon. L'acétylation semble être indépendante du phénotype d'acétylation du patient.
On pense que l'acétyl mésalazine est cliniquement et toxicologiquement inactive, mais cela n'a pas encore été confirmé.
Élimination
La demi-vie plasmatique de la mésalamine est d'environ 40 minutes et celle de l'acétyl-mésalazine d'environ 70 minutes. En raison de la libération continue de PENTASA dans le tractus gastro-intestinal, la demi-vie d'élimination de la mésalazine ne peut pas être déterminée après administration orale.Cependant, l'état d'équilibre est atteint après une période de traitement de 5 jours après l'administration orale.
Les deux substances sont excrétées dans l'urine et les fèces.
L'excrétion urinaire est principalement constituée d'acétyl mésalazine.
Chez les patients présentant une insuffisance hépatique ou rénale, la diminution de la vitesse d'élimination et l'augmentation de la concentration systémique de mésalazine peuvent constituer un risque accru d'effets néphrotoxiques indésirables.
05.3 Données de sécurité préclinique -
Un effet toxique sûr sur le rein a été démontré chez toutes les espèces. En général, les doses toxiques dépassent de 5 à 10 fois celles utilisées chez l'homme.
Aucune toxicité significative associée au tractus gastro-intestinal, au foie ou au système hématopoïétique n'a été rapportée chez les animaux.
Test in vitro et études in vivo ils n'ont pas montré d'effets mutagènes et clastogènes. Les études sur le potentiel cancérogène menées chez la souris et le rat n'ont pas montré d'augmentation liée à la substance de l'incidence des tumeurs.
Les études animales avec la mésalazine orale n'ont pas indiqué d'effets nocifs directs ou indirects sur la fertilité, la grossesse, le développement embryo-fœtal, la parturition ou le développement postnatal.
06.0 INFORMATIONS PHARMACEUTIQUES -
06.1 Excipients -
Comprimés : polyvinylpyrrolidone, éthylcellulose, stéarate de magnésium, talc, cellulose microcristalline.
Sachets : éthylcellulose, cellulose microcristalline.
06.2 Incompatibilité "-
Non pertinent.
06.3 Durée de validité "-
Comprimés à libération modifiée : 3 ans.
Granulés à libération prolongée : 2 ans. Une fois les sachets ouverts, le
le granulé est stable pendant 24 heures.
06.4 Précautions particulières de conservation -
Ce médicament ne nécessite aucune condition particulière de conservation.
06.5 Nature du conditionnement primaire et contenu de l'emballage -
Comprimés à libération modifiée :
Boîte de 50 comprimés (5 plaquettes thermoformées aluminium/polyamide-aluminium-chlorure de vinyle).
Granulés à libération prolongée :
Pack de 50 sachets de granulés. Les granulés sont contenus dans des sachets en aluminium.
06.6 Instructions d'utilisation et de manipulation -
Pas d'instructions particulières.
Les médicaments non utilisés et les déchets dérivés de ce médicament doivent être éliminés conformément aux réglementations locales.
07.0 TITULAIRE DE « L'AUTORISATION DE MISE SUR LE MARCHE » -
Ferring S.p.A. - Via Senigallia 18/2 - 20161 MILAN
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ -
500 mg comprimés à libération modifiée, 50 comprimés : 027130071
1 g de granulés à libération prolongée, 50 sachets : 027130083
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION -
Sachets Pentasa 1g :
Immatriculation 22 octobre 1998 - Renouvellement d'autorisation juin 2010












.jpg)