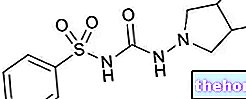
Ingrédients actifs : Tyrotricine, Bromure de cétrimonio, Benzocaïne
GOLAMIXIN® spray pour muqueuse buccale
Pourquoi Golamixin est-il utilisé? Pourquoi est-ce?
CATÉGORIE PHARMACOTHERAPEUTIQUE
Préparé pour la cavité oropharyngée - ATC R02A
INDICATIONS THÉRAPEUTIQUES
Thérapie topique de la stomatite bactérienne sensible à la thyrotricine.
Contre-indications Quand Golamixin ne doit pas être utilisé
Sujets présentant une hypersensibilité connue aux composants, en particulier à la thyrotricine.
Sujets dont la prédisposition marquée aux allergies est connue.
Précautions d'emploi Quelles sont les informations à connaître avant de prendre Golamixine
Chez la femme enceinte et dans la très petite enfance, le produit doit être administré en cas de besoin réel et sous le contrôle direct du médecin.
En cas d'absence de réponse dans les deux jours suivant le début de l'administration, interrompre le traitement pour une éventuelle colonisation de souches bactériennes ou de champignons (notamment Candida) résistants à la thyrotricine.L'utilisation, surtout si prolongée, de produits topiques peut entraîner une sensibilisation. phénomènes. Dans ce cas, la thérapie doit être interrompue et une thérapie appropriée instituée.
Un comportement similaire doit être observé en cas de développement de microorganismes non sensibles.
Interactions Quels médicaments ou aliments peuvent modifier l'effet de Golamixin
Compte tenu des faibles doses administrées, aucune interaction grave avec d'autres médicaments n'est attendue.
Avertissements Il est important de savoir que :
Gardez ce médicament hors de la portée et de la vue des enfants.
Pour ceux qui pratiquent des activités sportives, l'utilisation de médicaments contenant de l'alcool éthylique peut déterminer des tests de dopage positifs par rapport aux limites de concentration en alcool indiquées par certaines fédérations sportives.
Posologie et mode d'utilisation Comment utiliser Golamixin : Posologie
Adultes : 2-3 applications à la fois, 3-4 fois par jour.
Enfants : 1 à 2 applications à la fois, 3 à 4 fois par jour.
La distribution est dosée avec précision par une valve doseuse particulière.Appuyez sur le bouton bas pour obtenir une distribution dosée.
Surdosage Que faire si vous avez pris trop de Golamixine
Il n'y a pas de manifestations connues de toxicité par surdosage.
Effets secondaires Quels sont les effets secondaires de Golamixin
Avec l'utilisation de la thyrotricine, un noircissement de la langue, rarement une glossite et des réactions de sensibilisation ont été rapportés qui, cependant, régressent avec l'interruption du traitement. Les symptômes de sensibilisation aux anesthésiques, y compris les troubles musculaires, les convulsions, peuvent être plus pertinents.
Le patient est invité à signaler tout effet indésirable autre que ceux indiqués ci-dessus à son médecin ou son pharmacien.
Expiration et conservation
Golamixin doit être conservé dans un endroit frais.
Validité : 36 mois.
Attention : ne pas utiliser le médicament après la date de péremption indiquée sur l'emballage.
Délai "> Autres informations
COMPOSITION
100 ml contiennent :
- INGRÉDIENTS ACTIFS : Thyrotricine 0,02 g ; 0,05 g de bromure de cétrimone; Benzocaïne g 0,05.
- EXCIPIENTS : Huile essentielle de menthe poivrée ; De l'alcool.
FORMULAIRE PHARMACEUTIQUE
flacon "spray muqueux buccal" 10 ml.
Notice d'emballage source : AIFA (Agence italienne des médicaments). Contenu publié en janvier 2016. Les informations présentes peuvent ne pas être à jour.
Pour avoir accès à la version la plus récente, il est conseillé d'accéder au site Internet de l'AIFA (Agence Italienne du Médicament). Avis de non-responsabilité et informations utiles.
01.0 DÉNOMINATION DU MÉDICAMENT -
GOLAMIXINE
02.0 COMPOSITION QUALITATIVE ET QUANTITATIVE -
100 ml contiennent :
Ingrédients actifs : thyrotricine 0,02 g ; 0,05 g de bromure de cétrimone; benzocaïne g 0,05.
03.0 FORME PHARMACEUTIQUE -
Spray pour muqueuse buccale.
04.0 INFORMATIONS CLINIQUES -
04.1 Indications thérapeutiques -
Thérapie topique de la stomatite bactérienne sensible à la thyrotricine.
04.2 Posologie et mode d'administration -
Adultes : 2-3 applications à la fois, 3-4 fois par jour.
Enfants : 1 à 2 applications à la fois, 3 à 4 fois par jour.
04.3 Contre-indications -
Sujets présentant une hypersensibilité connue aux composants individuels, en particulier à la thyrotricine. Sujets dont la prédisposition marquée aux allergies est connue.
04.4 Mises en garde spéciales et précautions d'emploi appropriées -
Dans la toute petite enfance, le produit doit être administré en cas de besoin réel et sous le contrôle direct du médecin.
En cas d'absence de réponse dans les deux jours suivant le début de l'administration, interrompre le traitement pour l'éventuelle colonisation de souches ou de champignons (notamment Candida) résistants à la thyrotricine.L'utilisation, surtout si prolongée, de produits topiques peut donner lieu à des phénomènes de sensibilisation. . Dans ce cas, la thérapie doit être interrompue et une thérapie appropriée instituée. Un comportement analogue doit être observé en cas de développement de micro-organismes non sensibles.
Tenir hors de portée des enfants.
04.5 Interactions avec d'autres médicaments et autres formes d'interactions -
Compte tenu des faibles doses administrées, aucune interaction grave avec d'autres médicaments n'est attendue.
04.6 Grossesse et allaitement -
Chez la femme enceinte, le produit doit être administré en cas de besoin réel, et sous le contrôle direct du médecin.
04.7 Effets sur l'aptitude à conduire des véhicules et à utiliser des machines -
L'administration de Golamixin n'affecte pas la durée d'attention, il n'y a donc aucun obstacle à la conduite et à l'utilisation de machines de précision.
04.8 Effets indésirables -
Avec l'utilisation de la thyrotricine, un noircissement de la langue, rarement une glossite et des réactions de sensibilisation ont été rapportés qui, cependant, régressent avec l'interruption du traitement. Les symptômes de sensibilisation aux anesthésiques, y compris les troubles musculaires, les convulsions, peuvent être plus pertinents.
04.9 Surdosage -
Il n'y a pas de manifestations connues de toxicité par surdosage.
05.0 PROPRIÉTÉS PHARMACOLOGIQUES -
05.1 "Propriétés pharmacodynamiques -
La golamixine est constituée par l'association d'un antibiotique, la thyrotricine, avec un désinfectant, le cétrimide, et un anesthésique local, la benzocaïne.Les tests pharmacologiques réalisés avec la Golamixine ont confirmé que la spécialité possède et exerce les activités déjà connues exercées par l'individu actif. ingrédients qu'il contient.
05.2 "Propriétés pharmacocinétiques -
Les ingrédients actifs contenus dans Golamixin n'interfèrent pas avec la flore bactérienne intestinale et ne sont pas absorbés par l'organisme à des concentrations telles qu'elles déterminent une activité microbienne systémique.
05.3 Données de sécurité préclinique -
Des tests de toxicité aiguë réalisés chez le rat et la souris par voie orale et sous-cutanée ont fourni des valeurs de DL50 supérieures respectivement à 50 mg/kg et 20 mg/kg, conférant au produit une marge de sécurité importante. Le traitement chronique pendant 24 semaines a été bien toléré par l'organisme des rats et des chiens traités par voie orale.Le traitement par la golamixine n'a pas affecté la fertilité des rats et des lapins traités par voie orale, et n'a pas non plus montré de toxicité maternelle et d'activité tératogène. pas d'effets circulatoires aigus appréciables chez le lapin anesthésié.
06.0 INFORMATIONS PHARMACEUTIQUES -
06.1 Excipients -
Huile essentielle de menthe poivrée ml 0,25; alcool au goût à 100 ml.
06.2 Incompatibilité "-
Il n'y a pas d'incompatibilités connues avec d'autres médicaments.
06.3 Durée de validité "-
3 ans dans un emballage intact.
06.4 Précautions particulières de conservation -
Rien.
06.5 Nature du conditionnement primaire et contenu de l'emballage -
Flacon en verre neutre, type III, fermé avec pompe doseuse et équipé d'un doseur oral.
Vaporisateur 10 ml
06.6 Instructions d'utilisation et de manipulation -
07.0 TITULAIRE DE « L'AUTORISATION DE MISE SUR LE MARCHE » -
Teofarma S.r.l. - via F.lli Cervi, 8 -27010 Valle Salimbene (PV)
08.0 NUMÉRO D'AUTORISATION DE MISE SUR LE MARCHÉ -
Spray bucco-muqueux flacon 10 ml - A.I.C. 016703035 -
09.0 DATE DE PREMIÈRE AUTORISATION OU DE RENOUVELLEMENT DE L'AUTORISATION -
Octobre 1960 / Juin 2010
10.0 DATE DE REVISION DU TEXTE -
juin 2010