Ingrédients actifs : Acide tranexamique
UGUROL 500 mg/5 ml solution injectable pour voie intraveineuse, pour usage oral et local
Les notices d'emballage Ugurol sont disponibles pour les packs :- UGUROL 500 mg/5 ml solution injectable pour voie intraveineuse, pour usage oral et local
- UGUROL 250 mg comprimés
Pourquoi Ugurol est-il utilisé ? Pourquoi est-ce?
UGUROL contient de l'acide tranexamique, qui appartient à un groupe de médicaments appelés antihémorragiques, antifibrinolytiques, acides aminés.
UGUROL est utilisé chez les adultes et les enfants à partir d'un an pour prévenir et traiter les saignements dus à un processus qui inhibe la coagulation du sang appelé fibrinolyse.
Les indications spécifiques sont :
- cycle menstruel lourd;
- saignement gastro-intestinal;
- troubles hémorragiques des voies urinaires, suite à une chirurgie de la prostate ou une chirurgie des voies urinaires ;
- chirurgie cardiaque, abdominale ou gynécologique;
- saignement après un traitement avec d'autres médicaments pour dissoudre les caillots sanguins.
Contre-indications Quand Ugurol ne doit pas être utilisé
Ne prenez jamais UGUROL :
- si vous êtes allergique à l'acide tranexamique ou à l'un des autres composants contenus dans ce médicament
- si vous souffrez d'une maladie qui entraîne des caillots sanguins ;
- si vous souffrez d'une maladie appelée « coagulopathie de consommation » où le sang commence à coaguler dans différentes parties du corps ;
- si vous avez des problèmes rénaux ;
- si vous avez eu des crises dans le passé.
En raison du risque d'œdème cérébral et de convulsions, l'injection intrathécale et intraventriculaire et l'application intracérébrale ne sont pas recommandées.
Si vous pensez que l'un de ces cas vous concerne, ou si vous avez d'autres questions, parlez-en à votre médecin avant de prendre UGUROL.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Ugurol
Informez votre médecin si l'un des cas suivants s'applique à vous pour l'aider à décider si UGUROL vous convient :
- si vous avez remarqué du sang dans vos urines, cela pourrait être dû à une obstruction des voies urinaires ;
- si vous présentez un risque de caillots sanguins ;
- si vous avez des caillots ou des saignements excessifs dans tout votre corps (coagulation intravasculaire disséminée), UGUROL peut ne pas vous convenir, sauf si vous présentez des saignements aigus sévères et que des analyses sanguines ont montré que le processus qui inhibe la coagulation du sang, appelé fibrinolyse, est activé ;
- si vous avez eu des convulsions, UGUROL ne doit pas être administré. Votre médecin doit utiliser la dose la plus faible possible pour éviter les crises dues au traitement par UGUROL ;
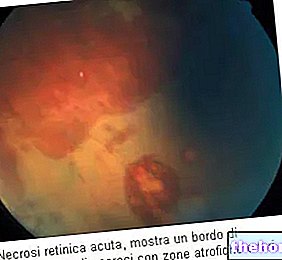
- si vous suivez un traitement prolongé par UGUROL, une attention particulière doit être portée aux troubles possibles de la vision des couleurs et, si nécessaire, le traitement doit être arrêté. En cas d'utilisation prolongée d'UGUROL solution injectable, des examens ophtalmologiques réguliers sont recommandés (examens de la vue comprenant l'acuité visuelle, la vision des couleurs, le fond d'œil, le champ de vision, etc.). En cas d'évolutions ophtalmologiques pathologiques, notamment de maladies de la rétine, votre médecin devra décider, après consultation d'un spécialiste, de la nécessité d'une utilisation prolongée d'UGUROL solution injectable dans votre cas.
Interactions Quels médicaments ou aliments peuvent modifier l'effet d'Ugurol
Informez votre médecin ou pharmacien si vous prenez ou avez pris récemment tout autre médicament, y compris des médicaments sans ordonnance, des vitamines, des minéraux, des médicaments à base de plantes ou des compléments alimentaires.
En particulier, vous devez informer votre médecin si vous prenez :
- d'autres médicaments qui aident à la coagulation du sang appelés antifibrinolytiques ;
- médicaments qui empêchent la coagulation du sang appelés thrombolytiques;
- contraceptifs oraux.
Avertissements Il est important de savoir que :
La grossesse et l'allaitement
Demandez conseil à votre médecin si vous êtes enceinte ou si vous allaitez avant de prendre UGUROL.
L'acide tranexamique étant excrété dans le lait maternel, l'utilisation d'UGUROL est déconseillée pendant l'allaitement.
Conduire et utiliser des machines
Aucune étude sur l'aptitude à conduire des véhicules et à utiliser des machines n'a été réalisée.
Dose, mode et heure d'administration Comment utiliser Ugurol : Posologie
Prenez ce médicament en suivant exactement les indications de votre médecin. En cas de doute, consultez votre médecin ou votre infirmier/ère.
Utilisation chez l'adulte par voie orale
- Prophylaxie
Si la solution est prise par voie orale, en diluant le contenu de l'ampoule avec un peu d'eau sucrée, la posologie journalière est de 1½-2 ampoules d'Ugurol de 500 mg, en commençant l'administration au moins 1 jour avant la chirurgie et en poursuivant le traitement pendant une période d'au moins 3-4 jours après l'opération.
- Thérapie
Si la solution est prise par voie orale, en diluant le contenu de l'ampoule dans un peu d'eau sucrée, la posologie journalière est de 1 à 2 ampoules d'Ugurol à 500 mg 3 fois par jour ou ½ à 1 ampoule d'Ugurol à 500 mg 6 fois par jour .
L'administration orale est particulièrement indiquée :
- dans les manifestations hémorragiques survenant en médecine interne, en oto-rhino-laryngologie et en dentisterie ; pour la préparation d'interventions chirurgicales dans lesquelles il est supposé que des hémorragies dues à l'activation plasminique peuvent survenir ;
- dans l'hyperménorrhée;
- dans les gemmes gynécologiques, dans les cystites et rectites hémorragiques consécutives à une radiothérapie pour carcinome génital ;
- pour le maintien des thérapies initiées par voie intraveineuse afin de prévenir la récurrence de l'hémorragie.
Utilisation intraveineuse chez l'adulte
La solution d'UGUROL est administrée en injection lente dans une veine. Votre médecin décidera de la dose qui vous convient et de la durée pendant laquelle vous devez la prendre.
Utilisation chez l'adulte en application locale
Pour l'application locale d'UGUROL 500 mg/5ml solution, utiliser le contenu de 1 flacon et le verser directement sur le site du saignement ou l'appliquer à l'aide d'une compresse de gaze préalablement imbibée.
L'application locale directe ou au moyen de compresses de gaze préalablement trempées dans la solution est particulièrement indiquée pour les saignements au niveau oro-nasopharyngé pour lesquels une hémostase rapide est souhaitée.
Utilisation chez les enfants
Si la solution d'UGUROL est administrée à un enfant à partir d'un an, la dose sera calculée en fonction du poids de l'enfant. Votre médecin décidera de la dose appropriée pour votre enfant et de la durée pendant laquelle vous devrez la prendre.
Utilisation chez les personnes âgées
Il n'est pas nécessaire de réduire la dose sauf en cas d'insuffisance rénale avérée.
Utilisation chez les patients souffrant de problèmes rénaux
Si vous avez des problèmes rénaux, votre dose d'acide tranexamique sera réduite sur la base d'un test sanguin (taux de créatinine sérique).
Utilisation chez les patients ayant des problèmes de foie
Il n'est pas nécessaire de réduire la dose.
Mode d'administration
La solution d'UGUROL doit être administrée lentement dans une veine
La solution d'UGUROL ne doit pas être injectée dans un muscle.
Surdosage Que faire si vous avez pris trop d'Ugurol
Si vous avez reçu plus d'UGUROL que la dose recommandée
Si vous recevez plus d'UGUROL que la dose recommandée, vous pouvez avoir une chute transitoire de la tension artérielle. Informez immédiatement votre médecin ou votre pharmacien.
Si vous oubliez de prendre UGUROL
Ne prenez pas de dose double pour compenser une dose oubliée.
Si vous avez d'autres questions sur l'utilisation de ce médicament, demandez plus d'informations à votre médecin ou votre pharmacien.
Effets secondaires Quels sont les effets secondaires d'Ugurol
Comme tous les médicaments, ce médicament peut provoquer des effets indésirables, bien que tout le monde n'y soit pas sujet.
Les effets indésirables suivants ont été observés avec UGUROL :
Fréquent (peut affecter jusqu'à 1 patient sur 10)
- effets sur l'estomac et les intestins : nausées, vomissements, diarrhée.
Peu fréquent (peut affecter 1 à 10 utilisateurs sur 1000)
- effets sur la peau : éruption cutanée
Fréquence indéterminée (la fréquence ne peut être estimée à partir des données disponibles)
- malaise avec hypotension (pression artérielle basse), en particulier si l'injection a été administrée trop rapidement ;
- caillots sanguins;
- effets sur le système nerveux : convulsions ;
- effets sur les yeux : troubles visuels, y compris une altération de la vision des couleurs ;
- effets sur le système immunitaire : réactions allergiques.
Déclaration des effets secondaires
Si vous ressentez un quelconque effet indésirable, parlez-en à votre médecin ou votre pharmacien, y compris tout effet indésirable éventuel non mentionné dans cette notice. Vous pouvez également signaler les effets indésirables directement via le système national de déclaration à l'adresse https://www.aifa.gov.it/content/segnalazioni-reazioni-avverse. En signalant les effets indésirables, vous pouvez contribuer à fournir plus d'informations sur la sécurité de ce médicament.
Expiration et conservation
Gardez ce médicament hors de la vue et de la portée des enfants
N'utilisez pas ce médicament après la date de péremption indiquée sur la boîte après « Expire le ». La date d'expiration fait référence au dernier jour de ce mois.
Ne jetez aucun médicament au tout-à-l'égout ou avec les ordures ménagères.Demandez à votre pharmacien comment jeter les médicaments que vous n'utilisez plus.Cela contribuera à protéger l'environnement.
Ce que contient UGUROL
L'ingrédient actif est l'acide tranexamique.
Chaque flacon de 5 ml contient 500 mg d'acide tranexamique. L'autre composant est l'eau pour préparations injectables.
A quoi ressemble UGUROL et contenu de l'emballage extérieur
UGUROL 500 mg/5ml solution injectable pour voie intraveineuse, à usage oral et local, boîte de 5 ampoules
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT
UGUROL 500 MG / 5 ML SOLUTION INJECTABLE POUR VOIE INTRAVEINEUSE POUR USAGE ORALE OU LOCAL
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE
une ampoule de 5 ml contient : principe actif : acide tranexamique 500 mg
Pour la liste complète des excipients, voir rubrique 6.1
03.0 FORME PHARMACEUTIQUE
Solution injectable pour usage intraveineux, à usage oral ou local.
04.0 INFORMATIONS CLINIQUES
04.1 Indications thérapeutiques
Prévention et traitement des saignements dus à une fibrinolyse généralisée ou locale chez l'adulte et l'enfant à partir d'un an.
Les indications spécifiques sont :
• saignements dus à une fibrinolyse généralisée ou locale tels que :
- ménorragie et métrorragie,
- saignements gastro-intestinaux,
- troubles de la coagulation urinaire, suite à une chirurgie prostatique ou à des interventions chirurgicales impliquant les voies urinaires ;
• Chirurgie ORL (adénoïdectomie, amygdalectomie, extractions dentaires) ;
• chirurgie gynécologique ou troubles obstétricaux ;
• chirurgie thoracique et abdominale et autres chirurgies majeures telles que la chirurgie cardiovasculaire ;
• prise en charge des saignements dus à l'administration d'un fibrinolytique.
04.2 Posologie et mode d'administration
Dosage
Adultes
- Prophylaxie orale
Si la solution est prise par voie orale, en diluant le contenu de l'ampoule avec un peu d'eau sucrée, la posologie journalière est de 1½-2 ampoules d'Ugurol de 500 mg, en commençant l'administration au moins 1 jour avant la chirurgie et en poursuivant le traitement pendant une période d'au moins 3-4 jours après l'opération.
- Thérapie orale
Si la solution est prise par voie orale, en diluant le contenu de l'ampoule dans un peu d'eau sucrée, la posologie journalière est de 1 à 2 ampoules d'Ugurol à 500 mg 3 fois par jour ou ½ à 1 ampoule d'Ugurol à 500 mg 6 fois par jour .
L'administration orale est particulièrement indiquée :
- dans les manifestations hémorragiques survenant en médecine interne, en oto-rhino-laryngologie et en dentisterie ;
- pour la préparation d'interventions chirurgicales dans lesquelles il est supposé que des saignements dus à l'activation plasminique peuvent survenir;
- dans l'hyperménorrhée ;
- dans les gemices gynécologiques, les cystites et les rectites hémorragiques consécutives à une radiothérapie pour carcinome génital ;
- pour le maintien des thérapies initiées par voie intraveineuse afin de prévenir la récidive hémorragique.
- Thérapie intraveineuse
Adultes
Sauf prescription contraire, les doses suivantes sont recommandées :
1. traitement standard de la fibrinolyse locale :
0,5 g (1 ampoule de 5 ml) à 1 g (2 ampoules de 5 ml) d'acide tranexamique en injection intraveineuse lente (= 1 ml/minute) deux ou trois fois par jour
2. traitement standard de la fibrinolyse généralisée :
1 g (2 ampoules de 5 ml) d'acide tranexamique en injection intraveineuse lente (= 1 ml/minute) toutes les 6-8 heures, soit 15 mg/kg de poids corporel.
Insuffisance rénale
En cas d'insuffisance rénale pouvant entraîner un risque d'accumulation, l'utilisation de l'acide tranexamique est contre-indiquée chez les patients présentant une insuffisance rénale sévère (voir rubrique 4.3). Chez les patients présentant une insuffisance rénale légère à modérée, la dose d'acide tranexamique doit être réduite en fonction du taux de créatinine sérique.
Insuffisance hépatique
Aucun ajustement posologique n'est nécessaire chez les patients présentant une insuffisance hépatique.
personnes agées
Il n'est pas nécessaire de réduire la dose sauf en cas d'insuffisance rénale avérée.
- Thérapie pour application locale
Le contenu de 1 ampoule est normalement utilisé, qui doit être versé directement sur le site du saignement ou appliqué avec une compresse de gaze préalablement imbibée.
L'application locale directe ou à l'aide de compresses de gaze préalablement trempées dans la solution est particulièrement indiquée pour les saignements au niveau buccal rhino-pharyngé pour lesquels une hémostase rapide est souhaitée.
Population pédiatrique
Les données sur l'efficacité, la posologie et la sécurité pour les indications actuellement approuvées telles que décrites dans la rubrique 4.1 sont limitées.
- Prophylaxie orale
Administrer la solution par voie orale à une dose quotidienne de 5 à 10 mg/kg en commençant l'administration au moins 1 jour avant l'opération et en poursuivant le traitement pendant une période d'au moins 3 à 4 jours après l'opération.
En cas de prise de la solution par voie orale, diluer le contenu du flacon avec un peu d'eau sucrée.
- Thérapie orale
Administrer la solution par voie orale à la dose de 10-20 mg/kg 3 fois par jour ou 5-10 mg/kg 6 fois par jour.
Pour prendre la solution voir "Prophylaxie".
- Thérapie intraveineuse
Chez l'enfant à partir d'un an, pour les indications actuellement approuvées décrites dans la rubrique 4.1, la dose est d'environ 20 mg/kg/jour. Cependant, il existe peu de données sur l'efficacité, la posologie et la tolérance pour ces indications.
Il n'existe pas d'évaluations exhaustives de l'efficacité, de la posologie et de l'innocuité de l'acide tranexamique chez les enfants subissant une chirurgie cardiaque. Les données actuellement disponibles sont limitées et sont présentées dans la rubrique 5.1.
Mode d'administration
a) Par voie intraveineuse
L'administration doit nécessairement avoir lieu par injection intraveineuse lente.
b) Par voie orale
Par voie orale, la solution est principalement indiquée chez l'enfant ou chez les patients ayant des difficultés à avaler.
c) Pour application locale
Instructions pour l'ouverture du flacon
Les flacons sont équipés d'une pré-ouverture de sécurité et doivent être ouverts comme suit :
- positionner le flacon comme indiqué sur la figure 1 ;
- exercer une pression avec le pouce placé sur le POINT DE COULEUR comme indiqué sur la figure 2.
04.3 Contre-indications
Hypersensibilité à la substance active ou à l'un des excipients mentionnés à la rubrique 6.1.
Thrombose veineuse ou artérielle aiguë (voir rubrique 4.4).
Affections fibrinolytiques dues à une coagulopathie de consommation sauf en cas d'activation prédominante du système fibrinolytique avec hémorragie aiguë sévère (voir rubrique 4.4).
Insuffisance rénale sévère (risque d'accumulation).
Histoire de convulsions.
Injection intrathécale et intraventriculaire, application intracérébrale (risque d'œdème cérébral et de convulsions).
04.4 Mises en garde spéciales et précautions d'emploi appropriées
Les indications ci-dessus et le mode d'administration doivent être strictement observés :
• les injections intraveineuses doivent être administrées lentement
• L'acide tranexamique ne doit pas être administré par voie intramusculaire.
Convulsions
Des cas de convulsions ont été rapportés en association avec le traitement à l'acide tranexamique. Dans la chirurgie de pontage aorto-coronarien (PAC), la plupart des cas sont survenus après une injection intraveineuse (IV) de fortes doses d'acide tranexamique. L'incidence des crises postopératoires était la même que chez les patients non traités.
Troubles visuels
Une attention particulière doit être portée aux troubles visuels possibles, y compris la déficience visuelle, la vision floue, la vision des couleurs altérée et, si nécessaire, le traitement doit être arrêté. En cas d'utilisation prolongée de la solution injectable d'acide tranexamique, des examens ophtalmologiques réguliers sont recommandés (examens de la vue comprenant l'acuité visuelle, la vision des couleurs, le fond d'œil, le champ visuel, etc.) En cas de modifications ophtalmologiques pathologiques, notamment avec des pathologies du rétine, le médecin doit décider, après consultation d'un spécialiste, de la nécessité d'une utilisation prolongée de la solution injectable d'acide tranexamique dans chaque cas individuel.
Hématurie
En cas d'hématurie des voies urinaires supérieures, il existe un risque d'obstruction urétrale.
Événements thromboemboliques
Avant d'utiliser l'acide tranexamique, les facteurs de risque de maladie thromboembolique doivent être pris en compte. Chez les patients ayant des antécédents de maladie thromboembolique ou chez ceux ayant une « incidence élevée d'événements thromboemboliques dans les » antécédents familiaux (patients à haut risque de thrombophilie), Solution d'acide tranexamique pour injection ne doit être administré que sur indication expresse du médecin, après consultation d'un expert en hémostase et sous surveillance médicale étroite (voir rubrique 4.3).
L'acide tranexamique doit être administré avec prudence chez les patientes prenant des contraceptifs oraux en raison du risque accru de thrombose (voir rubrique 4.5).
Coagulation intravasculaire disséminée
Les patients présentant une coagulation intravasculaire disséminée (CIVD) dans la plupart des cas ne peuvent pas être traités par l'acide tranexamique (voir rubrique 4.3). Si une décision est prise d'administrer de l'acide tranexamique, cela ne doit être fait que chez les patients chez lesquels il existe une « activation prédominante de la système fibrinolytique avec saignement aigu sévère. Normalement, le profil hématologique se rapproche de ce qui suit : temps de lyse réduit du caillot d'euglobuline ; temps de prothrombine prolongé ; diminution des taux plasmatiques de fibrinogène, de facteurs V et VIII, de fibrinolysine de plasminogène et d'alpha-2 macroglobuline ; taux plasmatiques normaux de le complexe prothrombine, c'est-à-dire les facteurs II (prothrombine), VIII et X ; taux plasmatiques élevés de produits de dégradation du fibrinogène ; numération plaquettaire normale. Ce qui précède suppose que la maladie sous-jacente ne modifie pas pour elle-même les divers éléments de ce profil. Dans ces cas aigus cas une dose unique de 1 g d'acide tranexamique habituellement ou est-ce suffisant pour contrôler les saignements. L'administration d'acide tranexamique dans la CIVD ne doit être envisagée que si un équipement de laboratoire hématologique adéquat est disponible et en présence de personnel spécialisé.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions
Aucune étude d'interaction n'a été réalisée. Un traitement concomitant par anticoagulants ne peut avoir lieu que sous la surveillance étroite d'un médecin expérimenté dans ce domaine. Les médicaments agissant sur l'hémostase doivent être administrés avec prudence chez les patients traités par l'acide tranexamique. Il existe un risque théorique d'augmentation potentielle de la formation de thrombus, comme cela se produit avec les œstrogènes. En variante, l'action antifibrinolytique du médicament peut être antagonisée avec des médicaments thrombolytiques.
04.6 Grossesse et allaitement
Les femmes en âge de procréer doivent utiliser des contraceptifs efficaces pendant le traitement.
Grossesse
Les données cliniques sur l'utilisation de l'acide tranexamique chez la femme enceinte sont insuffisantes. Par conséquent, même si les études animales ne rapportent pas d'effets tératogènes, par précaution d'emploi, l'utilisation de l'acide tranexamique est déconseillée au cours du premier trimestre de la grossesse. Des données cliniques limitées sur l'utilisation de l'acide tranexamique dans diverses affections hémorragiques au cours des deuxième et troisième trimestres de la grossesse n'ont pas rapporté d'effet délétère sur le fœtus. L'acide tranexamique ne peut être utilisé pendant la grossesse que si les bénéfices attendus justifient le risque potentiel.
L'heure du repas
L'acide tranexamique étant excrété dans le lait maternel, l'allaitement n'est donc pas recommandé.
La fertilité
Il n'y a pas de données cliniques sur les effets de l'acide tranexamique sur la fertilité.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines
Aucune étude sur l'aptitude à conduire des véhicules et à utiliser des machines n'a été réalisée.
04.8 Effets indésirables
Les effets indésirables rapportés dans les essais cliniques et sur la base de l'expérience post-commercialisation sont listés ci-dessous par classe de système d'organe.
Tableau avec la liste des effets indésirables
Les effets indésirables rapportés sont inclus dans le tableau ci-dessous et sont répertoriés par classe de système d'organe primaire MedDRA. Au sein de chaque classe de systèmes d'organes, les effets indésirables sont classés par fréquence. Dans chaque catégorie de fréquence, les effets indésirables sont classés par ordre décroissant de gravité. Les catégories de fréquence sont définies comme suit : très fréquent (≥1/10) ; commun (≥1 / 100
Déclaration des effets indésirables suspectés
La déclaration des effets indésirables suspectés survenant après l'autorisation du médicament est importante car elle permet un suivi continu du rapport bénéfice/risque du médicament. Les professionnels de santé sont invités à déclarer tout effet indésirable suspecté via le système national de déclaration.
04.9 Surdosage
Aucun cas de surdosage n'a été rapporté.
Les signes et symptômes peuvent être des étourdissements, des maux de tête, une hypotension et des convulsions. Il a été démontré que les convulsions surviennent plus fréquemment avec l'augmentation de la dose.
La prise en charge du surdosage doit consister en des soins de soutien.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES
05.1 Propriétés pharmacodynamiques
Classe pharmacothérapeutique : antihémorragiques, antifibrinolytiques
Code ATC : B02AA02
L'acide tranexamique a une activité anti-hémorragique en inhibant les propriétés fibrinolytiques de la plasmine.
Un complexe est formé qui comprend l'acide tranexamique et le plasminogène; l'acide tranexamique se lie au plasminogène lorsqu'il est transformé en plasmine.
L'activité du complexe acide tranexamique-plasmine sur l'activité de la fibrine est inférieure à l'activité de la plasmine libre seule.
Éducation in vitro ont montré que des doses élevées d'acide tranexamique réduisaient l'activité du complément.
Population pédiatrique
Enfants à partir d'un an :
Dans la littérature, 12 études d'efficacité en chirurgie cardiaque pédiatrique ont été identifiées qui ont inclus 1073 enfants, 631 traités par acide tranexamique. La plupart des études étaient contrôlées contre placebo. La population étudiée était hétérogène en termes d'âge, de type de chirurgie et de schémas posologiques. Les résultats des études sur l'acide tranexamique indiquent moins de pertes de sang et moins de besoins en produits sanguins en chirurgie cardiaque pédiatrique avec circulation extracorporelle (CEC) lorsqu'il existe un risque élevé de saignement, en particulier chez les patients cyanosés ou chez les patients subissant des interventions chirurgicales répétées. Le schéma posologique le plus approprié s'est avéré être :
- premier bolus de 10 mg/kg après induction de l'anesthésie et avant incision cutanée,
- perfusion continue de 10 mg/kg/h ou injection dans le liquide d'amorçage de la pompe CPB à une dose adaptée à la procédure CPB, ou selon le poids du patient à une dose de 10 mg/kg, ou selon le volume d'amorçage de la pompe CPB, avec la dernière injection de 10 mg/kg à la fin de l'opération de circulation extracorporelle.
Bien que les études aient porté sur un nombre très limité de patients, les quelques données disponibles indiquent que la perfusion continue est préférable, car elle maintient les concentrations plasmatiques thérapeutiques tout au long de la chirurgie.
Aucune étude dose-effet et pharmacocinétique spécifique n'a été réalisée chez l'enfant.
05.2 Propriétés pharmacocinétiques
Absorption
Les concentrations plasmatiques maximales d'acide tranexamique sont atteintes rapidement après une courte perfusion intraveineuse, après quoi les concentrations plasmatiques diminuent de manière multi-exponentielle.
Distribution
La liaison aux protéines plasmatiques de l'acide tranexamique est d'environ 3 % aux taux plasmatiques thérapeutiques et semble être entièrement due à sa liaison au plasminogène. L'acide tranexamique ne se lie pas à l'albumine sérique. Le volume initial de distribution est d'environ 9 à 12 litres.
L'acide tranexamique traverse le placenta.Suite à l'administration d'une injection intraveineuse de 10mg/kg à 12 femmes enceintes, la concentration sérique d'acide tranexamique était comprise entre 10 et 53 mcg/ml, tandis que celle dans le sang du cordon ombilical était comprise entre 4 et 31 mcg/ ml. L'acide tranexamique diffuse rapidement dans le liquide synovial et la membrane synoviale.Après l'administration d'une injection intraveineuse de 10 mg/kg à 17 patients opérés du genou, les concentrations dans le liquide synovial étaient similaires à celles observées dans les échantillons de sérum correspondants. La concentration d'acide tranexamique dans un certain nombre d'autres tissus correspond à une fraction de celle observée dans le sang (un centième dans le lait maternel ; un dixième dans le liquide céphalo-rachidien ; un dixième dans l'humeur aqueuse). L'acide tranexamique a été détecté dans le sperme, où il inhibe l'activité fibrinolytique mais n'affecte pas la migration des spermatozoïdes.
Excrétion
Il est principalement excrété dans les urines sous forme inchangée.L'excrétion urinaire par filtration glomérulaire est la principale voie d'élimination. La clairance rénale est égale à la clairance plasmatique (110-116 ml/min). L'excrétion d'acide tranexamique est d'environ 90 % au cours des 24 premières heures suivant l'administration intraveineuse de 10 mg/kg de poids corporel. La demi-vie de l'acide tranexamique est d'environ 3 heures.
Populations particulières
Les concentrations plasmatiques augmentent chez les patients insuffisants rénaux.
Aucune étude pharmacocinétique spécifique n'a été réalisée chez l'enfant.
05.3 Données de sécurité précliniques
Les données non cliniques ne révèlent aucun danger spécifique chez l'homme sur la base des études conventionnelles de pharmacologie de sécurité, de toxicité à doses répétées, de génotoxicité, de potentiel cancérogène et de toxicité pour la reproduction.
Une activité épileptogène a été observée en cas d'utilisation intrathécale d'acide tranexamique chez l'animal.
06.0 INFORMATIONS PHARMACEUTIQUES
06.1 Excipients
Un flacon de 500 mg d'acide tranexamique / 5 ml contient :
eau pour préparations injectables.
06.2 Incompatibilité
Rien.
06.3 Durée de validité
5 années
06.4 Précautions particulières de conservation
Rien
06.5 Nature du conditionnement primaire et contenu de l'emballage
boite de 5 ampoules de 500 mg/5 ml
boîte de 6 ampoules de 500 mg/5 ml
06.6 Instructions d'utilisation et de manipulation
07.0 TITULAIRE DE L'AUTORISATION DE MISE SUR LE MARCHE
Rottapharm S.p.A. - Galleria Unione, 5 - 20122 Milan
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ
A.I.C. 021458029 - "500 mg/5 ml solution injectable pour voie intraveineuse, à usage oral et local" 5 ampoules
A.I.C. 021458031 - "500 mg/5 ml solution injectable pour voie intraveineuse, à usage oral et local" 6 ampoules
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION
Ugurol est sur le marché depuis mai 1970 / 31 mai 2005
10.0 DATE DE RÉVISION DU TEXTE
Décembre 2013